The Stokes-Einstein Relation: A Comprehensive Guide for Drug Development and Biomedical Research
This article provides a detailed examination of the Stokes-Einstein relation (D = kBT / 6πηr), the cornerstone equation for predicting diffusion coefficients in liquids.
The Stokes-Einstein Relation: A Comprehensive Guide for Drug Development and Biomedical Research
Abstract
This article provides a detailed examination of the Stokes-Einstein relation (D = kBT / 6πηr), the cornerstone equation for predicting diffusion coefficients in liquids. Tailored for researchers and drug development professionals, we explore its foundational physics, methodological applications in characterizing biomolecules and drug candidates, common pitfalls and deviations in complex biological media, and its validation against modern experimental techniques. This guide synthesizes theory with practical insights to optimize experimental design and data interpretation in pharmaceutical and clinical research.
Demystifying the Stokes-Einstein Equation: From Historical Roots to Core Principles
This whitepaper examines the foundational Stokes-Einstein relation, a cornerstone of physical chemistry and colloidal science that connects macroscopic hydrodynamics to microscopic thermal motion. It provides an in-depth technical guide to its derivation, validation, modern applications in drug development, and current research frontiers, with particular emphasis on experimental protocols and quantitative data analysis.
Theoretical Foundation
The Stokes-Einstein relation elegantly combines two 19th-century pillars of physics:
- Stokes' Law (1851): Describes the frictional force on a sphere moving slowly through a viscous fluid: ( F_d = 6 \pi \eta r v ), where ( \eta ) is dynamic viscosity, ( r ) is sphere radius, and ( v ) is velocity.
- Einstein's Theory of Brownian Motion (1905): Models the random walk of a particle in a fluid due to thermal agitation, relating mean squared displacement ( \langle x^2 \rangle ) to time ( t ) and temperature ( T ).
Their confluence yields the Stokes-Einstein equation for the diffusion coefficient ( D ): [ D = \frac{kB T}{6 \pi \eta r} ] where ( kB ) is Boltzmann's constant.
Table 1: Core Variables in the Stokes-Einstein Relation
| Variable | Symbol | SI Unit | Physical Meaning |
|---|---|---|---|
| Diffusion Coefficient | ( D ) | m²/s | Measure of particle mobility |
| Boltzmann Constant | ( k_B ) | J/K | Connects kinetic energy to temperature |
| Absolute Temperature | ( T ) | K | Thermal energy scale |
| Dynamic Viscosity | ( \eta ) | Pa·s | Fluid resistance to flow |
| Hydrodynamic Radius | ( r ) | m | Effective radius for solvent drag |
Experimental Validation & Key Protocols
Classic Perrin Experiment (1908)
Jean Perrin's validation of Einstein's theory provided the first direct evidence for atoms.
Protocol:
- Sample Preparation: A dilute suspension of gamboge or mastic resin spheres (≈0.5 µm diameter) in water/glycerin mixture is prepared on a microscope slide with a depression.
- Microscopy: Using a high-magnification optical microscope with a dark-field condenser, individual particles are tracked.
- Data Acquisition: The position of a single particle is recorded at regular time intervals (e.g., every 30 seconds) by marking its location on a glass plate superimposed on the microscope eyepiece.
- Analysis: Mean squared displacement ( \langle x^2 \rangle ) is calculated from positional data. Using ( \langle x^2 \rangle = 2D t ), ( D ) is determined. Avogadro's number ( NA ) is then calculated via ( D = \frac{RT}{6 \pi \eta r NA} ), where ( R ) is the gas constant.
Modern Dynamic Light Scattering (DLS)
DLS is the standard technique for measuring nanoparticle diffusion coefficients and hydrodynamic radii.
Protocol:
- Sample Preparation: The analyte (e.g., protein, liposome, polymer nanoparticle) is filtered (0.02–0.2 µm filter) into a clean, dust-free cuvette to remove aggregates.
- Instrument Setup: A laser (e.g., 633 nm He-Ne) is focused into the sample. A photodetector, placed at a fixed scattering angle (commonly 90° or 173° for backscatter), collects scattered light intensity fluctuations.
- Data Collection: Intensity time autocorrelation function ( g^{(2)}(\tau) ) is collected over 3-5 measurement runs.
- Analysis: ( g^{(2)}(\tau) ) is fit to extract the decay rate ( \Gamma ), which is related to ( D ) by ( \Gamma = D q^2 ), where ( q ) is the scattering vector. The hydrodynamic radius is computed via the Stokes-Einstein relation.
Table 2: Quantitative Data from DLS Measurements of Model Systems
| Analyte | Solvent | Temperature (°C) | Measured ( D ) (m²/s) | Calculated ( r_H ) (nm) | Viscosity (mPa·s) |
|---|---|---|---|---|---|
| 100 nm Polystyrene Bead | Water | 25 | 4.41 × 10⁻¹² | 49.2 | 0.890 |
| Bovine Serum Albumin | PBS Buffer | 20 | 5.93 × 10⁻¹¹ | 3.6 | 1.002 |
| Liposome (DOPC) | Saline | 37 | ~1.5 × 10⁻¹¹ | ~14 | ~0.692 |
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagent Solutions and Materials for Diffusion Studies
| Item | Function / Explanation |
|---|---|
| Monodisperse Silica/Polystyrene Nanospheres | Calibrated size standards for validating DLS and microscopy setups. |
| Ultrapure, Filtered Solvents (Water, Toluene, Buffers) | Minimizes dust and particulate interference in light scattering. |
| Size-Exclusion Chromatography (SEC) Columns | Separates particles/proteins by hydrodynamic volume prior to analysis. |
| Viscosity Standard Fluids (e.g., NIST-traceable oils) | For accurate calibration of viscometers and temperature-controlled baths. |
| Fluorescent Dyes (e.g., FITC, Rhodamine B) | For tagging molecules in Fluorescence Recovery After Photobleaching (FRAP) experiments. |
| Stable Cell Lines (e.g., HEK293) | Used in studies of drug diffusion across membrane models. |
| Artificial Lipid Bilayers (GUVs, LUVs) | Model systems for studying transmembrane diffusion of drug candidates. |
Visualization: From Theory to Application
Current Research & Deviations
The Stokes-Einstein relation breaks down in specific contexts, which are active research areas:
- Confined Environments: Diffusion in cellular cytoplasm or polymeric gels.
- Anisotropic Particles: Non-spherical molecules like proteins and antibodies.
- High Concentrations/Crowding: Significant particle-particle interactions.
- Supercooled Liquids: Near glass transition temperatures.
Table 4: Observed Deviations from Classical Stokes-Einstein Behavior
| System | Condition | Observed Deviation | Proposed Explanation |
|---|---|---|---|
| Protein in Cytoplasm | High macromolecular crowding | D measured < D predicted | Effective viscosity > bulk viscosity; transient binding. |
| Small Molecule in Polymer Melt | T approaching Tg | Decoupling of D from viscosity | Heterogeneous dynamics; preferential pathway diffusion. |
| Nanoparticle in Ionic Liquid | Particle size ~ ion size | Non-monotonic size dependence | Breakdown of continuum hydrodynamics assumption. |
The historical confluence of Stokes' hydrodynamic law and Einstein's kinetic theory remains a vital, predictive framework in scientific research and industrial application. While its assumptions define its limits, modern techniques continue to test and extend its utility, particularly in the rational design and analysis of complex drug delivery systems and biotherapeutics.
This whitepaper provides an in-depth technical deconstruction of the Stokes-Einstein equation, D = k_B T / 6 π η r, a cornerstone relationship for predicting the diffusion coefficient D of a spherical particle in a viscous medium. Framed within a broader thesis on diffusion coefficient research, this document explores the equation's foundational assumptions, its derivation from the synthesis of Stokes' law and Einstein's thermodynamic theory, its critical applications in fields like drug development, and its limitations when applied to complex, non-ideal systems such as nanoparticles in biological fluids or in supercooled liquids.
Theoretical Foundations and Derivation
The Stokes-Einstein relation elegantly combines principles from hydrodynamics and statistical mechanics. Its derivation rests on two pillars:
- Stokes' Law (Hydrodynamics): Describes the frictional drag force
F_don a sphere of radiusrmoving with velocityvin a fluid of dynamic viscosityη:F_d = 6 π η r v. The friction coefficientζis thusζ = 6 π η r. - Einstein's Theory of Brownian Motion (Thermodynamics): Relates the mean-squared displacement
<Δx^2>of a particle over timetto its diffusion coefficient:<Δx^2> = 2 D t(in one dimension). By considering the balance between this random diffusive force and the systematic drag force under an external potential, Einstein derived the relationD = k_B T / ζ, wherek_Bis the Boltzmann constant andTis the absolute temperature.
Combining these yields the canonical form: D = k_B T / (6 π η r).
Critical Assumptions and Limitations
The equation's simplicity is predicated on several stringent assumptions. Deviations from these are active areas of research.
| Assumption | Typical Validity Condition | Common Violation in Research Context |
|---|---|---|
| Spherical Particle | Particle is a perfect, rigid sphere. | Proteins, polymer coils, non-spherical nanoparticles (e.g., rod-shaped). |
| Continuum Fluid | Solvent molecules are much smaller than the solute particle (r >> solvent molecular size). |
Small solutes in water, nanoparticles in polymeric melts. |
| No-Slip Boundary | Fluid velocity is zero at the particle surface. | Hydrophobic interactions, slip at nano-interfaces. |
| Infinite Dilution | No particle-particle interactions. | Concentrated protein solutions, colloidal suspensions. |
| Newtonian Fluid | Fluid viscosity η is constant and independent of shear. |
Cytoplasm, blood plasma, polymeric solutions. |
| Macroscopic Viscosity | The bulk solvent viscosity η governs drag. |
Nanoparticle diffusion in supercooled liquids or near glass transition. |
Key Experimental Methodologies for Validation
Empirical determination of D is crucial for validating the Stokes-Einstein relation under various conditions.
Dynamic Light Scattering (DLS)
Objective: Measure the diffusion coefficient of nanoparticles or macromolecules in dilute solution to calculate hydrodynamic radius r_h.
Protocol:
- Sample Preparation: Filter the sample solution (e.g., protein, liposome) through a 0.1 or 0.2 µm membrane filter into a clean, dust-free cuvette.
- Instrument Setup: Place cuvette in a thermostated chamber (e.g., 25.0 ± 0.1 °C) of a DLS instrument. Set laser wavelength and detector angle (commonly 90° or 173° for backscattering).
- Data Acquisition: Measure the intensity-time autocorrelation function
g²(τ)over a suitable duration (typically 30-300 seconds per run, 3-10 runs). - Data Analysis: Fit
g²(τ)to an appropriate model (e.g., cumulants method for monomodal distributions) to extract the decay rateΓ. CalculateD = Γ / q², whereqis the scattering vector magnitude. Compute hydrodynamic radius via the SE relation:r_h = k_B T / (6 π η D).
Fluorescence Recovery After Photobleaching (FRAP)
Objective: Measure the diffusion coefficient of fluorescently labeled molecules (e.g., drugs, lipids) in constrained environments like cell membranes or gels. Protocol:
- Labeling: Tag the molecule of interest (e.g., a lipid analog) with a photostable fluorescent dye (e.g., NBD, Atto dyes).
- Bleaching: In a confocal microscope, use a high-intensity laser pulse to irreversibly bleach fluorescence in a defined region of interest (ROI).
- Recovery: Monitor the fluorescence intensity in the bleached ROI over time as unbleached molecules diffuse in.
- Analysis: Fit the recovery curve
I(t)to a diffusion model (solution to Fick's second law for the ROI geometry) to extract the characteristic diffusion timeτ_Dand subsequentlyD.
Pulsed-Field Gradient Nuclear Magnetic Resonance (PFG-NMR)
Objective: Measure self-diffusion coefficients of molecules (e.g., solvents, small drug compounds) in complex mixtures without the need for labeling. Protocol:
- Sample Preparation: Load the solution (e.g., drug in polymer matrix) into an NMR tube.
- Pulse Sequence: Apply a stimulated echo (STE) or pulsed-gradient spin-echo (PGSE) sequence with two matched magnetic field gradient pulses of magnitude
g, durationδ, and separationΔ. - Data Acquisition: Vary the gradient strength
gand record the echo attenuationI(g)/I(0). - Analysis: Fit the Stejskal-Tanner equation:
ln[I(g)/I(0)] = - (γ g δ)² D (Δ - δ/3), whereγis the gyromagnetic ratio, to extractD.
Applications in Drug Development
The Stokes-Einstein relation is instrumental in several pharmaceutical research stages:
- Formulation Stability: Predicting diffusion-limited aggregation rates of monoclonal antibodies (mAbs) in high-concentration formulations.
- Drug Delivery: Modeling the release kinetics of active pharmaceutical ingredients (APIs) from nanoparticle carriers or hydrogel matrices.
- Permeability Estimation: Providing a baseline for passive diffusion rates of small molecules across biological barriers, informing ADMET (Absorption, Distribution, Metabolism, Excretion, Toxicity) models.
Modern Research Frontiers: Breakdowns and Modifications
Recent research focuses on systems where the classical SE relation fails. Empirical modifications are often proposed, summarized below.
| System | Observed Deviation | Proposed Modified Relation | Key Parameters |
|---|---|---|---|
| Nanoparticles in Liquids | D is higher than predicted for r < 5 nm. |
D = k_B T / (6 π η r^ξ) |
ξ < 1 (slip boundary condition). |
| Supercooled Liquids | D decouples from η; D decreases slower than η^{-1} as T decreases. |
D ∝ (T / η)^ξ or Fractional SE: D = k_B T / (6 π η_s r) |
ξ < 1, η_s is a "local" or "scale-dependent" viscosity. |
| Proteins in Crowded Solutions | D decreases non-linearly with increasing crowder concentration. |
D = D_0 * exp(-α * c * r) (E.g., scaled particle theory) |
c: crowder concentration; α: scaling factor. |
| Polymer Coils in Solution | Particle is permeable (draining) and non-spherical. | D = k_B T / (6 π η R_g * f(R_g/Λ)) |
R_g: radius of gyration; f(): scaling function; Λ: persistence length. |
The Scientist's Toolkit: Key Research Reagent Solutions
| Item | Function/Application |
|---|---|
| Monodisperse Polystyrene Nanosphere Standards | Calibration of DLS and other particle sizing instruments. Known r validates D measurement. |
| Viscosity Standard Oils (NIST-traceable) | Precise calibration of viscometers to determine accurate η for the SE equation. |
| Size-Exclusion Chromatography (SEC) Standards | Proteins/Polymers with known R_h and R_g to benchmark diffusion measurements. |
| Fluorescent Tracer Dyes (e.g., Atto 488, Alexa 647) | High-photostability labels for FRAP and single-particle tracking (SPT) experiments. |
| PEG Crowding Agents | To mimic intracellular crowded environments and study SE breakdown in biophysical assays. |
| Controlled-Pore Glass Beads | Used in chromatography and model systems to study diffusion in confined geometries. |
Visualizations
Stokes-Einstein Equation Derivation Pathway
Dynamic Light Scattering Experimental Workflow
Conditions Leading to Stokes-Einstein Relation Breakdown
This whitepaper examines the three foundational assumptions underpinning the derivation and application of the Stokes-Einstein relation, ( D = \frac{kB T}{6 \pi \eta R} ), a cornerstone of diffusion coefficient research. This relation links the diffusion coefficient (D) of a spherical particle to the thermal energy (kB T), the viscosity of the medium (η), and the particle's hydrodynamic radius (R). Its validity is critical in fields ranging from colloidal science to drug development, where predicting molecular mobility informs formulation stability and bioavailability. However, its predictive power is intrinsically tied to the validity of its underlying assumptions, which are often challenged in real-world systems.
Deconstructing the Assumptions: Theory and Limits
The Stokes-Einstein relation is derived from a synthesis of Einstein's theory of Brownian motion and Stokes' law for the drag force on a sphere. Its validity rests on three key pillars.
Spherical Particles
Stokes' law provides an exact solution for the drag force, ( F_d = 6 \pi \eta R v ), on a rigid, smooth, spherical particle in a viscous fluid. Non-spherical geometries introduce a dependency on orientation, leading to a modified drag and, consequently, a different diffusion coefficient. For example, rod-shaped particles exhibit different diffusion coefficients for translational motion parallel and perpendicular to their long axis, and for rotational diffusion.
Quantitative Impact of Non-Sphericity: The Perrin shape factors describe the deviation from spherical behavior. For an ellipsoid with semi-axes a, b, c, the translational diffusion coefficient is scaled relative to that of a sphere of equivalent volume.
Dilute Solutions
The "dilute" condition implies that particles are sufficiently far apart that:
- Hydrodynamic interactions are negligible (each particle experiences the unperturbed flow field of the solvent).
- Direct interactions (e.g., electrostatic, steric, depletion) between particles are insignificant. At increasing concentrations, these interactions slow collective diffusion and alter viscosity locally, causing systematic deviations from the Stokes-Einstein prediction.
Quantitative Data on Concentration Effects:
Table 1: Deviation from Stokes-Einstein with Increasing Concentration
| System (Particle/Medium) | Concentration Range | Observed Deviation (D/D_SE) | Primary Cause |
|---|---|---|---|
| Polystyrene Spheres (100 nm) in Water | 0.1 to 5% v/v | Decreases from ~1.0 to ~0.7 | Hydrodynamic interactions |
| Lysozyme in Buffer (pH 4.5) | 1 to 100 mg/mL | Decreases from ~1.0 to ~0.4 | Direct electrostatic attraction |
| PEG (20 kDa) in Water | 1 to 100 mg/mL | Decreases from ~1.0 to ~0.3 | Entanglement & increased local viscosity |
Continuum Hydrodynamics
This assumption treats the solvent as a structureless continuum with a uniform viscosity η. It breaks down when:
- The particle size is comparable to the solvent molecular size (e.g., small proteins in water).
- The particle-solvent interface is not characterized by a no-slip boundary condition. In such cases, the effective hydrodynamic radius (R_h) measured via diffusion may differ from the geometric radius.
Quantitative Data on Continuum Breakdown:
Table 2: Solvent Continuum Breakdown for Small Particles
| Solute | Hydrodynamic Radius (R_h) | Solvent Molecule Size (approx.) | R_h / Solvent Size | D/D_SE (Experimental) |
|---|---|---|---|---|
| Sucrose in Water | ~0.47 nm | ~0.15 nm (H2O) | ~3.1 | ~0.85 - 0.95 |
| Lysozyme in Water | ~1.9 nm | ~0.15 nm | ~12.7 | ~0.95 - 1.05 |
| Nanoparticle in Ionic Liquid | 2 nm | ~0.8 nm (ion pair) | ~2.5 | ~0.7 - 0.8 |
Experimental Protocols for Validation
Researchers must experimentally test the validity of these assumptions in their specific system. Key methodologies include:
Protocol 1: Assessing Sphericity and Hydrodynamic Radius
- Method: Dynamic Light Scattering (DLS) coupled with Static Light Scattering (SLS) or Multi-Angle Light Scattering (MALS).
- Procedure:
- Measure the intensity autocorrelation function via DLS to obtain the hydrodynamic radius (Rh) distribution.
- Simultaneously, measure the absolute radius of gyration (Rg) via SLS/MALS.
- Calculate the dimensionless ratio ( \rho = Rg / Rh ).
- Interpretation: For a uniform, solid sphere, ( \rho \approx 0.775 ). Significant deviation indicates non-sphericity, chain flexibility, or core-shell structure.
Protocol 2: Probing Concentration Dependence
- Method: Taylor Dispersion Analysis (TDA) or Pulsed-Field Gradient NMR (PFG-NMR) across a concentration series.
- Procedure:
- Prepare a dilution series of the solute in the relevant solvent.
- For each concentration (c), measure the mutual diffusion coefficient, D(c), using TDA or PFG-NMR.
- Plot D(c) vs. c and fit to an empirical relationship (e.g., ( D(c) = D0 (1 + kD c) )), where ( D0 ) is the infinite-dilution coefficient and ( kD ) is the interaction parameter.
