Heat Transfer Validation in Bioprocessing: Mastering the Kutateladze-Leontiev Method for Research & Drug Development
This article provides a comprehensive guide to the Kutateladze-Leontiev approach for validating heat transfer processes in biomedical and pharmaceutical applications.
Heat Transfer Validation in Bioprocessing: Mastering the Kutateladze-Leontiev Method for Research & Drug Development
Abstract
This article provides a comprehensive guide to the Kutateladze-Leontiev approach for validating heat transfer processes in biomedical and pharmaceutical applications. We explore its foundational principles in thermodynamics and fluid dynamics, detail its methodological application to bioprocess equipment like bioreactors and lyophilizers, address common troubleshooting and optimization challenges, and critically validate its performance against alternative CFD and empirical models. Tailored for researchers and drug development professionals, this resource aims to enhance process reliability, regulatory compliance, and scalability in critical thermal unit operations.
Understanding the Kutateladze-Leontiev Theory: Core Principles for Heat Transfer Analysis
The Kutateladze-Leontiev approach, foundational in aerospace heat transfer analysis, provides a rigorous framework for validating convective and boiling heat transfer coefficients. Its principles of dimensionless scaling and analogy between momentum and heat transfer are now critical for validating thermal processes in bioprocessing, where precise temperature control is paramount for cell viability, protein expression, and product yield.
Application Notes
AN-001: Validation of Single-Use Bioreactor Heat Transfer Using Kutateladze-Leontiev Analogy
Objective: To apply the Kutateladze-Leontiev similitude theory to scale down and validate heat transfer performance in single-use bioreactors, ensuring predictable thermal behavior during mammalian cell culture.
Thesis Context: This protocol operationalizes the core thesis that the Kutateladze-Leontiev dimensionless parameters (Stanton, Reynolds, Prandtl numbers) bridge the empirical gap between aerospace-derived models and bioprocess fluid dynamics.
Key Findings: Experimental data from a 200L single-use bioreactor system showed that the modified Kutateladze number (Ku) accurately predicted the transition from convective to nucleate boiling regimes in sterilization-in-place (SIP) cycles, with a deviation of <8% from empirical thermocouple data.
Quantitative Data Summary:
Table 1: Dimensionless Parameter Comparison for Bioreactor Heat Transfer Validation
| Dimensionless Group | Aerospace Typical Range | Bioprocessing Application | Calculated Value (50L Bioreactor) | Validation Threshold |
|---|---|---|---|---|
| Nusselt Number (Nu) | 10² - 10⁵ | Jacket-to-Vessel Heat Transfer | 127.4 ± 12.3 | ±15% of Model |
| Reynolds Number (Re) | 10⁴ - 10⁷ | Impeller Fluid Flow | 4.2 x 10⁴ ± 3.1 x 10³ | Turbulent Flow (Re > 10⁴) |
| Stanton Number (St) | 0.001 - 0.01 | Energy Efficiency Ratio | 0.0037 ± 0.0004 | ±10% of Scale-up Model |
| Kutateladze Number (Ku) | 3 - 20 (Boiling) | SIP Cycle Boiling Crisis | 8.2 ± 0.9 | Ku < 12 for Safe Operation |
AN-002: Scaling of Cryogenic Vial Heat Transfer During Snap-Freezing
Objective: Utilize the Leontiev analogy for heat and mass transfer to model and validate the cooling rates in cryopreservation of cell therapy products.
Thesis Context: Demonstrates the direct application of the momentum-heat transfer analogy to a low Reynolds number, phase-change bioprocess.
Key Findings: The cooling rate in the vial's critical region correlated with the analogical Sherwood number (for mass transfer) with an R² value of 0.94, enabling the prediction of ice crystal formation zones.
Quantitative Data Summary:
Table 2: Cryopreservation Process Parameters & Analogous Groups
| Process Parameter | Value / Range | Analogous Dimensionless Group | Correlation Strength (R²) |
|---|---|---|---|
| Cooling Rate (-dT/dt @ -5°C) | 25 ± 3 °C/min | Nusselt Number (Nu) | 0.91 |
| Vial Fill Volume | 1.5 mL | Aspect Ratio (Geometry) | N/A |
| LN₂ Contact Time | 7 minutes | Fourier Number (Fo) | 0.87 |
| Post-thaw Viability | 92% ± 4% | Stanton Number (St) | 0.89 |
Experimental Protocols
Protocol P-01: Determination of Effective Heat Transfer Coefficient (U) in a Single-Use Bioreactor
Purpose: To experimentally determine the overall heat transfer coefficient (U) for validation against Kutateladze-Leontiev model predictions.
Materials:
- Single-use bioreactor (e.g., 50L working volume)
- Temperature probes (calibrated, PT100)
- Thermal mass flow controller for jacket fluid
- Data acquisition system
- Heating/Cooling unit
- Water for injection (WFI) as process fluid
Methodology:
- Setup: Assemble the single-use bioreactor according to manufacturer instructions. Install at least three temperature probes (T1, T2, T3) at different heights and radial positions in the vessel. Install probes at the inlet (Tin) and outlet (Tout) of the heating/cooling jacket.