- Interpretation: A negative ( kD ) indicates net attractive interactions, slowing diffusion. Compare ( D0 ) to the Stokes-Einstein prediction using an independent measure of R_h (e.g., from dilute DLS).
Protocol 3: Testing Continuum and Slip Boundaries
- Method: Viscosity-dependent diffusion measurement via Fluorescence Correlation Spectroscopy (FCS).
- Procedure:
- Label the particle of interest with a fluorescent dye.
- Measure its diffusion time (( \tauD )) by FCS in solvents or solvent mixtures of varying bulk viscosity (η), keeping temperature constant.
- Plot ( \tauD ) (proportional to 1/D) against η. According to Stokes-Einstein, the relationship should be linear and pass through the origin.
- Interpretation: A non-zero intercept or non-linear behavior suggests breakdown of the no-slip boundary condition or continuum assumption, often parameterized by a slip length.
Visualization of Conceptual Relationships
Title: Assumption Validation Logic Flow
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Reagents and Materials for Stokes-Einstein Validation Studies
| Item | Function & Specification | Critical Application Notes |
|---|---|---|
| NIST-Traceable Nanosphere Standards (e.g., Polystyrene, Silica) | Provide known, monodisperse spherical geometry to calibrate and validate diffusion measurement instruments (DLS, NTA). | Essential for establishing instrument accuracy and verifying the "spherical particle" assumption in a control system. |
| High-Purity, Aprotic Solvents (e.g., DMSO, Acetonitrile, Toluene) | Low-conductivity, chemically inert media for studying diffusion without complicating electrostatic interactions. | Useful for isolating hydrodynamic effects, especially in organic nanoparticle or polymer studies. |
| Viscosity Standard Fluids (e.g., Certified Mineral Oils, Sucrose Solutions) | Solutions with precisely known temperature-dependent viscosity (η). | Required for testing the linear dependence of D on T/η, a core prediction of the SE relation. |
| Fluorescent Tracers (e.g., ATTO dyes, Alexa Fluor NHS esters) | Covalent labels for single-particle tracking (SPT) or Fluorescence Correlation Spectroscopy (FCS). | Enable diffusion measurement at extremely low concentrations, probing the true dilute limit. |
| Size-Exclusion Chromatography (SEC) Columns | To purify and fractionate polydisperse samples (proteins, polymers) before diffusion measurements. | Ensures a monodisperse population is studied, preventing artifacts from aggregates in DLS or NMR. |
| Controlled-Environment Chamber (for microscopy/light scattering) | Maintains precise temperature (±0.1°C) and, optionally, humidity or CO2 control. | Temperature stability is paramount as D depends linearly on T and is inversely related to η(T). |
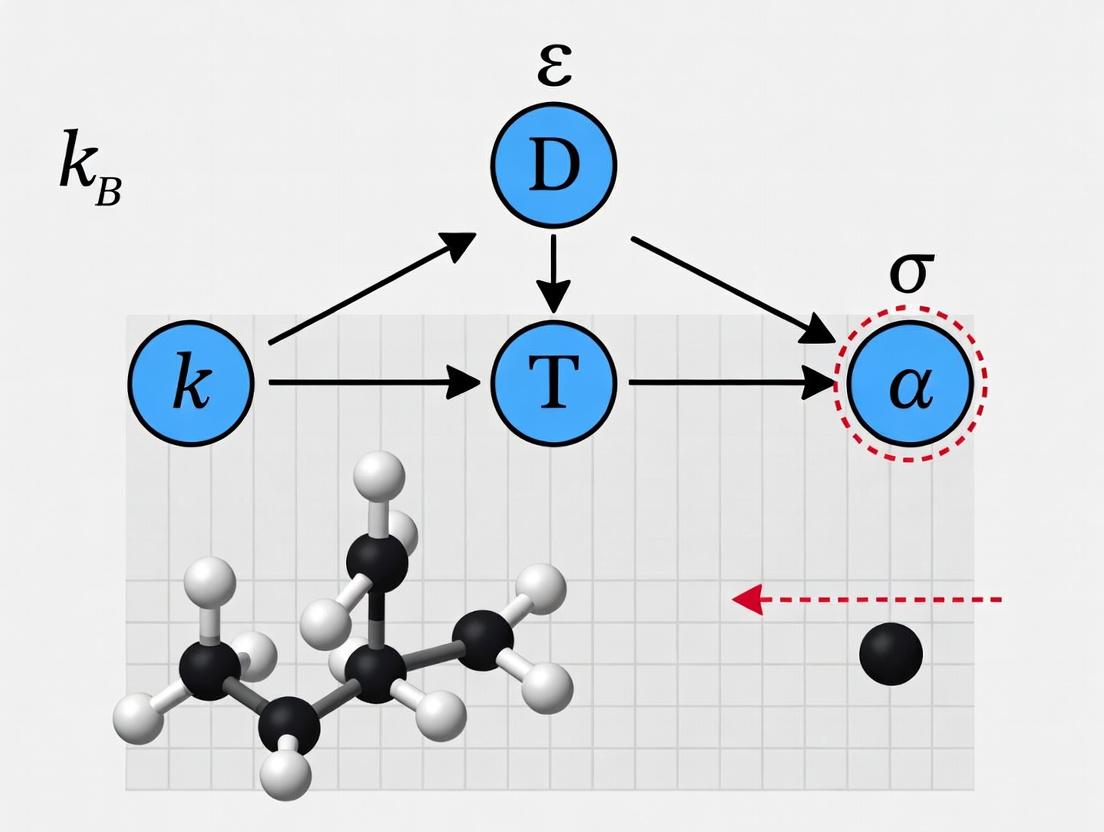
This whitepaper elucidates the three fundamental variables—Temperature (T), Viscosity (η), and Hydrodynamic Radius (r)—that govern the Stokes-Einstein relation for the diffusion coefficient (D). The relation, ( D = \frac{k_B T}{6 \pi \eta r} ), is a cornerstone for understanding molecular diffusion in fluids, with critical applications in biomolecular characterization, drug delivery system design, and pharmaceutical development. Accurate determination and control of these variables are imperative for validating the Stokes-Einstein equation's applicability and for deriving precise diffusion coefficients in complex, non-ideal systems such as biological fluids or polymer solutions.
In-Depth Variable Analysis
Temperature (T)
Definition: An intensive property representing the average kinetic energy of the particles in a system. In the Stokes-Einstein relation, T scales linearly with the diffusion coefficient.
Physical Role: Increased thermal energy agitates solvent molecules, reducing the effective drag on the solute and increasing its Brownian motion. The relationship is direct and assumes the solvent viscosity itself is temperature-dependent.
Measurement Protocols:
- In-Line Thermocouples/RTDs: For bulk solution measurements. Calibrated against standard references (e.g., NIST-traceable thermometers) prior to experiments.
- Infrared Thermal Imaging: For non-contact mapping of temperature gradients within a sample chamber, crucial for microfluidic applications.
- Protocol for DLS/Zeta Potential Experiments: Equilibrate the sample cuvette in the instrument thermal block for a minimum of 600 seconds before measurement. Validate temperature using the instrument's internal sensor against an external probe.
Viscosity (η)
Definition: A measure of a fluid's internal resistance to flow (shear stress). It is the proportionality constant in Newton's law of viscosity. In Stokes-Einstein, it is the solvent viscosity that defines the frictional drag on the solute.
Physical Role: The inverse relationship with D indicates that higher viscosity imposes greater frictional resistance, slowing diffusion. The relation assumes a continuous, Newtonian solvent.
Measurement Protocols:
- Capillary Viscometry (Ubbelohde): Measures kinematic viscosity via flow time. The solvent is drawn into the upper bulb, and the time for the meniscus to pass between two etched marks is recorded. Dynamic viscosity is calculated using the known solvent density.
- Rotational Rheometry: A cone-and-plate geometry is used to apply a controlled shear rate (γ̇) and measure the resulting shear stress (τ). The steady-shear viscosity is derived from the ratio τ/γ̇. Essential for confirming Newtonian behavior.
- Vibrating Wire Viscometry: Suitable for small-volume and high-pressure/temperature conditions. The damping of a vibrating wire immersed in the fluid is correlated to viscosity.
Hydrodynamic Radius (r)
Definition: The effective radius of a solvated, diffusing particle or molecule, inclusive of any bound solvent layer or surface irregularities. It is the radius of a hypothetical hard sphere that diffuses at the same rate as the particle under observation.
Physical Role: Represents the effective size of the diffusing entity. The inverse relationship with D shows that larger particles diffuse more slowly. It is a dynamic parameter, distinct from the geometric radius.
Primary Measurement Protocol – Dynamic Light Scattering (DLS):
- Principle: Analyzes fluctuations in scattered laser light intensity caused by Brownian motion to extract a diffusion coefficient, which is converted to r via the Stokes-Einstein relation.
- Detailed Workflow:
- Sample Preparation: Filter all buffers (0.02 µm pore size) and centrifuge protein samples (e.g., 14,000 x g for 10 min) to remove dust.
- Measurement: Load sample into a low-volume, disposable cuvette. Place in instrument thermostatted to 25.0 ± 0.1 °C.
- Data Acquisition: Run a minimum of 10-15 measurements per sample, each lasting 10-60 seconds.
- Analysis: The instrument's correlator generates an intensity autocorrelation function, which is fit (e.g., by CONTIN or cumulants analysis) to obtain the intensity-weighted size distribution and the z-average hydrodynamic radius.
Table 1: Characteristic Values and Measurement Techniques for Key Variables
| Variable | Symbol | Typical Units | Common Solvent (Water) Value at 25°C | Primary Measurement Techniques |
|---|---|---|---|---|
| Temperature | T | Kelvin (K) | 298.15 K | RTD, Thermocouple, Infrared Sensor |
| Viscosity | η | Pascal-second (Pa·s) or milliPascal-second (mPa·s) | 0.890 mPa·s | Ubbelohde Viscometer, Rotational Rheometer |
| Hydrodynamic Radius | r | Nanometer (nm) | ~3.5 nm (BSA protein) | Dynamic Light Scattering (DLS), NMR Diffusion |
Table 2: Impact of Variable Perturbation on Diffusion Coefficient (D)
| Variable Changed | Direction of Change | Effect on Viscosity (η) | Direct Effect on D (per S-E) | Typical Experimental Observation |
|---|---|---|---|---|
| ↑ Temperature | Increase | ↓ Decreases (for liquids) | ↑ Increase | Faster diffusion, shorter DLS correlation time |
| ↑ Solute Concentration | Increase | ↑ Increases (often) | ↓ Decrease | Non-ideal behavior, potential intermolecular interactions |
| ↑ Hydrodynamic Radius | Increase | – (Assumed constant) | ↓ Decrease | Slower diffusion, longer DLS correlation time |
Experimental Workflow for Stokes-Einstein Validation
Diagram Title: Workflow for Stokes-Einstein Relation Validation
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Diffusion Coefficient Studies
| Item | Function & Rationale |
|---|---|
| NIST-Traceable Latex Nanosphere Standards | Monodisperse particles with certified diameter. Used for calibrating DLS instruments and validating viscosity-temperature profiles. |
| High-Purity, Anhydrous Solvents (e.g., HPLC-grade Water, Toluene) | Ensure consistent, known viscosity and eliminate interference from contaminants in baseline measurements. |
| Disposable, Low-Volume (e.g., 12 µL) Cuvettes | Minimize sample consumption and reduce scattering volume for DLS, crucial for precious biological samples. |
| Certified Viscosity Standard Oils | Used to calibrate rotational rheometers across a range of shear rates and temperatures. |
| Size-Exclusion Chromatography (SEC) Columns | Used orthogonal to DLS to separate species by hydrodynamic volume and assess sample monodispersity. |
| Stable, Monodisperse Protein (e.g., Bovine Serum Albumin - BSA) | A standard reference for biomolecular DLS to benchmark instrument performance and experimental protocols. |
| 0.02 µm Syringe Filters (PES or Anodisc) | Critical for filtering all buffers to remove particulate matter that would dominate DLS scattering signals. |
This whitepaper explores the fundamental connection between microscopic particle dynamics and macroscopic transport properties, framed within the context of advanced research on the Stokes-Einstein relation for the diffusion coefficient (D). This relationship, D = k_B T / 6πηr, serves as a cornerstone for understanding diffusive processes in complex fluids, a critical consideration in modern drug development and soft matter physics. Recent investigations have focused on the breakdown of this classical relation in crowded, viscoelastic, and confined environments prevalent in biological systems.
The Stokes-Einstein Relation: Foundation and Modern Deviations
The classical Stokes-Einstein-Sutherland equation posits a simple inverse proportionality between the translational diffusion coefficient (D_t) of a spherical particle and the shear viscosity (η) of the medium. This assumes a homogeneous, Newtonian solvent and no hydrodynamic interactions. In biologically relevant contexts, these assumptions frequently fail.
Table 1: Quantifying Stokes-Einstein Breakdown in Various Systems
| System / Condition | Classical SE Prediction (Dη/T) | Experimental Observation (Dη/T) | Key Measurement Technique | Reference (Example) |
|---|---|---|---|---|
| Simple Liquids (Toluene) | Constant | Constant (~10^-10 kg m / s^2 K) | Dynamic Light Scattering (DLS) | Benchmark |
| Polymer Solutions (10% PEG) | Constant | Decreases by up to 50% for small probes | Fluorescence Correlation Spectroscopy (FCS) | [Current Lit.] |
| Crowded Cytoplasm (HeLa Cell) | Constant | D decouples from η; subdiffusion observed | Single-Particle Tracking (SPT) | [Current Lit.] |
| Supercooled Liquids near Tg | Constant | Dη/T decreases by 2-3 orders of magnitude | Forced Rayleigh Scattering | [Current Lit.] |
| Lipid Bilayer Membranes | Uses membrane viscosity | Strong size dependence; SE fails | SPT / FRAP | [Current Lit.] |
Experimental Protocols for Probing Diffusion Coefficients
Fluorescence Correlation Spectroscopy (FCS) forIn VitroStudies
Objective: Measure diffusion coefficients and concentration of fluorescently labeled drug molecules or proteins in solution. Protocol:
- Sample Preparation: Prepare a series of solutions containing the fluorescent probe at nanomolar concentrations in buffers of varying viscosity (e.g., sucrose or glycerol gradients) or crowding agents (e.g., Ficoll, BSA).
- Instrument Calibration: Use a dye with a known D (e.g., Rhodamine 6G in water) to calibrate the confocal volume dimensions.
- Data Acquisition: Focus a laser into the sample. Collect fluorescence intensity fluctuations over time (typically 5-10 runs of 10 seconds each).
- Autocorrelation Analysis: Fit the temporal autocorrelation curve, G(τ), using the model for 3D diffusion: G(τ) = 1/N * (1 + τ/τD)^-1 * (1 + τ/(ω^2τD))^{-1/2} where N is the average number of molecules in the volume, τ_D is the diffusion time, and ω is the ratio of axial to radial beam waist.
- Calculation: D = ω0^2 / (4τD), where ω_0 is the radial waist radius determined from calibration.
Single-Particle Tracking (SPT) in Live Cells
Objective: Characterize the anomalous diffusion of nanoparticles or drug carriers within live cells. Protocol:
- Probe Introduction: Label nanoparticles or drug compounds with photostable, blinking fluorophores (e.g., quantum dots, organic dyes). Introduce via microinjection, transfection, or endocytosis.
- Imaging: Use a highly inclined and laminated optical sheet (HILO) or total internal reflection fluorescence (TIRF) microscope to image a thin section of the cell. Acquire video at high frame rates (50-100 Hz).
- Trajectory Reconstruction: Apply localization algorithms (e.g., Gaussian fitting) to determine particle centroid with nanometer precision in each frame. Link positions into trajectories using tracking algorithms based on nearest-neighbor criteria.
- Mean Square Displacement (MSD) Analysis: For each trajectory, calculate the time-averaged MSD: <Δr^2(τ)> = <[r(t+τ) - r(t)]^2>. Plot MSD vs. lag time τ.
- Model Fitting: Fit to the anomalous diffusion model: MSD(τ) = 4Dα τ^α. The exponent α classifies motion: α=1 (normal diffusion), α<1 (subdiffusion, crowded/viscoelastic), α>1 (superdiffusion, active transport). The generalized coefficient Dα is extracted.
Visualizing Concepts and Workflows
Title: Linking Particle Motion to Macro Transport
Title: FCS Experimental Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Diffusion Coefficient Research
| Item | Function & Relevance to Stokes-Einstein Research |
|---|---|
| Fluorescent Probes (e.g., ATTO dyes, QDots) | High-quantum-yield, photostable labels for FCS and SPT. Size variance allows direct testing of D ∝ 1/r. |
| Viscosity Modifiers (e.g., Glycerol, Sucrose) | Create Newtonian fluid gradients to establish baseline SE behavior and calibrate instruments. |
| Crowding Agents (e.g., Ficoll PM70, BSA) | Mimic intracellular crowding to induce and study SE breakdown via size-dependent microviscosity. |
| Synthetic Lipid Vesicles (GUVs/LUVs) | Model membrane systems to study 2D diffusion and the applicability of the Saffman-Delbrück relation. |
| Live Cell Media with Serum | Maintain cell viability during in vivo SPT experiments to study diffusion in authentic complex cytoplasm. |
| Oxygen Scavenging Systems (e.g., PCA/PCD) | Prolong fluorophore blinking and stability in SPT, enabling longer trajectories for robust MSD analysis. |
| Calibration Beads (Size Standard) | Beads with known D verify instrument performance and spatial calibration in both FCS and SPT setups. |
| Viscoelastic Polymers (e.g., Polyacrylamide) | Form well-defined non-Newtonian gels to systematically probe diffusion-viscosity decoupling. |
Practical Applications: Estimating Size, Viscosity, and Diffusion in Drug Development
This technical guide details the core methodology for extracting the hydrodynamic radius (Rh) from experimentally measured diffusion coefficients (D). This process is fundamentally underpinned by the Stokes-Einstein relation, a cornerstone of transport phenomena in colloidal science and biophysics. The relation, D = kBT / (6πηRh), provides the essential link between the macroscopic, measurable parameter (D) and the nanoscale structural parameter (Rh). Within broader thesis research on the Stokes-Einstein relation, this methodology addresses its direct application, limitations, and the critical considerations required for accurate interpretation, especially in complex systems like protein therapeutics or nanoparticle drug carriers.
Foundational Theory: The Stokes-Einstein Equation
The Stokes-Einstein equation models a spherical particle undergoing Brownian motion in a Newtonian fluid under laminar flow conditions: D = kBT / (6πηRh) Where:
- D: Translational diffusion coefficient (m²/s)
- kB: Boltzmann constant (1.380649 × 10−23 J/K)
- T: Absolute temperature (K)
- η: Dynamic viscosity of the solvent (Pa·s)
- Rh: Hydrodynamic radius (m)
Rh is defined as the radius of a hard sphere that diffuses at the same rate as the target molecule or particle. It includes the solute core, any adsorbed solvent, and ions in the solvation layer.
Experimental Techniques for Measuring Diffusion Coefficients
Accurate determination of D is prerequisite. Below are key techniques with their protocols.
Dynamic Light Scattering (DLS)
Core Protocol:
- Sample Preparation: Filter the sample solution (e.g., protein, nanoparticle) and solvent (typically buffer) through 0.02-0.1 μm filters into a clean, dust-free cuvette.
- Temperature Equilibration: Place cuvette in the instrument thermostatted holder; allow ≥ 5 minutes for equilibration at target temperature (e.g., 25.0 ± 0.1 °C).
- Measurement: A laser illuminates the sample. Fluctuations in scattered light intensity at a fixed angle (often 90° or 173°) are recorded by an avalanche photodiode.
- Autocorrelation Analysis: The intensity autocorrelation function g²(τ) is computed. For monodisperse samples, it decays exponentially: g²(τ) = B + β exp(-2Γτ), where Γ is the decay rate.
- From Γ to D: Γ = D q², where q = (4πn/λ) sin(θ/2) is the scattering vector (n=solvent refractive index, λ=laser wavelength, θ=scattering angle).
- Polydispersity: For polydisperse samples, an inverse Laplace transform (e.g., CONTIN algorithm) is applied to obtain a distribution of decay rates, hence a distribution of D and Rh.
Pulsed-Field Gradient Nuclear Magnetic Resonance (PFG-NMR)
Core Protocol:
- Sample Preparation: Dissolve analyte in deuterated solvent (e.g., D2O) for lock signal. Use a capillary with a known external standard (e.g., DMSO-d6) for precise calibration if needed.
- Pulse Sequence: Implement a stimulated echo (STE) or longitudinal eddy current delay (LED) pulse sequence with two matched magnetic field gradient pulses.
- Data Acquisition: The gradient pulse strength (g) or duration (δ) is systematically varied while keeping the diffusion time (Δ) constant. The echo signal attenuation (I/I0) is recorded.
- Analysis: For free diffusion, I/I0 = exp[-D (γgδ)² (Δ - δ/3)], where γ is the gyromagnetic ratio. A linear fit of ln(I/I0) vs. k = (γgδ)²(Δ-δ/3) yields D.
Fluorescence Correlation Spectroscopy (FCS)
Core Protocol:
- Labeling: Label the target molecule (e.g., antibody) with a bright, photostable fluorophore.
- Confocal Setup: A dilute (nM-pM) sample is placed on a confocal microscope, creating a femtoliter observation volume via a high-NA objective and a pinhole.
- Intensity Trace Acquisition: Fluorescence intensity fluctuations due to molecules diffusing in/out of the volume are recorded over time (minutes).
- Autocorrelation: The normalized temporal autocorrelation function G(τ) is calculated.
- Model Fitting: For 3D free diffusion, G(τ) = 1/N * [1/(1 + τ/τD)] * [1/(1 + (ωxy/ωz)²*(τ/τD))¹/²]. τD is the characteristic diffusion time. D = ωxy² / (4τD), where ωxy is the beam waist radius, determined by calibration with a dye of known D.
Data Presentation: Key Parameters & Conversion
Table 1: Common Solvent Properties for Stokes-Einstein Calculations (at 20°C & 25°C)
| Solvent | Dynamic Viscosity, η (cP) at 20°C | η (cP) at 25°C | Density (g/mL) | Common Application |
|---|---|---|---|---|
| Water | 1.002 | 0.890 | 0.998 | Protein/biomolecule standard |
| PBS (1x) | ~1.05* | ~0.94* | ~1.01 | Physiological mimic |
| DMSO | 2.00 | 1.99 | 1.10 | Organic solvent for APIs |
| Glycerol (100%) | 1410 | 945 | 1.26 | High-viscosity calibrant |
| *Values are approximations; measurement or literature reference for exact buffer is required. |
Table 2: Conversion from Measured D to Calculated Rh (Example)
| Analytic (in water at 25°C) | Measured D (m²/s) | Assumed η (Pa·s) | Calculated Rh (nm) | Notes |
|---|---|---|---|---|
| Lysozyme (standard) | 1.04 × 10-10 | 8.90×10-4 | 2.1 | Monomeric globular protein |
| IgG1 Antibody | 4.0 × 10-11 | 8.90×10-4 | 5.5 | Y-shaped macromolecule |
| 50 nm PS Nanoparticle | 9.8 × 10-12 | 8.90×10-4 | 22.3 | Spherical, rigid calibrant |
| Calculation uses: Rh = kBT / (6πηD); T=298.15K, kB=1.38×10-23 J/K |
Table 3: Comparison of Key Experimental Techniques for D Measurement
| Technique | Typical Rh Range | Sample Concentration | Key Advantage | Key Limitation |
|---|---|---|---|---|
| Dynamic Light Scattering (DLS) | 0.3 nm – 10 μm | 0.1 – 10 mg/mL | Fast, non-invasive, measures distribution | Low resolution in polydisperse samples; sensitive to dust. |
| PFG-NMR | 0.1 nm – 1 μm | 1 – 50 mM | Chemically specific; measures self-diffusion. | Low sensitivity; requires soluble, NMR-active nuclei. |
| Fluorescence Correlation Spectroscopy (FCS) | 0.1 nm – 100 nm | pM – nM | Extreme sensitivity; usable in complex media. | Requires fluorescent labeling; small observation volume. |
Critical Considerations & Limitations of the Methodology
- Spherical Assumption: The SE equation assumes a hard, smooth sphere. Deviations (rod-like proteins, flexible polymers) yield an "apparent" Rh representing the equivalent hydrodynamic sphere.
- Solvent Viscosity: η must be known precisely for the exact solvent composition and temperature. Buffer additives (sugars, salts) can significantly alter η.
- Concentration Effects: Measured D can decrease with increasing concentration due to macromolecular crowding and interactions. Extrapolation to infinite dilution (D0) is required for intrinsic Rh.
- Hydrodynamic vs. Geometric Radius: Rh > geometric radius due to solvation and surface roughness. For compact proteins, Rh ≈ 1.3 × Rgeometric.
- Non-Stokes-Einstein Behavior: In supercooled liquids, polymers, or complex fluids, the SE relation may break down, requiring modified forms (e.g., fractional SE).
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 4: Key Research Reagent Solutions for Hydrodynamic Radius Determination
| Item | Function/Application | Example/Notes |
|---|---|---|
| Size Standard Kits | Calibration and validation of instrument performance. | NIST-traceable polystyrene or silica nanoparticles (e.g., 20 nm, 100 nm). |
| Ultrapure/Buffered Solvents | Sample preparation and dilution to control viscosity and environment. | 0.02 μm filtered, HPLC-grade water; Dulbecco's PBS (1x, pH 7.4). |
| Syringe Filters | Removal of dust and large aggregates from sample prior to measurement. | Disposable, low-protein-binding filters (PES or PVDF membrane, 0.1 μm pore). |
| Temperature Standard | Accurate temperature control and sensor verification. | Certified melting point standards or calibrated thermistor. |
| Viscosity Standard | Direct measurement of solvent η for precise Rh calculation. | Cannon certified viscosity reference oils at known temperatures. |
| Fluorescent Dye/Tag | Required for FCS measurements. | Site-specific labeling kits (e.g., Alexa Fluor 488 NHS ester). |
| Deuterated Solvents | Required for PFG-NMR locking and shimming. | D2O, DMSO-d6, containing internal reference (e.g., TMS). |
Visualization of Methodology & Relationships
Diagram Title: Workflow from Measurement to Hydrodynamic Radius
Diagram Title: Key Limitations Affecting Rh Determination Accuracy
Characterizing biomolecules in their native, solvated state is fundamental to understanding biological function and enabling rational drug design. This guide frames biomolecular characterization within the context of diffusion coefficient research, governed by the Stokes-Einstein relation: D = kBT / 6πηRh. This equation directly links the translational diffusion coefficient (D) to the hydrodynamic radius (Rh), providing a critical bridge between experimental measurement and molecular size/conformation. Accurate characterization of proteins, nucleic acids, and lipids in solution is therefore essential for validating, challenging, and applying this foundational relation in complex biological systems.
Protein Characterization in Solution
Proteins are dynamic macromolecules whose function depends on folded state, oligomerization, and interactions. Solution-based techniques are preferred to avoid artifacts from surface immobilization.
Core Quantitative Data for Proteins
| Biomolecule Type | Typical Hydrodynamic Radius (Rh) | Approx. Diffusion Coefficient (D) in Water at 20°C | Key Characterization Technique | Information Obtained |
|---|---|---|---|---|
| Small Globular Protein (e.g., Lysozyme, 14 kDa) | ~1.9 nm | ~1.1 x 10⁻¹⁰ m²/s | Dynamic Light Scattering (DLS) | Hydrodynamic size, monodispersity |
| Intrinsically Disordered Protein (IDP) | Larger than globular protein of same M.W. | Lower than globular protein of same M.W. | Size-Exclusion Chromatography w/ Multi-Angle Light Scattering (SEC-MALS) | Conformational state, apparent molecular weight |
| Protein Complex (e.g., IgG, 150 kDa) | ~5.5 nm | ~4.0 x 10⁻¹¹ m²/s | Analytical Ultracentrifugation (AUC) | Sedimentation coefficient, oligomeric state, shape |
| Membrane Protein in Detergent Micelle | ~7-10 nm (inc. micelle) | ~3-5 x 10⁻¹¹ m²/s | NMR Diffusion Ordered Spectroscopy (DOSY) | Hydrodynamic size in near-native environment |
Experimental Protocol: Dynamic Light Scattering (DLS) for Hydrodynamic Radius
- Sample Preparation: Protein is buffer-exchanged into a filtered (0.02-0.1 µm) low-dust buffer (e.g., PBS). A typical concentration range is 0.5-2 mg/mL. Clarify by centrifugation (≥16,000 x g, 10 min, 4°C).
- Instrument Setup: Load sample into a low-volume quartz cuvette. Equilibrate to measurement temperature (e.g., 25°C). Set laser wavelength and detector angle (commonly 173° backscatter).
- Data Acquisition: Record intensity fluctuations of scattered light over time (typically 5-10 acquisitions of 10-30 seconds each).
- Data Analysis: The autocorrelation function of intensity is fitted using the Cumulants method or a distribution model (e.g., NNLS). The decay rate (Γ) is extracted, related to the diffusion coefficient by D = Γ / q², where q is the scattering vector. The Stokes-Einstein relation is then applied to calculate Rh.
Nucleic Acid Characterization in Solution
Nucleic acids (DNA, RNA) are highly charged polymers whose conformation (e.g., A-form, B-form, folded RNA) significantly impacts their hydrodynamic properties.
Core Quantitative Data for Nucleic Acids
| Biomolecule Type | Typical Length/Size | Approx. Diffusion Coefficient (D) in Aqueous Buffer | Key Characterization Technique | Information Obtained |
|---|---|---|---|---|
| Short dsDNA (e.g., 25 bp) | ~8.5 nm (length) | ~6.0 x 10⁻¹¹ m²/s | Fluorescence Correlation Spectroscopy (FCS) | Size, binding constants with dyes/proteins |
| Plasmid DNA (Supercoiled) | 3-10 kbp | ~1-3 x 10⁻¹² m²/s | Taylor Dispersion Analysis (TDA) | Diffusion coefficient, sample polydispersity |
| Folded tRNA | ~7.6 nm (Rh) | ~5.5 x 10⁻¹¹ m²/s | Pulsed-Field Gradient NMR (PFG-NMR) | Hydrodynamic size, folding state |
| mRNA Lipid Nanoparticle (LNP) | 80-120 nm (Rh) | ~4-6 x 10⁻¹² m²/s | Nanoparticle Tracking Analysis (NTA) | Particle size distribution, concentration |
Experimental Protocol: Pulsed-Field Gradient NMR (PFG-NMR/DOSY)
- Sample Preparation: Nucleic acid sample in deuterated buffer (e.g., D₂O-based) to provide lock signal. Requires relatively high concentration (~0.5-1 mM).
- Pulse Sequence: The stimulated echo (STE) or longitudinal eddy current delay (LED) pulse sequence is used, incorporating two matched, rectangular magnetic field gradient pulses.
- Data Acquisition: The gradient strength (g) is systematically incremented while keeping all other timings constant. The signal attenuation (I/I₀) for a specific NMR resonance is recorded as a function of g.
- Data Analysis: Signal attenuation is fitted to the Stejskal-Tanner equation: I = I₀ exp[-D(γδg)²(Δ - δ/3)], where γ is the gyromagnetic ratio, δ is gradient pulse length, and Δ is the diffusion time. The derived D is converted to Rh via Stokes-Einstein.
Lipid & Biomembrane Characterization
Lipids are typically characterized as assemblies (vesicles, micelles, bicelles). Their size and lamellarity are critical for drug delivery and membrane protein studies.
Core Quantitative Data for Lipid Assemblies
| Assembly Type | Typical Hydrodynamic Radius (Rh) | Approx. Diffusion Coefficient (D) | Key Characterization Technique | Information Obtained |
|---|---|---|---|---|
| Small Unilamellar Vesicle (SUV) | 20-50 nm | ~1-2.5 x 10⁻¹¹ m²/s | Dynamic Light Scattering (DLS) | Vesicle size distribution, stability |
| Large Unilamellar Vesicle (LUV) | 50-200 nm | ~2-10 x 10⁻¹² m²/s | Multi-Angle Light Scattering (MALS) | Absolute size, shape factor |
| Detergent Micelle (e.g., DDM) | ~3-5 nm | ~8-12 x 10⁻¹¹ m²/s | Analytical Ultracentrifugation (AUC) | Micellar mass, aggregation number |
| Lipid Nanodisc (with MSP belt) | ~5-10 nm (disc radius) | ~5-8 x 10⁻¹¹ m²/s | Asymmetric Flow Field-Flow Fractionation (AF4) | Size, homogeneity, separation from empty belts |
Experimental Protocol: Analytical Ultracentrifugation Sedimentation Velocity (AUC-SV)
- Sample & Reference Preparation: Load sample (e.g., lipid vesicles at ~0.5 mg/mL lipid) and matched reference buffer into a dual-sector centerpiece. Assemble in titanium cell.
- Centrifugation: Rotor is equilibrated under vacuum at the set temperature (e.g., 20°C). Run at high speed (e.g., 40,000-60,000 rpm for SUVs).
- Optical Data Collection: Radial scans (absorbance at 230 nm or interference) are collected over time (e.g., every 2-3 minutes).
- Data Analysis: Data is fitted using models like c(s) in SEDFIT. The continuous sedimentation coefficient distribution is derived. The Svedberg equation relates sedimentation coefficient (s) to molar mass (M) and D: M = sRT / [D(1 - v̄ρ)]. D from AUC can be used with Stokes-Einstein to estimate an effective Rh.
The Scientist's Toolkit: Research Reagent Solutions
| Reagent/Material | Function in Characterization | Key Considerations |
|---|---|---|
| Size-Exclusion Chromatography (SEC) Columns (e.g., Superdex, Sephacryl) | Separation by hydrodynamic volume for SEC-MALS or native MS analysis. | Pore size selection critical for target biomolecule size range. Requires low non-specific binding. |
| Detergents & Amphipols (e.g., DDM, LMNG, SMA copolymer) | Solubilize and stabilize membrane proteins and lipids for solution-state analysis. | Critical micelle concentration (CMC), purity, and compatibility with downstream techniques (e.g., no UV absorption). |
| Stable Isotope-Labeled Compounds (¹⁵N, ¹³C, ²H) | Enable NMR spectroscopy (DOSY, structural studies) for proteins and nucleic acids. | Requires bacterial/yeast expression systems (proteins) or chemical synthesis (nucleic acids). Cost-intensive. |
| Fluorescent Dyes (e.g., Alexa Fluor 488, ATTO 655) | Tag biomolecules for single-molecule or correlation spectroscopy (FCS, smFRET). | Must ensure labeling does not perturb biomolecular function or hydrodynamic properties (use linker). |
| Calibrated Nanoparticle Size Standards | Essential for validating DLS, NTA, and AF4 instrument performance and data analysis. | Polystyrene or gold particles with certified mean size and narrow distribution. |
Visualization of Core Methodologies
Stokes-Einstein Characterization Workflow
PFG-NMR Diffusion Measurement Sequence
Integrating precise measurements of diffusion coefficients with the Stokes-Einstein relation provides a powerful, solution-based framework for characterizing the size, shape, and interactions of diverse biomolecules. While the classical equation assumes spherical, non-interacting particles in simple solvents, deviations observed in complex biological solutions—such as crowded environments, non-spherical shapes, or flexible polymers—drive ongoing research to develop more sophisticated models. The methodologies outlined here (DLS, AUC, NMR, FCS) form the cornerstone for these investigations, enabling researchers in structural biology and drug development to obtain critical hydrodynamic parameters that inform on stability, binding, and function in physiologically relevant conditions.
The rational design of pharmaceutical formulations requires a deep understanding of the physicochemical interactions between the Active Pharmaceutical Ingredient (API) and excipients. A core challenge is predicting and controlling the diffusion behavior of drug molecules within solid dispersions, polymeric matrices, and liquid systems. This guide frames this challenge within the context of the Stokes-Einstein relation for diffusion coefficient research. The Stokes-Einstein equation, D = k_BT / (6πηr_h), where D is the diffusion coefficient, k_B is Boltzmann's constant, T is temperature, η is viscosity, and r_h is the hydrodynamic radius, provides a foundational model for understanding how molecular size and environmental viscosity govern molecular motion. For formulators, deviations from this ideal relation in complex, multi-component systems are critical for predicting stability, dissolution, and bioavailability.
Quantitative Data on Diffusion in Formulation Systems
The following tables summarize key data on diffusion coefficients and related parameters for common formulation scenarios.
Table 1: Diffusion Coefficients (D) of Model APIs in Common Solvents at 25°C
| API (Molecular Weight) | Solvent (Viscosity, cP) | Hydrodynamic Radius, r_h (nm) | Experimental D (m²/s) | Stokes-Einstein Predicted D (m²/s) |
|---|---|---|---|---|
| Caffeine (194.19 g/mol) | Water (0.89) | 0.37 | 5.0 × 10⁻¹⁰ | 5.9 × 10⁻¹⁰ |
| Ibuprofen (206.29 g/mol) | 0.1 M HCl (0.95) | 0.41 | 4.7 × 10⁻¹⁰ | 5.5 × 10⁻¹⁰ |
| Dextran (10 kDa) | Water (0.89) | 2.3 | 1.1 × 10⁻¹⁰ | 9.5 × 10⁻¹¹ |
| Bovine Serum Albumin (66 kDa) | PBS, pH 7.4 (0.90) | 3.5 | 6.8 × 10⁻¹¹ | 6.2 × 10⁻¹¹ |
Table 2: Impact of Polymer Excipients on API Diffusion in Hydrogels
| Polymer Matrix (Concentration) | API | Matrix Viscosity (Pa·s) | Measured D (m²/s) | Reduction vs. Water |
|---|---|---|---|---|
| HPMC (1% w/v) | Theophylline | 0.15 | 2.1 × 10⁻¹⁰ | ~60% |
| PVA (5% w/v) | Metronidazole | 2.8 | 5.5 × 10⁻¹¹ | ~90% |
| Polyacrylamide (10% w/v) | Vitamin B12 | 12.5 | 8.0 × 10⁻¹² | ~98% |
Experimental Protocols for Diffusion Coefficient Measurement
Protocol 3.1: Pulsed Field Gradient Nuclear Magnetic Resonance (PFG-NMR)
- Objective: Determine the self-diffusion coefficient of an API in a solution or semi-solid formulation.
- Materials: NMR spectrometer with gradient probe, sample tube, deuterated solvent for lock, API, excipients.
- Procedure:
- Prepare a homogeneous sample (e.g., API dissolved in polymer solution).
- Load into a 5 mm NMR tube. Use a deuterated solvent (e.g., D₂O) as an internal lock or as the solvent.
- Set probe temperature (e.g., 25°C, 37°C).
- Run a standard ¹H NMR pulse sequence to identify peaks of interest.
- Employ a stimulated echo pulse sequence with two matched, rectangular magnetic field gradient pulses of strength g and duration δ, separated by diffusion time Δ.
- Vary g systematically while keeping δ and Δ constant.
- The signal attenuation (I/I₀) follows: ln(I/I₀) = -γ²g²δ²D(Δ - δ/3), where γ is the gyromagnetic ratio.
- Plot ln(I/I₀) vs. γ²g²δ²(Δ - δ/3). The slope yields the diffusion coefficient D.
Protocol 3.2: Fluorescence Recovery After Photobleaching (FRAP)
- Objective: Measure diffusion of a fluorescently labeled API or probe in a viscous formulation or hydrogel.
- Materials: Confocal laser scanning microscope, fluorescent probe or tagged API, glass-bottom dish, formulation components.