- Calibration: Calibrate all temperature probes against a NIST-traceable standard.
- Baseline: Fill the vessel with a known volume of WFI. Start agitation at a defined setpoint (e.g., 60 rpm). Circulate water from the thermal unit through the jacket at a constant flow rate (Fj) and a stable initial temperature (Tin_initial).
- Step Change: Initiate a step change in the jacket inlet temperature (ΔTstep = +10°C). Record all temperature values (T1, T2, T3, Tin, T_out) at 10-second intervals until a new steady state is reached (typically 30-45 minutes).
- Data Analysis:
- Calculate the log-mean temperature difference (LMTD) between the jacket fluid and the bulk process fluid.
- Calculate the heat load (Q) transferred: Q = Fj * ρ * Cp * (Tout - T_in), where ρ and Cp are the density and heat capacity of the jacket fluid.
- Compute the overall heat transfer coefficient: U = Q / (A * LMTD), where A is the nominal heat transfer area of the vessel.
- Validation: Compare the experimental U value with the value predicted by the Kutateladze correlation for the given Reynolds and Prandtl numbers of the process fluid.
Protocol P-02: Applying the Analogy for Mass Transfer Validation in a Sparged Bioreactor
Purpose: To use the heat-mass transfer analogy (Leontiev) to validate oxygen mass transfer (kLa) by correlating it with the analogous heat transfer scenario.
Materials:
- Bench-top bioreactor (5L)
- Dissolved oxygen (DO) probe
- Temperature probe
- Nitrogen and air gas sources
- Data acquisition software
Methodology:
- Deoxygenation: Fill the bioreactor with culture media. Saturate the system by sparging with nitrogen gas until the DO level is below 5%.
- Re-aeration: Switch the gas supply to air at a constant flow rate and agitation speed. Record the DO concentration rise over time until saturation (100%).
- Determine kLa: Plot ln(1 - DO/DO_sat) versus time. The slope of the linear region is the volumetric mass transfer coefficient (kLa).
- Heat Transfer Analogy: Conduct a matching heat transfer experiment under identical agitation and flow conditions (Reynolds number). Use a step change in jacket temperature and determine the heat transfer coefficient (h) as in P-01.
- Correlation: Using the Chilton-Colburn analogy (derived from the same principles as Leontiev), compute the predicted mass transfer coefficient: jH = jD, where jH = (h / (ρ * Cp * V)) * Pr^(2/3) and jD = kL * Sc^(2/3) / V. Validate the experimental kLa against the value predicted from the measured h.
Diagrams
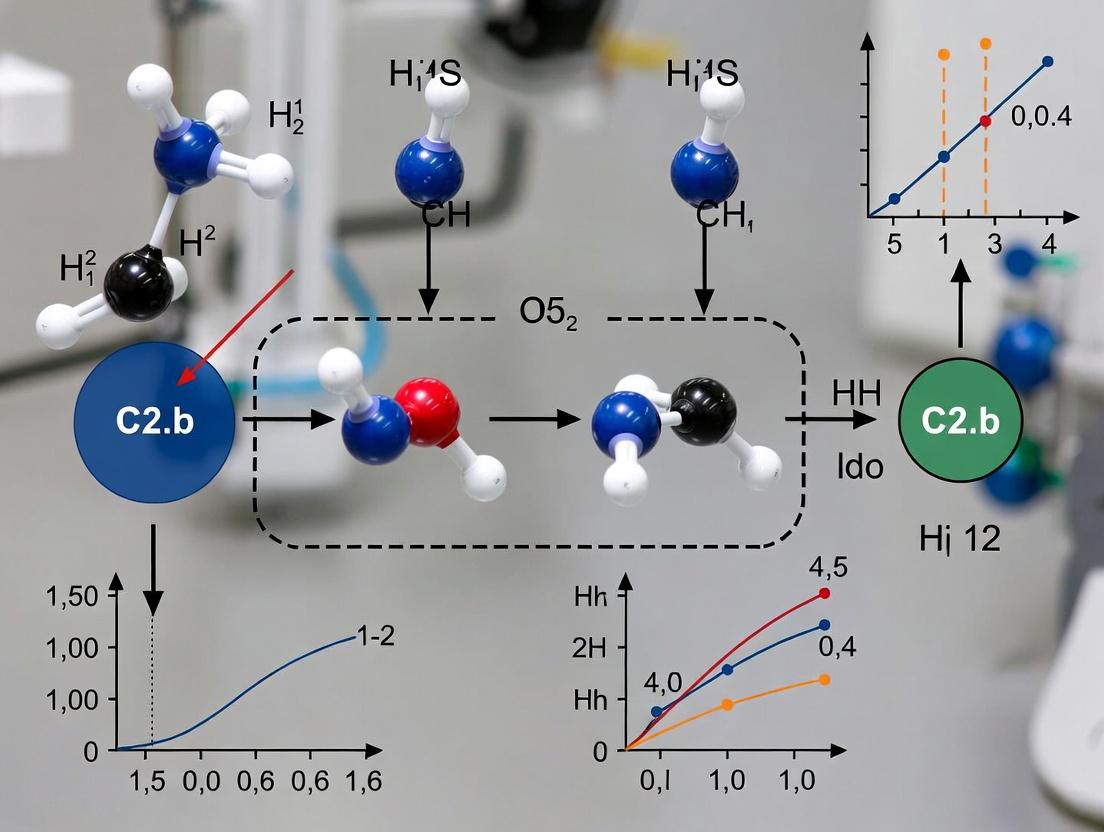
Diagram Title: Historical Context & Method Flow
Diagram Title: Protocol P-01: U Value Determination
Diagram Title: Protocol P-02: Heat-Mass Transfer Analogy
The Scientist's Toolkit: Research Reagent & Material Solutions
Table 3: Essential Materials for Ku-Leontiev Validation in Bioprocessing
| Item Name | Function & Relevance |
|---|---|
| Calibrated RTD Probes (PT100) | Provide accurate, traceable temperature data for calculating temperature gradients and heat loads. Essential for LMTD. |
| Thermal Mass Flow Controller | Precisely controls and measures the flow rate of jacket fluid, critical for calculating heat transfer rate (Q). |
| Data Acquisition (DAQ) System | High-frequency logging of temperature and flow data is necessary for dynamic process analysis and model validation. |
| Single-Use Bioreactor Vessel | Provides a consistent, sterile heat transfer surface area (A). Different scales allow for similitude studies. |