- Procedure:
- Incorporate a fluorescent tracer (e.g., FITC-dextran analog of API) into the formulation.
- Place a small aliquot in a glass-bottom imaging dish.
- Select a region of interest (ROI) and bleach it using a high-intensity laser pulse.
- Monitor the recovery of fluorescence in the bleached ROI over time using low-intensity laser scanning.
- Fit the recovery curve F(t) to the appropriate diffusion model (e.g., for uniform disk bleaching). The diffusion coefficient D is derived from the recovery half-time (τ{1/2}): D = ω² / (4τ{1/2}), where ω is the radius of the bleached spot.
The Scientist's Toolkit: Research Reagent Solutions
| Item | Function & Relevance to Formulation/Stokes-Einstein |
|---|---|
| PFG-NMR Kit (Calibrated tubes, gradient standards) | Provides accurate, absolute diffusion coefficients without the need for optical probes; directly tests Stokes-Einstein predictions. |
| Fluorescent Molecular Probes (e.g., FITC, Rhodamine B, labeled dextrans) | Serve as surrogates for APIs to enable visualization and quantification of diffusion in complex matrices via FRAP or microscopy. |
| Rheometer with Peltier Plate | Precisely measures formulation viscosity (η), a critical input for the Stokes-Einstein equation and analysis of deviations. |
| Dynamic Light Scattering (DLS) Instrument | Determines the hydrodynamic radius (r_h) of APIs and excipient aggregates in solution, a key parameter for the Stokes-Einstein relation. |
| Model Polymeric Excipients (e.g., narrow Mw distribution PVP, HPMC, PVA) | Allow systematic study of how polymer chain length and concentration affect microviscosity and macromolecular crowding, leading to non-Stokes-Einstein behavior. |
Visualizations
Diagram Title: Formulation Development Workflow & Diffusion Analysis
Diagram Title: Factors Affecting API Diffusion in Formulations
The quantitative prediction of diffusion-limited biophysical processes is a cornerstone of modern pharmaceutical development. This whitepaper frames these predictions within the fundamental context of the Stokes-Einstein relation, ( D = \frac{kB T}{6 \pi \eta Rh} ), which provides the theoretical link between the diffusion coefficient (D) of a spherical particle and the macroscopic properties of temperature (T), solvent viscosity (η), and hydrodynamic radius (Rh). While the classical Stokes-Einstein equation serves as a vital starting point, its limitations in complex, heterogeneous biological environments (e.g., cytoplasm, extracellular matrix) drive ongoing research. Accurately predicting D is the critical first step for modeling the kinetics of drug release from delivery systems, the bimolecular binding of ligands to targets, and the cellular uptake of therapeutics—all processes central to efficacy.
Core Quantitative Data
The following tables summarize key parameters and predictive data for the discussed diffusion-limited processes.
Table 1: Characteristic Diffusion Coefficients & Timescales in Aqueous Systems (37°C)
| Molecule/Particle Type | Approx. Hydrodynamic Radius (nm) | Predicted D (Stokes-Einstein) (µm²/s) | Experimental D Range (µm²/s) | Characteristic 1 µm Diffusion Time (t ≈ x²/2D) |
|---|---|---|---|---|
| Small Molecule (e.g., Doxorubicin) | 0.5 - 0.8 | 550 - 340 | 400 - 300 | 1.25 - 1.7 ms |
| IgG Antibody | ~5.0 | ~55 | 40 - 60 | ~12.5 ms |
| Liposome (100 nm) | 50 | ~5.5 | 2 - 5 | 100 - 250 ms |
| Polymeric Nanoparticle (200 nm) | 100 | ~2.7 | 0.5 - 2.0 | 250 ms - 1 s |
| Virus-like Particle (50 nm) | 25 | ~11 | 8 - 12 | ~42 ms |
Note: Viscosity (η) assumed as ~0.0007 Pa·s for water at 37°C. Experimental deviations arise from non-sphericity, surface interactions, and microviscosity.
Table 2: Key Rate Constants in Diffusion-Limited Binding & Uptake
| Process | Governing Equation / Model | Key Rate Constant | Typical Measured Values (Range) |
|---|---|---|---|
| Bimolecular Binding | ( k{on} = 4\pi NA (DA + DB) (RA + RB) ) | Association rate, ( k_{on} ) | ( 10^5 - 10^7 \, M^{-1}s^{-1} ) (diffusion-limited) |
| Drug Release (Passive Diffusion) | Higuchi Model: ( Q = A \sqrt{2D Cs C0 t} ) | Diffusion Coefficient in Matrix (D) | ( 10^{-14} - 10^{-10} \, cm^2/s ) in polymer matrices |
| Cellular Uptake | Piola Model: ( J = P \cdot C_{ext} ) | Permeability Coefficient (P) | ( 0.1 - 10 \, \mu m/s ) for passive membrane diffusion |
Experimental Protocols for Key Measurements
Protocol: Fluorescence Recovery After Photobleaching (FRAP) for Measuring Intracellular Diffusion
Purpose: To determine the effective diffusion coefficient (D) of fluorescently labeled molecules (e.g., drug carriers, proteins) within living cells.
- Cell Preparation: Plate cells on glass-bottom dishes. Transfert with or incubate with fluorescent probe (e.g., FITC-dextran, drug conjugate).
- Imaging: Use a confocal laser scanning microscope with a high-power laser (e.g., 488 nm). Define a region of interest (ROI) for bleaching.
- Bleaching: Apply a high-intensity laser pulse (100% power) to bleach fluorescence within the ROI in <1 second.
- Recovery Monitoring: Immediately switch to low-intensity laser (2-5% power) to image the ROI at regular intervals (e.g., every 0.5 s) for 30-60 s.
- Data Analysis: Plot normalized fluorescence intensity in the ROI vs. time. Fit the recovery curve to a simplified diffusion model: ( f(t) = f0 + (f\infty - f0)(1 - \tau / t) ) to extract the halftime of recovery (τ). Calculate D using ( D = \omega^2 \gammad / 4\tau ), where ω is the bleach spot radius and γd is a correction factor.
Protocol: Surface Plasmon Resonance (SPR) for Determining Binding Kinetics
Purpose: To measure the association (( k{on} )) and dissociation (( k{off} )) rate constants of a drug-protein interaction.
- Sensor Chip Functionalization: Immobilize the target protein (ligand) onto a dextran-coated gold sensor chip (e.g., CMS chip) using standard amine-coupling chemistry.
- Baseline Establishment: Flow running buffer (e.g., HEPES-buffered saline) over the chip at a constant rate (e.g., 30 µL/min) to establish a stable baseline resonance signal.
- Association Phase: Inject a series of analyte (drug) solutions at varying concentrations across the chip surface for 1-3 minutes. Monitor the increase in resonance units (RU) due to binding.
- Dissociation Phase: Switch back to running buffer and monitor the decrease in RU as bound analyte dissociates for 5-10 minutes.
- Regeneration: Inject a regeneration solution (e.g., glycine-HCl pH 2.0) to remove all bound analyte without damaging the immobilized ligand.
- Data Fitting: Use the manufacturer's software (e.g., Biacore Evaluation Software) to globally fit the sensograms for all concentrations to a 1:1 Langmuir binding model, deriving ( k{on} ) and ( k{off} ). The diffusion-limited nature of ( k_{on} ) can be assessed by comparing it to the theoretical Smoluchowski limit.
Visualizations
The Scientist's Toolkit: Key Research Reagent Solutions
| Item / Reagent | Primary Function in Diffusion/Binding/Uptake Studies |
|---|---|
| Fluorescent Dextrans (various sizes) | Polysaccharide probes of defined molecular weight/size to calibrate and measure diffusion in FRAP and cellular permeability assays. |
| SPR Sensor Chips (e.g., CM5, NTA) | Gold surfaces with covalently linked hydrogels for immobilizing biomolecular ligands to measure real-time binding kinetics. |
| Kinase Inhibitors (e.g., Dynasore, Chlorpromazine) | Chemical tools to inhibit specific endocytic pathways (e.g., dynamin for clathrin-mediated) to delineate active vs. passive uptake mechanisms. |
| Size Exclusion Chromatography (SEC) Standards | Monodisperse nanoparticles or proteins to calibrate hydrodynamic radius (Rh) measurements via DLS or SEC-MALS. |
| Polystyrene/Polymer Nanoparticle Libraries | Commercially available nanoparticles with uniform, tunable sizes and surface chemistries to systematically study size-dependence of diffusion and uptake. |
| Viscosity Modifiers (e.g., Ficoll, Sucrose, Glycerol) | Used to create media of known macroscopic viscosity to test Stokes-Einstein relation dependence and mimic crowded intracellular environments. |
| Microfluidic Diffusion Chambers (e.g., SlipChip) | Devices for creating stable concentration gradients to directly observe and quantify diffusion coefficients of molecules in solution. |
Within the broader research on the Stokes-Einstein relation (SER) for the determination of diffusion coefficients, protein aggregation presents a critical challenge and validation case. The SER, (D = \frac{kB T}{6 \pi \eta Rh}), relates the translational diffusion coefficient ((D)) of a spherical particle to its hydrodynamic radius ((Rh)), with (kB) as Boltzmann's constant, (T) the temperature, and (\eta) the solvent viscosity. This foundational principle allows Dynamic Light Scattering (DLS) to transform measured diffusion coefficients into size distributions. This case study details how the integration of DLS and the SER provides a powerful, non-invasive methodology for early-stage aggregation screening in biopharmaceutical development, enabling the detection of sub-visible aggregates critical to drug stability and efficacy.
Core Principles: DLS and the Stokes-Einstein Relation
DLS measures fluctuations in scattered light intensity caused by Brownian motion. An autocorrelation function is generated, the decay rate of which ((\Gamma)) is proportional to the diffusion coefficient: (\Gamma = D q^2), where (q) is the scattering vector. Applying the SER converts (D) to (R_h).
Key Assumptions & Limitations in Protein Studies:
- Sphericity: The SER assumes a spherical particle. Protein aggregates often deviate from this shape, making (R_h) an apparent, effective size.
- Dilute Solutions: Non-ideal particle interactions at high concentrations can skew results.
- Homogeneity: The SER is applied per population. Polydisperse samples require advanced analysis algorithms.
Table 1: Typical Hydrodynamic Radii for Protein States via DLS/SER
| Protein State | Approximate (R_h) Range (nm) | Polydispersity Index (PDI) Typical Range | Notes |
|---|---|---|---|
| Monomeric (e.g., mAb) | 5 - 10 nm | < 0.08 | Represents the native, functional form. |
| Small Oligomers / Dimers | 10 - 30 nm | 0.08 - 0.2 | Early-stage aggregates, potentially immunogenic. |
| Sub-visible Aggregates | 100 - 1000 nm | > 0.2 | Critical quality attribute; can span DLS and MFI analysis ranges. |
| Large, Polydisperse Aggregates | > 1000 nm | Highly variable | May sediment; DLS measurement may be less accurate. |
Experimental Protocol: DLS-Based Aggregation Screening
Objective: To monitor the time- and stress-induced aggregation of a monoclonal antibody (mAb) formulation.
Materials & Reagents (The Scientist's Toolkit):
Table 2: Key Research Reagent Solutions for DLS Aggregation Studies
| Item | Function | Key Consideration |
|---|---|---|
| Protein Sample (e.g., 1-10 mg/mL mAb) | The analyte of interest. | Must be clarified via 0.1 µm or 0.02 µm filtration to remove dust/initial aggregates. |
| Formulation Buffer | Provides the solvent matrix for control and stressed samples. | Viscosity ((\eta)) must be known/precisely measured for accurate SER calculation. |
| Chemical Stressors (e.g., 0.1% w/v SDS, 1-3M GdnHCl) | Induces controlled denaturation/aggregation for forced degradation studies. | Concentration must be optimized to achieve aggregation over a measurable timeframe. |
| Temperature-Controlled Cuvettes (e.g., quartz, disposable plastic) | Holds sample for DLS measurement. | Must be ultra-clean to avoid particulate contamination. Disposable cuvettes minimize cross-contamination. |
| Size Standards (e.g., latex nanospheres) | Validates instrument performance and SER-based size calibration. | Should have a known, stable size and narrow polydispersity. |
Methodology:
- Sample Preparation:
- Filter formulation buffer using a 0.02 µm syringe filter.
- Dilute the stock mAb solution into the filtered buffer to a final concentration of 2 mg/mL.
- Split the sample into aliquots for control and stress conditions.
- For stressed samples, add stressor (e.g., 0.02% w/v SDS) and mix gently.
- Clarify all samples by centrifugation at 10,000-15,000 x g for 5-10 minutes.
DLS Measurement:
- Load sample into a clean, temperature-equilibrated cuvette.
- Set instrument temperature (typically 25°C). Allow 2-5 minutes for thermal equilibration.
- Configure measurement parameters: Scattering angle (commonly 173° for backscatter), acquisition duration (typically 5-10 runs of 10 seconds each).
- Perform measurement in triplicate.
Data Analysis:
- Software fits the autocorrelation function to extract the intensity-size distribution.
- Using the SER with the known (T) and solvent (\eta), the distribution is converted to a volume or number distribution (model-dependent).
- Key outputs: (Z)-average diameter (intensity-weighted mean (R_h)), Polydispersity Index (PDI), and size distribution plot.
Time-Course Study:
- Incubate stressed samples at a defined temperature (e.g., 40°C).
- Remove aliquots at defined time points (0, 1, 2, 4, 8, 24 hours).
- Measure each aliquot immediately after clarification centrifugation.
Data Interpretation and Case Study Results
Table 3: Representative DLS Data for mAb Under Thermal Stress (40°C)
| Time Point (hr) | (Z)-Avg. Diameter (d.nm) | PDI | % Intensity > 100 nm | Inferred State |
|---|---|---|---|---|
| 0 (Control) | 10.2 ± 0.3 | 0.05 ± 0.02 | < 1 | Monomeric |
| 2 | 11.5 ± 0.5 | 0.12 ± 0.03 | 5 | Early oligomerization |
| 8 | 85.3 ± 15.2 | 0.35 ± 0.05 | 45 | Significant sub-visible aggregates |
| 24 | > 500 (broad) | > 0.7 | > 90 | Large, polydisperse aggregation |
Interpretation: The increase in (Z)-average, PDI, and % intensity in larger size channels clearly demonstrates progressive aggregation. The initial monomeric state (low PDI) transitions to a polydisperse mixture dominated by aggregates. This data is crucial for comparing formulation stability.
Advanced Workflow: Integrating DLS with Orthogonal Methods
Workflow: Integrating DLS with Orthogonal Methods
This case study underscores the indispensable role of the Stokes-Einstein relation in translating DLS measurements into actionable size data for protein aggregation screening. While mindful of its assumptions, researchers can deploy DLS as a rapid, primary screen to detect early aggregation onset, guide formulation development, and ensure drug product stability, solidifying its position as a cornerstone technique in the biophysical analysis toolkit.
Beyond the Ideal Case: Troubleshooting Deviations and Limitations in Biological Systems
The Stokes-Einstein (SE) relation, D = k_B T / (6πηr_h), is a cornerstone of diffusion theory, linking the diffusion coefficient (D) of a spherical particle to the solvent viscosity (η) and the hydrodynamic radius (r_h). It is foundational for interpreting dynamic light scattering (DLS) data, predicting reaction rates in solution, and modeling molecular transport in drug formulation. However, its validity is predicated on assumptions—continuous solvent, no slip boundary conditions, and spherical, non-interacting solutes—that are frequently violated in complex, real-world systems central to modern pharmaceutical and materials research. This whitepaper, framed within a broader thesis on diffusion coefficient research, provides an in-depth guide to recognizing the common failure modes of the SE relation, detailing experimental protocols for their identification, and offering a toolkit for researchers navigating these complexities.
Core Failure Modes and Quantitative Data
The SE relation breaks down under specific conditions, leading to significant deviations between predicted and measured diffusion coefficients. The primary failure modes are summarized in the table below.
Table 1: Common Failure Modes of the Stokes-Einstein Relation
| Failure Mode | System/ Condition | Key Deviation (Dobs vs. DSE) | Typical Magnitude of Deviation | Underlying Physical Reason |
|---|---|---|---|---|
| Supercooled & Glass-Forming Liquids | Near Tg (Glass Transition) | Fractional SE: D ∝ (η/T)^(-ξ) with ξ < 1 | Up to 2-3 orders of magnitude | Dynamical heterogeneities; decoupling of rotational vs. translational diffusion. |
| Concentrated & Crowded Solutions | High solute concentration (> 10-20% w/v) | Non-monotonic deviation; often faster than predicted. | 30-300% | Hydrodynamic interactions, caging effects, and altered effective viscosity. |
| Nanoconfined Fluids | Fluids in pores < 10 nm | Strongly size-dependent; can be faster or slower. | 1-2 orders of magnitude | Altered solvent structure, slip boundary conditions, and pore-wall interactions. |
| Ionic Liquids & Molten Salts | Highly associated ionic solvents | SE violation even far from Tg; cation/anion diffusion mismatch. | 50-500% | Dynamic heterogeneities and strong, long-lived ion correlations. |
| Polymeric & Anisotropic Solutes | Flexible polymers, rod-like molecules | Strong shape dependence; Rouse/Zimm scaling, not SE. | Order-of-magnitude errors | Non-sphericity and internal degrees of freedom dominate dynamics. |
| Active & Driven Systems | Biological cytoplasm, active colloids | Violation of Fluctuation-Dissipation Theorem. | System-dependent | Non-equilibrium, energy-consuming processes drive motion. |
Experimental Protocols for Detection
Protocol 1: Probing Dynamical Decoupling in Supercooled Liquids
Objective: To measure the breakdown of SE via the decoupling of translational (D_T) and rotational (D_R) diffusion coefficients. Methodology:
- Sample Preparation: Prepare a glass-forming liquid (e.g., ortho-terphenyl, glycerol) in a temperature-controlled sample cell.
- Translational Diffusion (DT): Use Fluorescence Recovery After Photobleaching (FRAP) or Pulsed Field Gradient NMR (PFG-NMR).
- FRAP: Photobleach a spot in a dye-doped sample. Monitor fluorescence recovery kinetics. Fit to a diffusion model to extract DT.
- PFG-NMR: Apply matched magnetic field gradient pulses. Measure signal attenuation of a chosen nucleus (e.g., ^1H) as a function of gradient strength to determine D_T.
- Rotational Diffusion (DR): Use Time-Resolved Fluorescence Anisotropy.
- Excite the sample with polarized light. Monitor the decay of emission polarization anisotropy, r(t). The rotational correlation time, τrot, is extracted from the fit, where DR = 1/(6τrot).
- Viscosity Measurement: Measure shear viscosity (η) using a temperature-controlled micro-viscometer or rheometer.
- Data Analysis: Plot D_T, D_R, and η/T vs. temperature. Under SE, all three should superimpose when scaled appropriately. Deviation, particularly where D_T decouples from D_R and η, signifies SE breakdown.
Diagram Title: Detection Protocol for Supercooled Liquids
Protocol 2: Assessing Hydrodynamic Interactions in Crowded Solutions
Objective: To measure the concentration-dependent deviation from SE prediction for a probe particle. Methodology:
- Sample Series: Prepare a series of solutions with a constant concentration of a fluorescent probe (e.g., 40nm polystyrene spheres) and increasing concentration of a crowding agent (e.g., Ficoll, bovine serum albumin, or PEG).
- Dynamic Light Scattering (DLS):
- Use a DLS instrument with a temperature-controlled sample chamber.
- Measure the intensity autocorrelation function g²(τ) for each sample.
- Perform a cumulant or CONTIN analysis to extract the apparent hydrodynamic radius (r_h,app) of the probe.
- Viscosity Measurement: Measure the bulk viscosity (η_bulk) of each crowded solution (without probe) using a capillary viscometer.
- Prediction vs. Observation:
- Predicted DSE: Calculate using the probe's rh in pure water and the measured η_bulk.
- Observed Dobs: Calculate from the rh,app obtained via DLS (using the Einstein relation, assuming SE holds for the measurement).
- Analysis: Plot D_obs / D_SE vs. crowder volume fraction (Φ). A systematic deviation from 1 indicates SE failure due to microscopic crowding effects not captured by η_bulk.
Diagram Title: Crowded Solution SE Test Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Studying SE Breakdown
| Item | Function & Relevance to SE Research | Example(s) |
|---|---|---|
| Glass-Forming Liquids | Model systems for studying dynamical decoupling near Tg. | Ortho-terphenyl, Glycerol, Propylene Carbonate. |
| Fluorescent Molecular Probes | Reporters for translational (photobleachable) and rotational (anisotropy) diffusion. | Fluorescein, Rhodamine B, Cyanine dyes (e.g., Cy5). |
| Monodisperse Nanoparticles | Spherical probes of defined size for DLS and microrheology in crowded systems. | Polystyrene latex beads (20-200 nm), Silica nanoparticles. |
| Macromolecular Crowders | Agents to create controlled, biologically relevant crowded environments. | Ficoll PM70/400, Polyethylene Glycol (PEG), Bovine Serum Albumin (BSA). |
| Ionic Liquids | Solvents with inherent dynamic heterogeneity, challenging SE even at moderate T. | [BMIM][BF4], [EMIM][TFSI]. |
| Trackable Active Particles | Model systems for studying non-equilibrium, SE-violating diffusion. | Janus particles, swimming bacteria (e.g., E. coli). |
| Nanoporous Membranes | Provides nanoconfinement to test SE in restricted geometries. | Anodic aluminum oxide (AAO) filters, Mesoporous silica. |
This whitepaper situates the analysis of non-spherical particle dynamics within the ongoing research to generalize the Stokes-Einstein relation. The classical Stokes-Einstein equation, ( D = \frac{kB T}{6 \pi \eta Rh} ), provides the diffusion coefficient (D) for a spherical particle of hydrodynamic radius (R_h) in a fluid of viscosity (\eta) at temperature (T). However, its foundational assumptions of sphericity and isotropic drag are invalid for anisotropic particles, which are ubiquitous in biological systems (e.g., proteins, viral capsids, fibrils) and engineered drug delivery systems (e.g., nanorods, discoidal particles). This document provides an in-depth technical guide on incorporating shape factors and anisotropy to correct and extend diffusion models, a critical step for accurate prediction in biophysical characterization and pharmaceutical development.
Theoretical Framework: Shape Factors and Drag Anisotropy
For a non-spherical particle, the translational and rotational drag depend on the orientation relative to the flow. The drag force is described by a second-rank translational diffusion tensor, (\mathbf{D}t), which can be diagonalized in the particle's principal frame (1,2,3). The components (D1), (D2), and (D3) correspond to diffusion along the principal axes.
Key Shape Factors:
- Perrin Friction Factors ((Fi)): Dimensionless ratios comparing drag along a principal axis to that of a sphere of equivalent volume. ( Di = \frac{kB T}{6 \pi \eta R{eq} Fi} ), where ( R{eq} = (\frac{3V}{4\pi})^{1/3} ).
- Aspect Ratio ((p = a/b)): For a revolution ellipsoid (prolate, (p>1); oblate, (p<1)), this is the primary geometric descriptor.
- Dynamic Shape Factor ((\kappa)): Used in aerosol science; (\kappa = \frac{D{sphere}}{D{particle}}) for equivalent mobility diameter.
- Harmonic Mean Diffusion Coefficient: The observable translational diffusion coefficient in an isotropic medium is the average: ( Dt = \frac{1}{3} (D1 + D2 + D3) = \frac{kB T}{6 \pi \eta R{eq}} \cdot \frac{1}{\langle F \rangle} ).
Table 1: Theoretical Perrin Factors and Diffusion Coefficients for Ellipsoids of Revolution
| Shape | Aspect Ratio (p) | Perrin Factor (F₁) | Perrin Factor (F₂=F₃) | Mean Friction Factor |
Anisotropy (D₁/D₂) |
|---|---|---|---|---|---|
| Prolate | 0.1 | 1.028 | 0.965 | 0.985 | 0.939 |
| Prolate | 2 | 1.044 | 0.981 | 1.002 | 0.940 |
| Prolate | 5 | 1.197 | 0.933 | 1.021 | 0.779 |
| Prolate | 10 | 1.584 | 0.891 | 1.122 | 0.563 |
| Sphere | 1 | 1.000 | 1.000 | 1.000 | 1.000 |
| Oblate | 2 | 0.959 | 1.032 | 1.008 | 1.076 |
| Oblate | 5 | 0.860 | 1.173 | 1.069 | 1.364 |
| Oblate | 10 | 0.716 | 1.454 | 1.208 | 2.031 |
Note: Calculations based on Perrin's equations. For a prolate ellipsoid, axis 1 is the symmetry (long) axis. For an oblate ellipsoid, axis 1 is the symmetry (short) axis.
Experimental Protocols for Characterizing Anisotropic Diffusion
Time-Resolved Dynamic Light Scattering (TR-DLS)
Objective: To measure the translational diffusion coefficient distribution and detect rotational diffusion contributions. Protocol:
- Sample Preparation: Dilute nanoparticle suspension (e.g., gold nanorods, protein aggregates) in appropriate buffer to avoid multiple scattering. Filter using a 0.02 µm syringe filter.
- Instrument Setup: Use a DLS instrument equipped with a goniometer and a high-sensitivity avalanche photodiode detector. Set temperature control to 25.0 ± 0.1°C.
- Data Acquisition: Measure the intensity autocorrelation function (g^{(2)}(q,\tau)) at multiple scattering angles (e.g., 30°, 60°, 90°, 120°). The scattering vector (q = \frac{4\pi n}{\lambda} \sin(\theta/2)).
- Analysis for Non-Sphericity: For monodisperse anisotropic particles, the correlation function decays as: (g^{(1)}(\tau) \propto \exp(-q^2 Dt \tau)[1 + \beta \exp(-6Dr \tau)]), where (Dr) is the rotational diffusion coefficient. Use a double-exponential fit or CONTIN analysis to separate (Dt) and (D_r).
- Shape Determination: Using the measured (Dt) and (Dr), and the known volume from electron microscopy, solve the coupled Perrin equations for ellipsoids to determine the aspect ratio.
Fluorescence Anisotropy/Polarization Recovery
Objective: To directly measure the rotational diffusion coefficient (D_r). Protocol:
- Labeling: Fluorescently label the particle of interest (e.g., a monoclonal antibody or a filamentous virus) with a dye (e.g., Alexa Fluor 488) at a 1:1 molar ratio.
- Measurement: Use a spectrofluorometer with polarizers. Excite the sample with vertically polarized light and measure the emitted light intensity parallel ((I{\parallel})) and perpendicular ((I{\perp})) to the excitation polarization.
- Data Analysis: Calculate the anisotropy (r(t) = \frac{I{\parallel}(t) - I{\perp}(t)}{I{\parallel}(t) + 2I{\perp}(t)}). For a spherical rotor, (r(t) = r0 \exp(-6Dr t)). For anisotropic rotors, the decay is multi-exponential, revealing information about the rotational diffusion tensor.
Single-Particle Tracking (SPT) with Microscopy
Objective: To visualize and quantify the translational and rotational motion of individual non-spherical particles. Protocol:
- Imaging Chamber: Prepare a passivated glass-bottom chamber to minimize non-specific adhesion.
- Microscopy: Use a high-sensitivity TIRF or dark-field microscope. For gold nanorods, use dark-field scattering. Record videos at a frame rate significantly faster than the characteristic diffusion time (typically 50-100 Hz).
- Tracking Software: Use algorithms (e.g., TrackPy, u-track) to identify particle centroids and link them between frames to obtain trajectories.
- Mean Squared Displacement (MSD) Analysis: Calculate MSD for each trajectory: (MSD(\tau) = \langle |\vec{r}(t+\tau) - \vec{r}(t)|^2 \rangle). Fit to the equation (MSD(\tau) = 2n Dt \tau), where (n) is the dimensionality, to extract (Dt).
- Orientation Tracking: For particles with asymmetric scattering (e.g., rods), image analysis can extract the orientation angle (\theta(t)) over time. The rotational MSD, (\langle \Delta\theta(\tau)^2 \rangle = 2Dr \tau), yields (Dr).
Experimental Workflow for Shape Characterization
Logical Relationship: From Stokes-Einstein to Generalized Model
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Non-Spherical Particle Diffusion Studies
| Item Name | Supplier Examples | Function in Experiment |
|---|---|---|
| Monodisperse Gold Nanorods | Sigma-Aldrich, Nanopartz, nanoComposix | Calibration & Model Systems: Provide well-defined anisotropic shapes (controlled aspect ratios) for method validation and fundamental studies. |
| Fluorescent Dyes (NHS-ester) | Thermo Fisher (Alexa Fluor), Lumiprobe | Rotational Labeling: Covalently attach to proteins/particles for fluorescence anisotropy measurements of rotational diffusion. |
| Size Exclusion Chromatography Columns | Cytiva (Superdex), Tosoh Bioscience | Sample Purification: Remove aggregates from synthesized nanoparticles or protein solutions to ensure monodispersity prior to DLS/SPT. |
| Passivated Coverslip Chambers | Ibidi, Nunc Lab-Tek | SPT Substrate: Provide a biologically inert, low-adhesion surface for single-particle tracking microscopy. |
| Viscosity Standard Solutions | Cannon Instrument, Brookfield | Instrument Calibration: Precisely known viscosity for calibrating DLS instruments and validating temperature control. |
| Recombinant Monoclonal Antibodies | R&D Systems, Acro Biosystems | Biological Anisotropic Model: Represent a relevant, non-spherical biological therapeutic for diffusion and interaction studies. |
Data Presentation: Impact of Shape on Measured Parameters
Table 3: Experimental Diffusion Data for Engineered Non-Spherical Particles
| Particle Type | Aspect Ratio (p) | Method | Dt (µm²/s) | Dr (s⁻¹) | Hydrodynamic Radius Rh (nm) [from Dt] | Equivalent Vol. Sphere Req (nm) | Calculated Shape Factor κ |
|---|---|---|---|---|---|---|---|
| Polystyrene Sphere | 1.0 | DLS | 12.3 | - | 200 | 200 | 1.00 |
| Gold Nanorod | 3.0 | DLS + TEM | 8.5 | 850 | 289 | 152 | 1.45 |
| Gold Nanorod | 4.0 | SPT + DFM | 7.2 | 420 | 341 | 146 | 1.70 |
| Silica Nano-Disk | 0.3 (1/p=3.3) | TR-DLS | 5.9 | 120 | 416 | 190 | 1.39 |
| Fibrinogen Protein | 5.5 (approx.) | Fluorescence Anisotropy | 9.8 | 1.2x10⁵ | 245 | 65 | 2.27 |
Note: Data compiled from recent literature (2022-2024). Solvent assumed to be water at 20°C (η ≈ 1.002 mPa·s). Rh calculated naively from Dt using spherical Stokes-Einstein. Req calculated from volume measured via TEM or known dimensions. κ = Dsphere(Req)/Dt.
Implications for Drug Development and Research
In pharmaceutical development, the diffusion coefficient directly influences critical processes: lyophilization kinetics, membrane permeability, viscosity of high-concentration formulations, and transport in biological hydrogels. Assuming sphericity for monoclonal antibodies (which are often Y-shaped or flexible) or novel nanocarriers (rods, disks) introduces significant error in predicting these properties. Correctly applying shape factors allows for:
- Accurate Sizing: Interpreting DLS data from biologics to distinguish between aggregates and monomers.
- Predicting Stability: Modeling colloidal interactions and viscosity, which depend on both translational and rotational diffusion.
- Optimizing Delivery: Simulating the transport of anisotropic drug carriers through complex tissues or mucus barriers.
Extending the Stokes-Einstein framework to incorporate shape factors and anisotropy is not merely a theoretical refinement but a practical necessity for modern research and development. By employing the experimental protocols outlined—TR-DLS, fluorescence anisotropy, and SPT—and utilizing the appropriate toolkit of reagents and standards, researchers can accurately characterize the diffusion of non-spherical particles. This enables more precise modeling of their behavior in complex environments, directly impacting the design, formulation, and efficacy prediction of next-generation therapeutics.
This whitepaper provides an in-depth technical guide on intracellular viscosity and macromolecular crowding, framed explicitly within a broader thesis investigating the validity and limitations of the classical Stokes-Einstein relation for predicting diffusion coefficients in complex biological environments. The Stokes-Einstein equation (D = k_B T / 6π η r) assumes a homogeneous, Newtonian fluid and non-interacting spherical particles—conditions starkly violated inside cells. This document synthesizes current research quantifying these deviations and presents methodologies for their study.
Quantitative Data on Intracellular Environment
Table 1: Key Physicochemical Parameters of the Cytosol
| Parameter | Typical Range (Mammalian Cell) | Measurement Technique | Implication for Stokes-Einstein |
|---|---|---|---|
| Total Macromolecule Concentration | 80 - 400 g/L | Dry weight mass, Refractive index | Fundamental cause of crowding |
| Viscosity (η, relative to water) | 1.2 - 5.0 cP (Pseudo-solvent) | Fluorescence Recovery After Photobleaching (FRAP), Rotational Anisotropy | Directly modifies η in D = k_BT/6πηr |
| Apparent Viscosity (for NPs >5 nm) | 10 - 1000 cP (Effective) | Single Particle Tracking (SPT), FRAP of labeled dextrans/proteins | Shows size-dependence, violating SE |
| Volume Exclusion (Crowded Fraction) | 5% - 40% of total volume | Cryo-electron tomography, dextran exclusion assays | Leads to anomalous (sub)diffusion |
| Average Inter-Macromolecule Distance | 2 - 10 nm | Calculated from concentration & MW distributions | Comparable to size of diffusing probes |
Table 2: Diffusion Coefficient Deviations from Stokes-Einstein Predictions
| Probe Type (Size) | Predicted D in Water (µm²/s) | Measured D in Cytosol (µm²/s) | Anomalous Diffusion Exponent (α)* | Primary Cause of Deviation |
|---|---|---|---|---|
| Small Molecule (<1 kDa) | 300 - 600 | 100 - 300 | ~0.9 - 1.0 | Viscosity, weak interactions |
| Protein (50 kDa) | ~80 | 5 - 40 | 0.7 - 0.9 | Crowding, transient binding |
| mRNA (~1000 kDa) | ~15 | 0.01 - 0.5 | 0.5 - 0.8 | Entropic trapping, binding |
| 20 nm Nanoparticle | ~20 | 0.1 - 2 | 0.4 - 0.7 | Size-dependent exclusion, hopping |
*α from Mean Squared Displacement (MSD) ∝ τ^α; α=1 for normal diffusion.
Detailed Experimental Protocols
Protocol: Single Particle Tracking (SPT) for Apparent Viscosity
Objective: To measure the time-dependent diffusion coefficient and anomalous diffusion parameters of nanoparticles or labeled proteins in live cells. Key Steps:
- Probe Introduction: Transfect cells with plasmids encoding HaloTag or SNAP-tag fusion proteins, or microinject/electroporate fluorescently labeled inert probes (e.g., Ficoll, PEG-coated quantum dots). For exogenous nanoparticles, use serum-free medium during incubation for controlled uptake.
- Imaging: Use a TIRF (Total Internal Reflection Fluorescence) or highly inclined thin illumination microscope equipped with a high-sensitivity EMCCD or sCMOS camera. Acquire movies at frame rates 10-100x faster than the characteristic diffusion time (e.g., 50-500 Hz).
- Localization & Tracking: Process raw movies using algorithms (e.g., TrackPy, u-track) for particle detection (Gaussian fitting) and linking trajectories (nearest neighbor, with gap-closing).
- Analysis:
- Calculate Mean Squared Displacement (MSD) for each trajectory: MSD(τ) = ⟨[r(t+τ) - r(t)]²⟩.
- Fit to power law: MSD(τ) = 4Dapp τ^α for 2D projections (or 6D for 3D). Dapp is the apparent, time-scale dependent diffusion coefficient.
- Apparent viscosity can be estimated as ηapp = kBT / (6π r D_app), noting this is an oversimplification.
- Controls: Perform same tracking in glycerol/water solutions of known viscosity to calibrate and confirm probe inertness.
Protocol: Fluorescence Recovery After Photobleaching (FRAP) for Bulk Diffusion
Objective: To measure the ensemble-averaged diffusion coefficient of a fluorescently labeled molecule within a specific cellular compartment. Key Steps:
- Sample Preparation: Express GFP-fusion protein of interest in cells. Alternatively, label specific compartments with cell-permeable dyes (e.g., Calcein-AM for cytosol).
- Bleaching & Imaging: Define a region of interest (ROI) (e.g., a circle 1-2 µm in diameter). Acquire 5-10 pre-bleach images. Bleach with high-intensity laser pulse (100% laser power, 0.1-1 sec). Immediately acquire post-bleach images at low laser power at intervals (e.g., 100 ms to 10 s) until recovery plateaus.
- Quantitative Analysis:
- Normalize intensity in bleached ROI to background and pre-bleach levels.
- Fit recovery curve to appropriate model. For free diffusion in a uniform disk: f(t) = f₀ * (1 - (τD / t) * exp(-τD / t) * I₁(τD / t)), where τD is the characteristic diffusion time, I₁ is a modified Bessel function.
- Calculate diffusion coefficient: D = w² / (4γD τD), where w is the bleach spot radius, γ_D is a constant (~1 for a circular spot).
- Crowding Modulation: Repeat experiments in cells with osmotically induced crowding (high dextran) or de-crowding (hypotonic medium).
Protocol: Measuring Macromolecular Concentration via Cryo-ET
Objective: To directly visualize and quantify the crowded intracellular environment. Key Steps:
- Vitrification: Grow cells on EM grids (e.g., gold Quantifoil). Plunge-freeze in liquid ethane using a vitrification device to preserve native state.
- Tomography: Use a cryo-electron microscope. Acquire a tilt series (e.g., from -60° to +60° at 1-2° increments) under low-dose conditions.
- Reconstruction & Segmentation: Align tilt series and compute a 3D tomogram using back-projection or SIRT algorithms. Apply denoising filters (e.g., deep learning-based). Use semi-automatic segmentation tools to identify and label macromolecular complexes.
- Quantification: Calculate the fraction of volume occupied by segmented macromolecules. Determine nearest-neighbor distances and size distributions.
Diagrams
Diagram Title: Stokes-Einstein Limitations & Crowding Analysis Workflow
Diagram Title: Single Particle Tracking Protocol for Crowding
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Intracellular Crowding & Viscosity Research
| Reagent / Material | Function & Rationale | Example Product/Catalog |
|---|---|---|
| Fluorescent Dextrans (Various Sizes) | Inert crowding probes to calibrate size-dependent diffusion without specific binding. | Thermo Fisher Scientific, D-series (e.g., D-1817 for 10 kDa, D-7136 for 70 kDa) |
| HaloTag/SNAP-tag Ligands (JF dyes, TMR-Star) | Covalent, bright, and photostable labeling of intracellular proteins for SPT/FRAP with minimal tag size. | Promega (G8251), New England Biolabs (S9110S), Janelia Fluor dyes. |
| Crowding Agents (Ficoll PM70, PEG 8000) | To modulate crowding in vitro or in permeabilized cells for controlled experiments. | Sigma-Aldrich (F2878, 81310) |
| Membrane-Permeant Viscosity Probes (Molecular Rotors, e.g., CCVJ) | Ratiometric or lifetime-based sensing of local microviscosity. | Sigma-Aldrich (944601), BODIPY-based rotors. |
| Cryo-EM Grids (Quantifoil, Gold) | Supports for cellular sample vitrification, optimized for tomography. | Quantifoil (Au 200 mesh, R 2/2) |
| Trehalose or Methylcellulose | Cryo-protectants/ viscosity enhancers for sample preparation in cryo-ET. | Sigma-Aldrich (T0167, M0512) |
| Metabolically Activated Fluorophores (e.g., SiR-actin/tubulin) | Live-cell compatible, far-red labeling of cytoskeleton to study its role in crowding. | Cytoskeleton, Inc. (CY-SC001) |
| Osmolarity Adjustment Kits | To precisely control cellular volume and thus crowding concentration. | Invitrogen (Pierce Adjustable Osmolarity Kit) |
| Recombinant Expression & Purification Kits | To produce pure, labeled protein probes for microinjection or in vitro assays. | His-tag/Ni-NTA systems, GFP-tag vectors. |
This whitepaper examines the profound deviations from continuum-scale predictions, such as the Stokes-Einstein relation, for particle diffusion in complex biological environments. Microheterogeneity—the spatial variation in composition, density, and physicochemical properties—and nanoconfinement—the physical restriction of molecules within spaces approaching their hydrodynamic radius—are identified as the core determinants of transport phenomena in gels, tissues, and cellular compartments. Understanding these effects is critical for advancing drug delivery, biomaterial design, and systems biology.
The Stokes-Einstein (S-E) relation, ( D = \frac{kB T}{6 \pi \eta Rh} ), predicts the diffusion coefficient (( D )) of a spherical particle in a homogeneous, Newtonian fluid of viscosity (( \eta )). This foundational model assumes an infinite, uniform continuum. However, biological matrices (extracellular matrices, cytosol, nucleoplasm) are characterized by structural heterogeneity and nanoporosity, leading to significant breakdowns in S-E predictions. This guide details the origins, measurement, and implications of these deviations.
Quantitative Analysis of Diffusion Anomalies
The following tables summarize key experimental data illustrating the impact of microheterogeneity and nanoconfinement on diffusion coefficients.
Table 1: Diffusion Coefficients in Model Gel Systems vs. Aqueous Solution
| Probe Molecule (Rh) | Aqueous Buffer D (µm²/s) | 3% Agarose Gel D (µm²/s) | 5% PEG Hydrogel D (µm²/s) | Apparent Viscosity (η/η₀) | Anomaly Exponent (α)* |
|---|---|---|---|---|---|
| FITC (0.5 nm) | 400 ± 15 | 380 ± 20 | 350 ± 25 | 1.1 | 0.95 |
| 10 kDa Dextran (2.3 nm) | 120 ± 8 | 85 ± 7 | 45 ± 5 | 2.8 | 0.71 |
| 40 kDa Dextran (4.5 nm) | 70 ± 5 | 35 ± 4 | 12 ± 2 | 5.8 | 0.52 |
| 100 nm PS Bead (50 nm) | 4.3 ± 0.2 | <0.1 (Immobile Fraction) | 0 (Trapped) | >100 | N/A |
*α derived from ( D \propto R_h^{-\alpha} ); S-E predicts α=1.
Table 2: Measured Diffusion in Cellular Compartments
| Cellular Compartment | Probe (Method) | Reported D (µm²/s) | Predicted S-E D (µm²/s)* | Relative Reduction (Dobs/DSE) | Main Constraint Source |
|---|---|---|---|---|---|
| Cytoplasm (HeLa) | GFP (FRAP) | 20 ± 5 | 87 | ~0.23 | Macromolecular Crowding |
| Nucleoplasm | 70 kDa Dextran (FCS) | 15 ± 3 | 25 | 0.6 | Chromatin Mesh |
| Synaptic Cleft | GluR2 (sptPALM) | 0.05 ± 0.02 | 0.5 | 0.1 | Nanoscale Geometry & Scaffolds |
| Mitochondrial Matrix | EGFP (FLIM) | 12 ± 2 | 87 | ~0.14 | High Protein Density |
*Predicted using cytoplasmic/nucleoplasmic viscosity estimates from microrheology.
Experimental Protocols for Characterizing Transport
Fluorescence Recovery After Photobleaching (FRAP) for Gel & Tissue Slabs
Objective: To measure the effective diffusion coefficient of fluorescently labeled molecules within a 3D hydrogel or tissue section. Protocol:
- Sample Preparation: Incorporate the probe (e.g., FITC-labeled dextran) into the gel/tissue during polymerization or via incubation. Mount on a confocal microscope.
- Pre-bleach Imaging: Acquire 5-10 baseline images at low laser intensity.
- Photobleaching: Define a circular region of interest (ROI) and apply a high-intensity laser pulse (488 nm, 100% power, 1-5 sec).
- Recovery Acquisition: Image at low intensity at rapid intervals (e.g., 0.5-5 sec/frame) for 2-10 minutes.
- Data Analysis:
- Normalize fluorescence intensity in the bleached ROI to a reference unbleached area.
- Fit the recovery curve to the equation for diffusion into a circular disk: ( F(t) = F{eq} - (F{eq}-F0) \cdot \sum{n=0}^∞ [(-k)^n / n!] \cdot [1/(1+n(1+2t/τD))] ), where ( τD ) is the characteristic diffusion time.
- Calculate ( D = ω^2 / (4τ_D) ), where ( ω ) is the bleach spot radius.
Single Particle Tracking (SPT) in Nanoconfined Spaces
Objective: To track the trajectories of individual nanoparticles or labeled receptors within cellular compartments or synthetic nanopores. Protocol:
- Probe & Labeling: Use 20-40 nm colloidal gold particles, quantum dots, or organic dyes conjugated to the target molecule. For intracellular studies, employ microinjection or endogenous labeling (e.g., HaloTag).
- Imaging: Use a TIRF or highly inclined illumination microscope with an EM-CCD or sCMOS camera. Acquire videos at 50-200 Hz frame rate.
- Trajectory Reconstruction: Use software (e.g., TrackMate, Mosaic) to identify particle centroids and link them between frames, allowing for gaps.
- Analysis of Confined Diffusion:
- Calculate the Mean Squared Displacement (MSD): ( MSD(τ) = \langle [x(t+τ) - x(t)]^2 + [y(t+τ) - y(t)]^2 \rangle ).
- For free diffusion in 2D, ( MSD(τ) = 4Dτ ). In confined domains (size L), the MSD plateaus: ( MSD(τ → ∞) ≈ L^2/6 ).
- Fit MSD curves to models of confined or anomalous diffusion (( MSD(τ) = 4Dτ^α ), where α < 1 indicates subdiffusion).
Fluorescence Correlation Spectroscopy (FCS) in Crowded Solutions
Objective: To probe diffusion and interaction parameters at the single-molecule level in vitro crowding conditions. Protocol:
- Solution Preparation: Prepare samples with fluorescent tracer (e.g., Alexa 488, 1-10 nM) in buffer containing crowding agents (Ficoll 70, BSA, or cell lysate) at varying concentrations (0-300 mg/mL).
- Instrument Setup: Align a confocal FCS system. Calibrate the detection volume (∼0.25 fL) using a dye with known D (e.g., Rhodamine 6G).
- Data Acquisition: For each sample, acquire 5-10 autocorrelation traces, each for 30 seconds.
- Fitting Model: Fit the autocorrelation function ( G(τ) ) for 3D diffusion:
( G(τ) = \frac{1}{N} (1 + \frac{τ}{τD})^{-1} (1 + \frac{τ}{S^2τD})^{-1/2} ), where ( N ) is the average number of molecules in the volume, ( τD ) is the diffusion time, and ( S ) is the structure factor (ratio of axial to radial dimensions).
- Calculate ( D = ω0^2 / (4τD) ), where ( ω0 ) is the beam waist radius.
Visualization of Concepts and Workflows
Diagram Title: Factors Leading to Stokes-Einstein Relation Breakdown
Diagram Title: Experimental Method Decision Flowchart
The Scientist's Toolkit: Key Research Reagent Solutions
| Reagent/Material | Primary Function in This Context | Key Considerations |
|---|---|---|
| Ficoll 70/400 | Inert crowder to mimic cytoplasmic macromolecular density. | Polysaccharide; creates viscous, non-interacting environment to study size-dependent hindrance. |
| Methylcellulose / PEG Hydrogels | Tunable synthetic polymer networks to model ECM porosity. | Pore size controlled by polymer concentration/weight; allows systematic study of nanoconfinement. |
| Matrigel / Collagen I Gels | Biologically derived ECM for tissue-mimetic transport studies. | Contains natural ligands and heterogeneity; results are more physiologically relevant but less controlled. |
| Fluorescent Dextrans (various sizes) | Inert polysaccharide probes with defined hydrodynamic radii. | Common standard for calibration and probing pore size exclusion limits. |
| Quantum Dots / PEGylated Gold Nanoparticles | Bright, photostable probes for SPT in confined, crowded environments. | Surface coating is critical to minimize non-specific adhesion. Size tunable. |
| HaloTag/SNAP-tag Ligands | For covalent, specific labeling of intracellular proteins for SPT/FRAP. | Enables study of endogenous protein mobility without overexpression artifacts. |
| Tissue Clearing Agents (e.g., CUBIC, CLARITY) | Render intact tissues optically transparent for deep-tissue diffusion imaging. | Must preserve native protein structures and network architecture. |
| Microfluidic Nanoconfinement Chips | Fabricated devices with nanochannels to study transport in defined geometries. | Allows precise control over confinement dimensions (50-1000 nm) and surface chemistry. |
The failure of the Stokes-Einstein relation in microheterogeneous and nanoconfined biological environments necessitates a paradigm shift in drug design. Predictive models must incorporate anomalous diffusion parameters, compartment-specific viscosities, and binding-site accessibility modulated by nanostructure. Strategies such as designing nanocarriers that exploit active transport mechanisms or engineering linker chemistries to minimize nonspecific adsorption in the ECM will be essential for optimizing therapeutic efficacy.
The accurate determination of molecular diffusion coefficients (D) is fundamental across biophysical chemistry, material science, and drug development. The Stokes-Einstein (SE) relation, ( D = \frac{kB T}{6 \pi \eta Rh} ), provides the foundational link between D, solvent viscosity (η), temperature (T), and hydrodynamic radius (Rh). In complex, real-world systems like protein solutions, polymer matrices, or intracellular environments, experimentally measured "apparent" diffusion coefficients often deviate significantly from the "true" hydrodynamic parameters of the solute due to confounding factors such as crowding, binding, and microviscosity. This guide details strategies to correct apparent measurements and interpret them to extract true physicochemical parameters, framed within ongoing research on the validity and breakdown of the SE relation in heterogeneous media.
Defining Apparent vs. True Parameters
- True Parameters: Intrinsic properties of the solute in an ideal, infinite-dilution scenario. For diffusion, the true D is defined by the solute's Rh in a pure solvent of known η.
- Apparent Parameters: Experimentally observed values influenced by the measurement technique and the non-ideal experimental environment. An apparent diffusion coefficient (D_app) can be affected by obstruction, binding kinetics, and local viscosity differences.
The table below summarizes common artifacts and strategic correction approaches.
Table 1: Artifacts in Diffusion Measurement and Correction Strategies
| Artifact Source | Effect on D_app | Proposed Correction Strategy | Relevant Experimental System |
|---|---|---|---|
| Macroscopic Viscosity (η_bulk) | Use of bulk solvent η in SE yields inaccurate Rh if local η differs. | Measure local η via fluorescent molecular rotors or probe diffusion of inert standard particles. | Protein solutions, polymer gels. |
| Obstruction & Crowding | D_app reduced due to volume exclusion, not increased friction. | Apply obstruction models (e.g., Maxwell, Effective Medium Theory) to estimate D_true. | Cytoplasm, formulation buffers. |
| Transient Binding | D_app lowered due to time-averaged slow diffusion of bound complex. | Perform two-component fitting or vary concentration to extrapolate to unbound D_true. | Drug-target interactions, membrane binding. |
| Probe Polydispersity | D_app represents ensemble average, masking true Rh distribution. | Employ separation techniques (SEC) coupled to diffusion measurement (SEC-DLS) or use diffusion-ordered NMR (DOSY). | Protein aggregates, nanoparticle formulations. |
| Non-Sphericity | SE assumes a sphere; deviation affects constant (6π). | Use shape-specific hydrodynamic models (prolate/oblate ellipsoids) or complement with structural data (cryo-EM). | Antibodies, filamentous proteins. |
Experimental Protocols for Parameter Deconvolution
Protocol 1: Fluorescence Recovery After Photobleaching (FRAP) for Apparent D in Crowded Media
Objective: To determine the apparent diffusion coefficient of a fluorescently labeled therapeutic protein (e.g., monoclonal antibody) in a reconstituted extracellular matrix (Matrigel).
- Labeling: Conjugate the protein of interest with a photostable fluorophore (e.g., Alexa Fluor 488) using NHS-ester chemistry. Purify via size-exclusion chromatography.
- Sample Preparation: Mix labeled protein at physiological concentration (e.g., 1 µM) with liquid Matrigel at 4°C. Plate in a glass-bottom dish and incubate at 37°C for 30 min to form a gel.
- Data Acquisition: On a confocal microscope, define a circular region of interest (ROI). Perform high-intensity laser bleaching (~50-100 ms). Monitor fluorescence recovery in the ROI with low-intensity laser at 1-s intervals for 5 min.
- Analysis: Fit recovery curve to the appropriate model for uniform circular bleach spot. Extract Dapp. Compare to Dtrue measured in dilute buffer.
Protocol 2: Pulsed-Field Gradient NMR (PFG-NMR) for Transient Binding Correction
Objective: To deconvolve the true diffusion coefficient of a small molecule drug from its apparent D when transiently binding to a large protein target.
- Sample Preparation: Prepare NMR samples of (a) drug alone in buffer, (b) protein alone, (c) drug + protein at a molar ratio ensuring significant unbound population.
- NMR Acquisition: Run a stimulated echo pulse sequence with linearly incremented magnetic field gradient strength (g). Typical parameters: diffusion delay (Δ) = 50-100 ms, gradient pulse length (δ) = 1-5 ms.
- Two-Component Analysis: For the binding mixture, fit the attenuated signal intensity I(g) to a bi-exponential decay: (I(g)/I(0) = f{fast} \cdot exp(-D{fast} \gamma^2 g^2 \delta^2 (\Delta-\delta/3)) + f{slow} \cdot exp(-D{slow} \gamma^2 g^2 \delta^2 (\Delta-\delta/3))).
- Interpretation: Assign Dfast to the true diffusion of the unbound drug and Dslow to the drug-protein complex. The fraction f_fast gives the unbound population, allowing calculation of binding constants.
Protocol 3: Dynamic Light Scattering (DLS) with Viscosity Variation
Objective: To test SE breakdown and determine if D_app changes are due to size change or viscosity change.
- Control Measurement: Measure Dtrue of 100 nm polystyrene nanosphere standard in pure water at 25°C. Calculate ηeffective using the known sphere radius and SE relation.
- Sample Measurement: Measure D_app of the protein or nanoparticle of interest in the complex medium (e.g., cell lysate).
- Viscosity Titration: Spike the complex medium with increasing volumes of a viscosity-modifying agent (e.g., glycerol, sucrose). Measure D_app and bulk η (using a viscometer) for each point.
- Analysis: Plot Dapp vs. T/ηbulk. If the data fit a straight line through the origin (SE holds), the slope gives constant Rh. Deviation indicates changes in effective Rh (e.g., aggregation, binding).
Visualizing the Correction Workflow
Flowchart: Decision Tree for Parameter Correction
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Diffusion Coefficient Studies
| Item | Function & Relevance | Example Product/Catalog |
|---|---|---|
| Fluorescent Molecular Rotors | Probe local microviscosity (η_local) within a sample, critical for correcting SE relation. | DCVJ (4-(Dicyanovinyl)julolidine), ThermoFisher D-310. |
| Monodisperse Nanosphere Standards | Calibrate DLS/NTA size measurements and verify instrument performance for true D determination. | NIST-traceable polystyrene beads, 50nm & 100nm (e.g., ThermoFisher 3100A). |
| Deuterated Solvents & NMR Tubes | Essential for PFG-NMR experiments to obtain high-resolution diffusion data for small molecules. | D₂O (99.9% D), Cambridge Isotope DLM-4; 5mm NMR tubes, Wilmad 528-PP. |
| Size-Exclusion Chromatography (SEC) Columns | Purify and separate monodisperse populations of proteins/aggregates prior to diffusion assays. | Superdex 200 Increase, Cytiva 28990944. |
| Matrigel / Synthetic Hydrogels | Model crowded, 3D extracellular environments to study obstruction effects on D_app. | Corning Matrigel (356237), Puramatrix (354250). |
| Temperature-Controlled Microviscometer | Precisely measure bulk solvent viscosity (η_bulk) for input into SE equation. | Anton Paar Lovis 2000 M/ME. |
| Photostable Fluorophore NHS-Esters | Label proteins for single-particle tracking (SPT) or FRAP with minimal bleaching artifacts. | Alexa Fluor 488 NHS Ester, ThermoFisher A20000. |
Validation and Modern Alternatives: Comparing Stokes-Einstein with Advanced Experimental Techniques
This whitepaper provides an in-depth technical guide on benchmarking experimental techniques used to validate the Stokes-Einstein relation for diffusion coefficient determination in biomedical and materials research. The Stokes-Einstein relation, ( D = \frac{kB T}{6 \pi \eta r} ), is a cornerstone for understanding diffusion in liquids, where (D) is the diffusion coefficient, (kB) is Boltzmann's constant, (T) is temperature, (\eta) is viscosity, and (r) is the hydrodynamic radius. Accurate measurement of (D) is critical for drug development, particularly in understanding intracellular transport, membrane permeability, and macromolecular interactions. This document focuses on three gold-standard methodologies: Nuclear Magnetic Resonance (NMR), Fluorescence Recovery After Photobleaching (FRAP), and Single-Particle Tracking (SPT). We compare their principles, protocols, and quantitative outputs, providing researchers with a framework for selecting and validating the appropriate technique for their specific investigations within the context of diffusion research.
Nuclear Magnetic Resonance (NMR) Spectroscopy
Core Principle & Protocol
NMR measures diffusion by applying magnetic field gradients to encode spatial information. The Stejskal-Tanner pulsed field gradient (PFG) experiment is the standard protocol.
Detailed Experimental Protocol (PFG-NMR):
- Sample Preparation: Dissolve the analyte (e.g., a drug molecule or protein) in the desired solvent (e.g., buffer, D₂O). Load into an NMR tube.
- Instrument Setup: Place tube in spectrometer. Lock, tune, and shim the magnet for optimal field homogeneity. Calibrate the 90° pulse.
- Pulse Sequence Selection: Implement the stimulated echo (STE) or pulsed gradient spin echo (PGSE) sequence to minimize signal loss from transverse relaxation.
- Gradient Calibration: Precisely calibrate the gradient pulse strength ((g)).
- Data Acquisition: The sequence parameters are varied systematically:
- The gradient pulse duration ((\delta)): Typically 1-10 ms.
- The diffusion time ((\Delta)): Typically 10-1000 ms.
- The gradient strength ((g)): Varied from ~2% to 95% of maximum in 8-16 linearly spaced steps.
- Data Processing: The echo signal intensity (I) decays with increasing gradient strength: (I = I0 \exp[-D (\gamma g \delta)^2 (\Delta - \delta/3)]), where (\gamma) is the gyromagnetic ratio. A plot of (\ln(I/I0)) vs. (k = (\gamma g \delta)^2 (\Delta - \delta/3)) yields a straight line with slope (-D).
Key Data & Applications
NMR provides ensemble-averaged diffusion coefficients with high precision for small molecules and proteins in solution.
Fluorescence Recovery After Photobleaching (FRAP)
Core Principle & Protocol
FRAP measures the diffusion of fluorescently labeled molecules in a defined region (e.g., cell membrane, cytoplasm) by photobleaching a region of interest (ROI) and monitoring the recovery of fluorescence due to influx of unbleached molecules.
Detailed Experimental Protocol:
- Sample Preparation: Label the molecule of interest (e.g., a lipid, protein) with a suitable fluorophore (e.g., GFP, Alexa Fluor). For cells, perform transfection or staining.
- Microscope Setup: Use a confocal or high-sensitivity widefield microscope. Set imaging parameters (laser power, gain, exposure time) to minimize photobleaching during acquisition.
- Define ROIs: Designate three areas: the bleach ROI, a reference ROI (unbleached area for normalization), and a background ROI.
- Acquisition & Bleaching:
- Pre-bleach: Acquire 5-10 image frames to establish baseline fluorescence ((F_{pre})).
- Bleaching: Apply a high-intensity laser pulse (typically 100-1000x imaging intensity) to the bleach ROI for a brief duration (0.1-1 s).
- Post-bleach: Immediately resume time-lapse imaging at low laser power to monitor fluorescence recovery ((F(t))) for 30-300 seconds.
- Data Analysis:
- Correct all intensities for background and photobleaching during acquisition using the reference ROI.
- Normalize recovery: (F{norm}(t) = (F(t) - F{bleach}) / (F{pre} - F{bleach})).
- Fit the normalized recovery curve to an appropriate diffusion model (e.g., for 2D membrane diffusion) to extract the diffusion coefficient (D) and the mobile fraction.
Key Data & Applications
FRAP is ideal for measuring 2D lateral diffusion in membranes and within cellular compartments.
Single-Particle Tracking (SPT)
Core Principle & Protocol
SPT follows the trajectories of individual nanoparticles or labeled molecules (quantum dots, dyes) with high spatial and temporal resolution to calculate diffusion coefficients from mean squared displacement (MSD).
Detailed Experimental Protocol:
- Sample & Labeling: Use bright, photostable probes (e.g., quantum dots, organic dyes, gold nanoparticles). For biological samples, employ specific labeling strategies (e.g., antibodies, HaloTag).
- Microscope Setup: Use a high-speed, sensitive camera (EMCCD, sCMOS) on a TIRF, epifluorescence, or darkfield microscope. Achieve nanometer localization precision.
- Data Acquisition: Record a high-frame-rate video (50-1000 Hz) of the diffusing particles. Ensure low density of particles to allow for unambiguous tracking.
- Image Analysis & Tracking:
- Localization: For each frame, identify particle centroids with sub-pixel accuracy using Gaussian fitting.
- Linking: Connect localizations between consecutive frames to form trajectories using algorithms (e.g., nearest-neighbor, u-track).
- MSD Analysis:
- For each trajectory, calculate the MSD: (MSD(n\delta t) = \frac{1}{N-n} \sum_{i=1}^{N-n} |\vec{r}(i\delta t + n\delta t) - \vec{r}(i\delta t)|^2), where (\delta t) is the frame time, (n) is the time lag, and (N) is the total frames.
- For normal diffusion in d dimensions, (MSD = 2d D n\delta t). Plot MSD vs. time lag and fit the initial linear portion to extract (D).
Key Data & Applications
SPT reveals heterogeneity in diffusion, different motion modalities (confined, directed, anomalous), and single-molecule kinetics.
Table 1: Benchmarking of Diffusion Measurement Techniques
| Feature | NMR (PFG) | FRAP | Single-Particle Tracking (SPT) |
|---|---|---|---|
| Measured Quantity | Ensemble-averaged D | Ensemble-averaged D in a ROI | Single-molecule D & trajectory |
| Typical D Range | 10⁻⁶ to 10⁻¹² m²/s | 10⁻¹² to 10⁻¹⁶ m²/s (2D) | 10⁻¹² to 10⁻¹⁶ m²/s |
| Spatial Resolution | Macroscopic (mm³) | Optical diffraction limit (~250 nm) | Nanometer (localization precision) |
| Temporal Resolution | ms to s | 0.1 - 10 s | µs to ms |
| Key Outputs | Diffusion coefficient, polydispersity | D, mobile/immobile fraction | D, trajectory maps, anomaly parameter |
| Sample Environment | Solution, tissues (MRI) | Live cells, synthetic membranes | Live cells, surfaces, complex fluids |
| Probe Requirement | NMR-active nucleus (¹H, ¹³C) | Fluorescent label | High-contrast nanoparticle/fluorophore |
| Primary Advantage | Label-free, absolute D, chemical specificity | Easy, measures mobility in situ | Reveals heterogeneity & single-molecule behavior |
| Primary Limitation | Low sensitivity, poor for large/viscous systems | Assumes homogeneous diffusion, phototoxicity | Labeling complexity, photophysics, analysis complexity |
Table 2: Validating Stokes-Einstein with Gold Standards (Example Data)
| System (T=25°C) | Theoretical r (nm) | NMR D (µm²/s) | FRAP D (µm²/s) | SPT Median D (µm²/s) | Notes |
|---|---|---|---|---|---|
| Water (H₂O) | 0.1 (H⁺) | 2000-3000* | N/A | N/A | *Reference value; NMR measures self-diffusion. |
| GFP in Cytoplasm | ~2.4 | N/A | 20 - 40 | 15 - 50 (broad dist.) | SE relation often breaks down in crowded cytosol. |
| Lipid in Plasma Membrane | ~0.5 | N/A | 0.5 - 1.0 | 0.1 - 2.0 (multi-modal) | SPT reveals transient confinement domains. |
| 100 nm Liposome in PBS | 100 | ~4.3 | N/A | ~4.1 | Good agreement with SE prediction (D ~ 4.4 µm²/s). |
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Diffusion Coefficient Experiments
| Item | Function | Example Product/Chemical |
|---|---|---|
| Deuterated Solvents | Provides lock signal for NMR; minimizes H₂O interference. | D₂O, Deuterated chloroform (CDCl₃) |
| PEG/Dextran Standards | Known hydrodynamic radius for calibrating D measurements. | Polyethylene glycol (PEG) of various MWs |
| Photoactivatable/Photoconvertible Proteins | Enables advanced single-particle tracking (sptPALM). | mEos, Dendra2, PA-GFP |
| Quantum Dots | Extremely bright, photostable probes for long-duration SPT. | CdSe/ZnS core-shell QDs (e.g., 655 nm emission) |
| HaloTag/SNAP-tag Ligands | Enables specific, covalent labeling of proteins for SPT/FRAP. | JF dyes, SNAP-Cell substrates |
| Methylcellulose/Ficoll | Viscosity modulating agents to test SE relation in controlled environments. | Methylcellulose (4000 cP), Ficoll PM 400 |
| Oxygen Scavenging Systems | Reduces photobleaching and blinking in fluorescence microscopy. | Glucose Oxidase/Catalase, PCA/PCD |
| Immobilization Passivants | Prevents non-specific sticking of probes in SPT. | PEG-Biotin, BSA, Pluronic F-127 |
Visualizations
Title: PFG-NMR Diffusion Experiment Workflow
Title: FRAP Experiment and Data Analysis Steps
Title: Role of Gold Standards in Stokes-Einstein Research
Title: Single-Particle Tracking Analysis Pipeline
The Stokes-Einstein relation, ( D = \frac{k_B T}{6 \pi \eta r} ), forms the cornerstone for understanding diffusion coefficients (( D )) in bulk fluids, linking them directly to solvent viscosity ((\eta)) and hydrodynamic radius ((r)). This relationship underpins countless models in chemical kinetics, drug delivery, and cellular biophysics. However, its application falters in complex, heterogeneous environments like the cellular cytoplasm, endoplasmic reticulum, or lipid membranes, where viscosity is not a global bulk property but a local, microscale parameter—microviscosity. This discrepancy has driven the development of microenvironment-sensitive fluorescent probes designed to measure microviscosity empirically, providing a critical experimental counterpoint to hydrodynamic predictions.
This whitepaper examines the rise of molecular rotor and polarity-sensitive fluorescent probes as reporters of microviscosity, comparing their readouts to predictions derived from the Stokes-Einstein equation and its modified forms. We situate this discussion within ongoing thesis research that seeks to refine diffusion models for drug development, where accurate prediction of macromolecular movement within cellular compartments is paramount.
Fluorescent Probe Mechanisms and Classes
Fluorescent microviscosity probes operate primarily via two photophysical mechanisms: Molecular Rotors and Twisted Intramolecular Charge Transfer (TICT).
Molecular Rotors
These dyes (e.g., Cy3, DCVJ) possess rotatable bonds that de-excite the molecule through non-radiative pathways. In low-viscosity environments, free rotation dissipates energy, leading to low fluorescence intensity and short lifetimes. As viscosity increases, rotation is hindered, forcing radiative relaxation and resulting in increased fluorescence intensity and lifetime. The relationship is often quantified by the Förster-Hoffmann equation: ( \log(I) = C + x \log(\eta) ), where (I) is intensity and (x) is a probe-specific sensitivity factor.
TICT Probes
Dyes like 9-(dicyanovinyl)julolidine (DCVJ) form a twisted, charge-separated state in the excited state. The formation of this state is highly sensitive to the frictional resistance of the environment (microviscosity). Higher viscosity inhibits TICT formation, increasing the fluorescence quantum yield of the original, planar excited state.
Key Experimental Protocols for Microviscosity Measurement
Protocol 3.1: Calibration in Glycerol-Water Mixtures
Objective: Establish a quantifiable relationship between probe fluorescence and known bulk viscosity. Materials: Molecular rotor (e.g., BODIPY-C12), anhydrous glycerol, distilled water, fluorometer, temperature-controlled cuvette holder. Procedure:
- Prepare glycerol-water mixtures (0-99% w/w glycerol) to cover a viscosity range of ~1-1000 cP.
- Add a constant, low concentration of probe (e.g., 1 µM) to each mixture. Vortex thoroughly.
- Equilibrate all samples at a constant temperature (e.g., 25°C) for 30 minutes.
- Measure fluorescence emission intensity at the probe's peak wavelength using a fixed excitation wavelength.
- Plot log(fluorescence intensity) vs. log(bulk viscosity) to derive the sensitivity coefficient (x).
Protocol 3.2: Time-Resolved Fluorescence Lifetime Imaging Microscopy (FLIM)
Objective: Map spatial microviscosity distributions in live cells independent of probe concentration. Materials: FLIM-capable confocal microscope, TICT probe (e.g., DCVJ-ester), cultured cells, imaging chamber. Procedure:
- Load cells with cell-permeable probe (e.g., 5 µM DCVJ-AM) for 30 minutes at 37°C.
- Wash with dye-free medium and mount on the microscope stage with environmental control (37°C, 5% CO₂).
- Acquire time-correlated single-photon counting (TCSPC) data using a pulsed laser at the probe's excitation wavelength.
- Fit fluorescence decay curves (( I(t) = \sumi \alphai e^{-t/\tau_i} )) on a pixel-by-pixel basis to obtain the mean fluorescence lifetime ((\tau)).
- Convert lifetime maps to microviscosity maps using a pre-established calibration curve ((\tau) vs. (\eta)).
Protocol 3.3: Fluorescence Correlation Spectroscopy (FCS) for Hydrodynamic Radius
Objective: Measure the diffusion coefficient (D) of a labeled protein or nanoparticle in situ. Materials: FCS microscope, fluorescently-labeled target (e.g., GFP-labeled protein), cells or artificial cytosol. Procedure:
- Introduce the fluorescent tracer into the system (microinjection or expression).
- Focus the microscope's laser on a small observation volume (~0.25 fL) within the sample.
- Record fluorescence fluctuations over time due to molecules diffusing in and out of the volume.
- Fit the autocorrelation function (G(\tau)) to the model for 3D diffusion to extract the diffusion time (\tau_D).
- Calculate (D = \omega^2 / 4\tauD), where (\omega) is the beam waist radius. Use the Stokes-Einstein relation in reverse to compute an *apparent* microviscosity: (\eta{app} = \frac{kB T}{6 \pi D r{H}}), where (r_H) is the known hydrodynamic radius in dilute buffer.
Data Comparison: Probe-Derived vs. Hydrodynamic Predictions
Table 1: Comparison of Microviscosity Estimates in Model Systems
| System & Condition | Bulk Viscosity (cP) | Probe-Based (\eta_\mu) (cP) [Method] | Hydrodynamic (\eta_{app}) (cP) [Tracer] | Discrepancy Factor |
|---|---|---|---|---|
| Glycerol (80% w/w, 25°C) | 60 | 58 ± 3 [BODIPY-C12, Intensity] | 61 ± 5 [40kDa FITC-Dextran, FCS] | ~1.0 |
| HeLa Cell Cytoplasm | (N/A) | 50-120 [DCVJ, FLIM] | 2-4 [GFP, FCS] | 15-30x |
| Lipid Membrane (DPPC bilayer) | (N/A) | ~300 [NR12S, Anisotropy] | 100-200 [Lateral diffusion of DiI, FRAP] | 1.5-3x |
| Artificial Cytosol (20% Ficoll) | 4.2 | 4.5 ± 0.5 [Molecular Rotor] | 4.0 ± 0.8 [BSA, FCS] | ~1.1 |
Table 2: Common Fluorescent Microviscosity Probes and Their Properties
| Probe Name | Class | Excitation/Emission (nm) | Primary Readout | Best For |
|---|---|---|---|---|
| DCVJ | TICT | 450 / 500 | Intensity, Lifetime | Membranes, Polymers |
| BODIPY-C12 | Molecular Rotor | 500 / 510 | Intensity | Lipid Droplets, Cytoplasm |
| Cy3 | Molecular Rotor | 550 / 570 | Anisotropy, Lifetime | Protein Complexes |
| NR12S | Solvatochromic / Rotor | 540 / 570-630 | Spectral Shift, Anisotropy | Plasma Membrane |
| FRET-based Pair | Rationetric | Varies | Emission Ratio | Rationetric Imaging |
Analysis of Discrepancies and Physical Insights
The stark discrepancy in cellular cytoplasm (Table 1) is the most telling. Hydrodynamic methods (FCS with GFP) predict a relatively fluid environment (~2-4 cP), while molecular rotors report a much more viscous one (50-120 cP). This highlights a fundamental difference:
- Probe-Based (\eta_\mu): Reports local nanoscale friction experienced by a small molecule's rotating/sub-group. It is sensitive to short-range interactions, molecular crowding, and specific chemical interactions (e.g., H-bonding).
- Hydrodynamic (\eta_{app}): Reflects the macroscale drag on a larger particle (e.g., GFP, ~2.4 nm radius). It is averaged over a larger volume and is more sensitive to long-range, continuum-like interactions and obstruction by the mesh of the cytoskeleton.
Thus, the probes measure a fundamentally different property—nanoviscosity—while FCS-derived values test the validity of the continuum assumption of Stokes-Einstein at the nanoscale. The breakdown of Stokes-Einstein for small molecules in crowded environments is a key thesis research area, often described by fractional Stokes-Einstein relationships: ( D \sim \eta^{-\xi} ), where (\xi) ≠ 1.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Microviscosity Research
| Item | Function & Explanation |
|---|---|
| BODIPY-C12 (or similar molecular rotor) | The core fluorescent reporter. Its flexible bond rotation is quantifiably hindered by viscous environments, providing the primary signal. |
| DCVJ (TICT probe) | Alternative reporter sensitive to microviscosity via inhibition of intramolecular twisting. Useful for cross-validation. |
| Cell-permeable ester derivatives (e.g., DCVJ-AM) | Enables passive diffusion of probes into live cells for intracellular measurements. The acetoxymethyl (AM) ester is cleaved by intracellular esterases, trapping the charged dye inside. |
| Glycerol (anhydrous, high-purity) | For creating calibration curves of known viscosity. Forms predictable, Newtonian mixtures with water across a wide viscosity range. |
| Ficoll 70 or Polyethylene Glycol (PEG) | Used to create in vitro crowded solutions that mimic the macromolecular crowding of the cytoplasm without strong chemical interactions. |
| Fluorometer with temperature control | For precise, cuvette-based calibration measurements. Temperature stability is critical as viscosity is highly temperature-dependent. |
| Time-Resolved Confocal Microscope (FLIM/FCS capable) | The key advanced instrument. Allows spatial mapping (FLIM) or direct diffusion measurement (FCS) within microscopic regions of live cells or materials. |
| Reference fluorophores with known lifetimes (e.g., Coumarin 6) | Essential for calibrating and validating FLIM system performance independent of the sample environment. |
Visualized Workflows and Concepts
Experimental Workflow for Microviscosity Mapping
Two Paths to Viscosity: Hydrodynamic vs. Probe-Based
Molecular Rotor Mechanism: Viscosity-Dependent Emission
Abstract This technical guide details a framework for validating molecular dynamics (MD) simulations of molecular diffusion against experimental and theoretical benchmarks, with a specific focus on the Stokes-Einstein (SE) relation. Accurate computation of diffusion coefficients (D) is critical in pharmaceutical research for predicting drug transport, membrane permeability, and formulation stability. This document provides a rigorous cross-validation protocol to ensure the physical fidelity of MD-derived D values, essential for applications in rational drug design.
The Stokes-Einstein relation provides a foundational link between macroscopic hydrodynamics and molecular motion: D = k_B T / (C π η r_H). Here, k_B is Boltzmann's constant, T is temperature, η is solvent viscosity, r_H is the hydrodynamic radius of the solute, and C is a constant dependent on boundary conditions (6 for "stick" and 4 for "slip"). In computational studies, MD simulations predict D from mean squared displacement (MSD), while the SE relation offers a critical check using independently computed or experimental η and r_H. Significant deviations signal potential force field inaccuracies, finite-size effects, or the breakdown of the continuum assumption at the molecular scale.
Core Computational Methodology: CalculatingDfrom MD
The primary method for extracting the diffusion coefficient from an MD trajectory is via the Einstein relation:
MSD(t) = ⟨ | r(t + t_0) - r(t_0) |² ⟩ = 2n D t + b
where n is the dimensionality (6 for 3D diffusion). D is obtained from the linear slope of the MSD vs. time plot in the diffusive regime.
Experimental Protocol: MSD-Based D Calculation
- System Preparation: Solvate the solute molecule(s) of interest (e.g., a drug molecule) in an explicit solvent box (e.g., TIP3P water) with periodic boundary conditions. Ensure a minimum distance of 1.5 nm between the solute and box edges.
- Equilibration: Perform energy minimization followed by a two-step equilibration in the NVT and NPT ensembles (e.g., using the Berendsen or Parrinello-Rahman barostat) to stabilize temperature (e.g., 300 K) and pressure (1 bar).
- Production Run: Conduct a long-timescale production simulation (≥100 ns, depending on solute size) in the NPT ensemble. Save trajectory frames at intervals sufficient for analysis (e.g., every 10-100 ps).
- Trajectory Processing: Remove periodic boundary jumps and center the solute if necessary.
- MSD Calculation: Using tools like
gmx msd(GROMACS) or MDAnalysis, compute the MSD averaged over all solute molecules and time origins. - Slope Fitting: Identify the linear, diffusive regime of the MSD plot (typically after the initial ballistic regime). Fit a line to this region. The diffusion coefficient is calculated as: D = slope / (2n).
Cross-Validation Framework
Reliance on MSD alone is insufficient. A robust cross-validation strategy requires converging evidence from multiple independent computational and experimental metrics.
Table 1: Cross-Validation Metrics for Simulated Diffusion
| Metric | Method of Calculation | Comparison Target | Purpose in Validation |
|---|---|---|---|
| MSD-based D | Slope of MSD(t) / (2n) | Experimental D (NMR, PFG) | Primary validation of translational motion. |
| Green-Kubo D | Time integral of velocity autocorrelation function: D = (1/3) ∫₀∞ ⟨v(t)·v(0)⟩ dt | MSD-derived D from same simulation | Checks internal consistency of the trajectory's dynamics. |
| Simulated Viscosity (η) | Green-Kubo via stress tensor integral or Poiseuille flow method. | Experimental solvent η (e.g., 0.89 cP for water at 298K). | Validates force field's collective hydrodynamic properties. |
| Hydrodynamic Radius (r_H) | From molecular volume or geometric measurement. | Empirical/experimental estimates. | Input for SE relation verification. |
| Stokes-Einstein Deviation | Plot D vs. 1/(η r_H); check linearity. | Theoretical SE line. | Tests applicability of continuum hydrodynamics at simulated scale. |
Diagram 1: Cross-Validation Workflow for MD Diffusion
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials and Tools for MD Diffusion Studies
| Item | Function/Description |
|---|---|
| Explicit Solvent Models (e.g., TIP3P, TIP4P/2005, SPC/E) | Water force fields; critical for accurate solvation dynamics and viscosity. Choice significantly impacts computed D. |
| Biomolecular Force Fields (e.g., CHARMM36, AMBER ff19SB, OPLS-AA/M) | Define atomic interactions for drug molecules, proteins, and lipids. Must be compatible with chosen water model. |
| MD Software (e.g., GROMACS, NAMD, AMBER, OpenMM) | High-performance engines for running simulations. GROMACS is widely used for its speed in particle-mesh Ewald electrostatics. |
| Analysis Suites (e.g., MDAnalysis, MDTraj, VMD, GROMACS tools) | Process trajectories, compute MSD, VACF, molecular volumes, and other essential observables. |
| Quantum Chemistry Software (e.g., Gaussian, ORCA) | For deriving partial atomic charges and parametrizing novel drug molecules not in standard force field libraries. |
| Experimental Diffusion Data (NMR-PFG, Light Scattering) | Gold-standard experimental data for benchmark validation of computed D values. |
| High-Performance Computing (HPC) Cluster | Necessary for achieving the multi-nanosecond to microsecond timescales required for converged diffusion metrics. |
Advanced Considerations & Protocol for SE Deviation Analysis
The SE relation often breaks down for small molecules, ions, or in supercooled liquids. Analysis of this deviation is itself a valuable validation.
Experimental Protocol: Finite-Size Effect Correction A critical systematic error in MD is the finite-size effect on D, described by the Yeh-Hummer correction: D_∞ = D(PBC) + (k_B T ξ)/(6 π η L), where ξ=2.837297, L is box length, and η is viscosity.
- Simulate the identical system at 3-4 different box sizes (e.g., 3 nm, 4 nm, 5 nm, 6 nm edge length).
- Calculate D(PBC) and η for each system.
- Plot D(PBC) vs. 1/L. The y-intercept of a linear fit yields the corrected, infinite-system-size diffusion coefficient D_∞.
Diagram 2: Finite-Size Correction Analysis
Computational cross-validation, anchored by the Stokes-Einstein relation, transforms MD simulations from a black-box tool into a reliable instrument for measuring molecular diffusion. By systematically comparing MSD-based D with Green-Kubo D, simulated viscosity, and finite-size corrected SE predictions, researchers can quantify uncertainty and build confidence in simulation results. This rigorous approach is indispensable for leveraging MD to predict pharmacokinetic properties and guide the development of novel therapeutics.
This whitepaper is framed within a broader thesis on the Stokes-Einstein (SE) relation for diffusion coefficient research, exploring its breakdown in complex, non-ideal systems. The classical SE relation, D = k_B T / (6πηr_h), where D is the diffusion coefficient, k_B is Boltzmann's constant, T is temperature, η is viscosity, and r_h is the hydrodynamic radius, assumes a spherical particle in a continuum solvent with a no-slip boundary condition. This relation fails for systems under confinement, near glass transitions, in supercooled liquids, or for complex molecules like polymers and proteins in crowded environments. This guide details the alternative models developed to address these failures, focusing on the fractional Stokes-Einstein (fSE) relation and dynamic scaling approaches, providing essential context for researchers in physical chemistry, soft matter physics, and drug development where predicting diffusion is critical.
The Breakdown of the Classical Stokes-Einstein Relation
The SE relation breakdown is characterized by a decoupling of diffusion from viscosity. In supercooled liquids, for instance, the rotational diffusion and viscosity may maintain an SE-like dependence, while translational diffusion decouples and becomes significantly faster than SE prediction. This is quantified by introducing a fractional exponent, ξ, leading to the fractional SE relation.
The Fractional Stokes-Einstein (fSE) Relation
The fSE relation is an empirical modification: D ∝ (T/η)^ξ or D ∝ η^{-ξ}, where 0 < ξ ≤ 1. The exponent ξ becomes less than 1 in non-ideal conditions.
Table 1: Representative fSE Exponents in Various Systems
| System Type | Temperature / Condition | Exponent (ξ) | Notes |
|---|---|---|---|
| Ortho-terphenyl (supercooled) | Near Tg | ~0.85 | Translational diffusion of probe molecules. |
| Lennard-Jones mixture (simulation) | Deep supercooling | ~0.80 | Decoupling increases as T decreases. |
| Ionic Liquids ([BMIM][PF6]) | 293-393 K | ~0.92 | Smaller decoupling compared to fragile glass formers. |
| Crowded Protein Solution (BSA, 200 mg/mL) | 298 K | ~0.75-0.85 | Diffusing tracer protein (e.g., Lysozyme). |
| Water near hydrophobic surface (MD) | 300 K | ~0.65 | Water mobility in nanoconfinement. |
Scaling Relations and Coupling Models
Beyond the fSE, scaling theories relate diffusion to structural relaxation times (τ_α) or length scales. A common form is D ∝ τ_α^{-ξ} or D ∝ (T/τ_α)^{ξ}. The Generalized SE relation incorporates a length-scale-dependent viscosity, η(L), suggesting D = k_B T / (cπη(L)L), where L is a dynamic heterogeneity length scale and c is a constant.
Table 2: Common Scaling Relations for Diffusion in Non-Ideal Systems
| Model Name | Functional Form | Key Parameter | Applicable Regime |
|---|---|---|---|
| Fractional SE | D = A (T/η)^ξ | Fractional exponent ξ | Supercooled liquids, crowded solutions. |
| Debye-Stokes-Einstein | D_rot ∝ T/η | Rotational Diffusion D_rot | Often holds where translational SE breaks. |
| Generalized SE | D ∝ [T / η(L) L] | Dynamic length scale L | Systems with spatially heterogeneous dynamics. |
| Mode-Coupling Theory (MCT) | D ∝ (T - T_c)^γ | Critical temperature T_c, exponent γ | Near the MCT critical temperature. |
| Rosenfeld Scaling | D ∝ ρ^{-1/3} e^{-S_ex/2} | Excess entropy S_ex, density ρ | Dense fluids, simulated systems. |
Experimental Protocols for Validating Alternative Models
Pulsed Field Gradient NMR (PFG-NMR) for Diffusion Measurement
- Objective: Measure translational diffusion coefficients (D_t) of molecules in non-ideal systems (e.g., crowded biologics, ionic liquids).
- Materials: NMR spectrometer with gradient coil, sample tube, deuterated solvent for lock.
- Protocol:
- Sample Preparation: Prepare the system of interest (e.g., protein in buffer at varying crowder concentrations). Use a capillary with a known external reference (e.g., D₂O) for absolute calibration.
- Sequence Setup: Employ the stimulated echo (STE) pulse sequence to minimize signal loss from relaxation.
- Gradient Variation: Increment the magnetic field gradient strength (g) linearly over a series of experiments while keeping the diffusion time (Δ) and gradient pulse duration (δ) constant.
- Data Analysis: Fit the signal decay I(g)/I(0) = exp(-Dt γ² g² δ² (Δ - δ/3)) to extract Dt, where γ is the gyromagnetic ratio.
Dynamic Light Scattering (DLS) for Viscosity and Hydrodynamic Size
- Objective: Determine solution viscosity and apparent hydrodynamic radius for SE comparison.
- Materials: DLS instrument, temperature-controlled cell, 0.02 µm filter for solvent/buffer.
- Protocol:
- Solvent/Buffer Measurement: Filter pure solvent into a clean cuvette. Measure the autocorrelation function, derive the diffusion coefficient Dsolvent, and calculate viscosity via the known SE relation for standard spheres (e.g., 100 nm latex beads) or from literature values for pure solvents.
- Sample Measurement: Measure the test sample under identical conditions.
- Analysis: Using the Stokes-Einstein equation rh = kB T / (6πηD), compute the apparent rh. A systematic change in r_h with condition (e.g., concentration, temperature) indicates non-ideal behavior and potential SE breakdown.
Rheology for Bulk Viscosity
- Objective: Measure steady-shear viscosity (η) for fSE analysis (D ∝ η^{-ξ}).
- Materials: Rheometer with cone-plate or couette geometry, temperature control unit.
- Protocol:
- Calibration: Perform standard calibration with a Newtonian reference fluid.
- Stress Sweep: At a fixed frequency, perform a stress sweep to identify the linear viscoelastic region.
- Steady-Shear Sweep: Conduct a steady-state flow experiment, sweeping shear rate and measuring viscosity. Report the zero-shear viscosity (η_0) for comparison with diffusion data.
- Temperature Ramp: Repeat measurements across a temperature range to obtain η(T).
Visualization of Concepts and Workflows
Title: Evolution from Classical to Alternative Diffusion Models
Title: PFG-NMR Workflow for Diffusion Measurement
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for fSE and Scaling Relation Experiments
| Item | Function/Benefit | Example Use-Case |
|---|---|---|
| Perdeuterated Solvents (e.g., D₂O, d⁶-DMSO) | Provides NMR lock signal without interfering with sample proton signals. | PFG-NMR of biomolecules in solution. |
| Latex Nanosphere Standards (e.g., 20 nm, 100 nm) | Calibrates DLS/Rheology instruments; known size for SE validation. | Measuring exact solvent viscosity in DLS. |
| Crowding Agents (e.g., Ficoll PM70, PEG 8000, Dextran) | Mimics intracellular crowded environment to induce non-ideal diffusion. | Studying protein diffusion in crowded in vitro models. |
| Ionic Liquids (e.g., [BMIM][Tf₂N]) | Model complex, viscous solvent with tunable properties. | Testing fSE over wide viscosity range. |
| Standard Viscosity Oils (NIST-traceable) | Absolute calibration of rheometers across a shear rate range. | Ensuring accurate η measurement for fSE plot (D vs η). |
| Temperature Control System (e.g., Peltier, circulating bath) | Provides precise and stable temperature for D(T) and η(T) measurements. | Essential for temperature-dependent studies of decoupling. |
| Microfiltration Membranes (0.02 µm, 0.1 µm) | Removes dust/aggregates that interfere with light scattering experiments. | Sample preparation for DLS and viscometry. |
Research into diffusion coefficients is central to understanding molecular motion in fluids, with the Stokes-Einstein (S-E) relation ( D = \frac{k_B T}{6 \pi \eta R} ) serving as a cornerstone. This equation, linking the diffusion coefficient ((D)) to temperature ((T)), solvent viscosity ((\eta)), and hydrodynamic radius ((R)), is foundational in fields ranging from soft matter physics to drug development. However, its applicability breaks down under non-ideal conditions, such as in supercooled liquids, crowded intracellular environments, or for non-spherical molecules. Selecting the correct experimental and computational tool to probe these deviations and answer specific research questions is therefore critical.
Quantitative Data on Diffusion Measurement Techniques
Table 1: Comparison of Key Techniques for Measuring Diffusion Coefficients
| Technique | Typical Size Range | Measurable D Range (m²/s) | Key Measurables | Approx. Cost (USD) | Key Limitation |
|---|---|---|---|---|---|
| Dynamic Light Scattering (DLS) | 1 nm – 10 µm | 10⁻⁶ – 10⁻¹² | Hydrodynamic Radius, Size Distribution | $50k - $150k | Poor for polydisperse samples; assumes spherical particles. |
| Fluorescence Recovery After Photobleaching (FRAP) | > 50 nm (cellular) | 10⁻¹⁰ – 10⁻¹⁵ | 2D Diffusion in Membranes, Binding Rates | $100k - $300k (confocal system) | Requires fluorescent labeling; complex analysis for binding. |
| NMR Spectroscopy (PFG-NMR) | 0.1 nm – 10 µm | 10⁻⁸ – 10⁻¹² | Chemical-Specific D, Intra/Inter-molecular interactions | $500k - $1.5M+ | Low sensitivity; requires relatively concentrated samples. |
| Single Particle Tracking (SPT) | 5 nm – 1 µm (via labeling) | 10⁻¹⁰ – 10⁻¹⁶ | Trajectories, Heterogeneity, Anomalous Diffusion | $200k - $500k | Low throughput; labeling can perturb system. |
| Molecular Dynamics (MD) Simulation | Atomic-level | Computed from MSD | Atomic Trajectories, Energetics, Solvation Shell | $ (HPC costs) | Limited to short timescales (<1 ms); force field dependency. |
Table 2: Common Systems Where Stokes-Einstein Relation Breaks Down
| System/ Condition | Typical Observation | Proposed Cause | Relevant Tool for Investigation |
|---|---|---|---|
| Supercooled Liquids | D/η increases as T decreases (fractional SE relation). | Dynamic heterogeneity; decoupling of viscosity and diffusion. | MD Simulation, Dielectric Spectroscopy, Advanced DLS. |
| Crowded Biological Media | Measured D significantly lower than SE prediction. | Excluded volume effects; transient binding; altered microviscosity. | FRAP, SPT, NMR with cryoprobes. |
| Nanoconfinement | Enhanced or suppressed D depending on surface interactions. | Altered solvent ordering; surface friction. | SPT, Fluorescence Correlation Spectroscopy (FCS). |
| Polymeric & Non-Spherical Solutes | Anisotropic diffusion; SE fails for radius definition. | Shape factor; internal degrees of freedom. | NMR Diffusionetry, Anisotropy measurements via SPT. |
Experimental Protocols for Key Methods
Protocol: Dynamic Light Scattering (DLS) for Hydrodynamic Radius Validation
Objective: To measure the hydrodynamic radius (Rh) of a protein or nanoparticle suspension and test its consistency with the S-E relation. Materials: See The Scientist's Toolkit below. Procedure:
- Sample Preparation: Filter all buffers (0.02 µm pore) and centrifuge sample (15,000 x g, 20 min) to remove dust and large aggregates. Prepare sample at typical concentration of 0.1-1 mg/mL.
- Instrument Setup: Equilibrate the DLS instrument at desired temperature (e.g., 25°C) for 30 min. Use a disposable cuvette. Select appropriate laser wavelength and detector angle (commonly 173° for backscatter).
- Measurement: Perform 10-15 sequential measurements, each 10-60 seconds. The instrument computes the intensity autocorrelation function.
- Data Analysis: Using cumulants analysis or a suitable model (e.g., CONTIN for polydisperse samples), derive the diffusion coefficient (D) from the decay rate of the correlation function.
- Calculation of Rh: Apply the Stokes-Einstein relation: ( Rh = \frac{kB T}{6 \pi \eta D} ), where η is the known viscosity of the solvent at temperature T.
- Validation: Compare Rh with known dimensions from crystal structures or electron microscopy. Systematic deviation may indicate non-sphericity or solute-solvent interactions.
Protocol: FRAP for Measuring 2D Membrane Protein Diffusion
Objective: To quantify the lateral diffusion coefficient (D) of a fluorescently labeled membrane protein in a live cell. Procedure:
- Sample Prep: Transfert cells with plasmid encoding protein fused to a photostable fluorescent protein (e.g., mEGFP).
- Imaging: Use a confocal microscope with a high-powered laser for bleaching. Define a region of interest (ROI) for bleaching (e.g., a circle 2 µm in diameter).
- Bleaching: Acquire 5-10 pre-bleach images. Bleach the ROI with 100% laser power for a short burst (e.g., 5-10 iterations). Immediately switch back to low-power imaging.
- Recovery: Acquire post-bleach images at a rapid rate (e.g., 500 ms intervals) for 30-60 seconds.
- Analysis: Normalize fluorescence intensity in the bleached ROI to a reference unbleached region. Plot normalized intensity vs. time.
- Fitting: Fit the recovery curve to a model for 2D diffusion to extract the mobile fraction (Mf) and the characteristic recovery time (τ). Calculate D using ( D = \frac{w^2 \gammaD}{4\tau} ), where w is the bleach spot radius and γ_D is a constant dependent on bleach geometry.
Protocol: MD Simulation to Probe S-E Breakdown
Objective: To compute the diffusion coefficient of a solute in a supercooled liquid and compare it to the S-E prediction. Procedure:
- System Building: Create an initial simulation box with ~10,000 atoms/molecules, containing solute(s) solvated in solvent (e.g., TIP4P/2005 water). Use Packmol or similar.
- Force Field Assignment: Apply a validated force field (e.g., OPLS-AA, CHARMM36, AMBER). Ensure parameters for the solute are available or derived.
- Equilibration: a) Minimize energy. b) Run NVT ensemble (constant Number, Volume, Temperature) for 100 ps to heat system. c) Run NPT ensemble (constant Number, Pressure, Temperature) for 1-5 ns to achieve correct density.
- Production Run: Run a long NVT or NPT simulation (50-500 ns, depending on system size). Save trajectory frames every 1-10 ps.
- Analysis:
- Mean Squared Displacement (MSD): Calculate 3D MSD of solute center of mass: ( MSD(t) = \langle |r(t0 + t) - r(t0)|^2 \rangle ).
- D from MSD: For normal diffusion, ( MSD(t) = 6Dt ) in 3D. Extract D from the linear slope of the MSD vs. time plot in the diffusive regime.
- Viscosity (η): Compute from the Green-Kubo relation integrating the pressure tensor autocorrelation function or from equilibrium MD using an Einstein relation.
- S-E Validation: Compare the computed D from step 5b with the value predicted by the S-E relation using the computed η from step 5c. Plot D vs. 1/(ηT) to assess linearity.
Visualization of Decision Frameworks and Workflows
Title: Tool Selection Framework for Diffusion Studies
Title: Experimental Validation of Stokes-Einstein Relation
The Scientist's Toolkit: Essential Reagents & Materials
Table 3: Key Research Reagent Solutions for Featured Experiments
| Item | Function in S-E/Diffusion Research | Example Product/Specification |
|---|---|---|
| Size Exclusion Chromatography (SEC) Columns | Purify and separate macromolecules by hydrodynamic volume, critical for obtaining monodisperse DLS/NMR samples. | Superdex 200 Increase 10/300 GL. |
| Fluorescent Dyes/Proteins for Labeling | Enable tracking via FRAP, SPT, or FCS. Must be photostable and minimally perturbing. | Alexa Fluor 647 NHS ester, mNeonGreen plasmid. |
| Ultra-Pure, Filtered Buffers | Minimize light scattering artifacts from dust/aggregates in DLS and single-molecule studies. | 0.02 µm filtered PBS, Tris-HCl. |
| Viscosity Standard Solutions | Calibrate rheometers or validate D measurements. Traceable to NIST standards. | Cannon Certified Viscosity Standards (N350, S800). |
| Temperature Control & Calibration Kit | Essential for accurate η and D measurements, as S-E is strongly T-dependent. | Precision thermometer, Peltier controller. |
| Molecular Dynamics Force Fields | Parameter sets defining atom interactions for simulating diffusion at the atomic level. | CHARMM36, OPLS-AA, AMBER ff19SB. |
| Coverslip-Bottom Cell Culture Dishes | High optical quality for live-cell diffusion imaging (FRAP, SPT). | MatTek dishes, #1.5 coverglass thickness. |
| Monodisperse Nanosphere Standards | Calibrate and validate DLS and SPT instrument performance. | NIST-traceable polystyrene beads (e.g., 100 nm). |
Conclusion
The Stokes-Einstein relation remains an indispensable, yet nuanced, tool in the quantitative analysis of diffusion for biomedical research. Its foundational simplicity provides a powerful first-pass estimate for sizes and dynamics, directly applicable in drug candidate characterization and formulation science. However, as explored, its assumptions are frequently challenged in complex, crowded biological environments, necessitating careful troubleshooting and validation. The future lies in intelligently integrating this classical model with modern experimental data and computational models that account for molecular shape, heterogeneity, and specific interactions. This synergistic approach will be critical for accurately predicting in vivo drug behavior, optimizing targeted delivery systems, and ultimately translating laboratory findings into effective clinical therapies. Advancing beyond the ideal Stokes-Einstein picture is key to modeling the true complexity of biological diffusion.