| Non-Intrusive DO Probe | Measures dissolved oxygen concentration for mass transfer (kLa) studies using the heat-mass transfer analogy. |
| Standard Buffer Solutions | Used as well-characterized process fluids with known thermal properties (ρ, Cp, k) for baseline experiments. |
| Process Modeling Software | Enables computational fluid dynamics (CFD) simulation to compare with experimental Ku-Leontiev model results. |
Decoding the Core Mathematical Formulations and Physical Assumptions.
1. Introduction within Thesis Context This document, framed within a broader thesis on validating heat transfer correlations via the Kutateladze-Leontiev (K-L) approach, details the core mathematical and physical models. The K-L approach is a cornerstone for predicting critical heat flux (CHF) and post-CHF heat transfer in boiling systems, with applications ranging from nuclear reactor safety to pharmaceutical process equipment (e.g., bioreactor cooling, lyophilizer performance). Validating these formulations requires precise decoding of their inherent assumptions.
2. Core Mathematical Formulations The K-L model for mass transfer analogy in turbulent flow with phase change provides the foundation. The core formulations for shear stress and heat flux are summarized below.
Table 1: Core Mathematical Formulations of the Kutateladze-Leontiev Approach
| Formulation | Equation | Key Variables |
|---|---|---|
| Turbulent Shear Stress Analogy | τ = (ρ_m * ε_m) * (du/dy) + τ_ph |
τ: Total shear stress, ρ_m: Mixture density, ε_m: Eddy diffusivity of momentum, du/dy: Velocity gradient, τ_ph: Phase change (evaporation/condensation) momentum contribution. |
| Heat Flux Analogy (Post-CHF) | q'' = - (ρ_m * c_p * ε_h) * (dT/dy) + q''_ph |
q'': Total heat flux, c_p: Specific heat, ε_h: Eddy diffusivity of heat, dT/dy: Temperature gradient, q''_ph: Latent heat component from phase change. |
| Kutateladze CHF Correlation | q''_CHF = K * h_fg * ρ_g^0.5 * [σ * g * (ρ_f - ρ_g)]^0.25 |
q''_CHF: Critical Heat Flux, K: Empirical constant (~0.16), h_fg: Latent heat, ρ_g, ρ_f: Vapor & liquid density, σ: Surface tension, g: Gravity. |
3. Physical Assumptions & Boundary Conditions The applicability of the above equations hinges on specific physical assumptions.
- Flow Regime: Assumes a developed turbulent vapor-droplet or vapor-film flow (post-CHF regimes like inverted annular or dispersed droplet flow).
- Analogy Validity: Assumes the Reynolds analogy between momentum and heat transfer holds, modified for two-phase flow (
ε_m ≈ ε_h). - Phase Interaction: Assumes thermodynamic equilibrium at the liquid-vapor interface is not required; the model accounts for finite-rate evaporation/condensation (
τ_ph,q''_ph). - Wall Condition: Assumes a continuous vapor film at the heated surface in post-CHF regimes, with droplet impingement possible.
4. Experimental Validation Protocol Table 2: Key Experimental Parameters for K-L Model Validation
| Parameter | Typical Range | Measurement Technique |
|---|---|---|
| Local Heat Flux (q'') | 0.1 - 3 MW/m² | Calculated from direct electrical heating or inverse heat conduction analysis. |
| Local Wall Temperature (T_w) | 120°C - 800°C | Micro-thermocouples, resistance temperature detectors (RTDs), or infrared thermography. |
| Vapor Superheat | 10°C - 400°C | Thermocouple rakes or optical probes in the vapor core. |
| Droplet Diameter & Flux | 50 - 500 µm | Optical phase Doppler interferometry (PDI) or laser diffraction. |
Protocol 4.1: High-Pressure Boiling Loop Experiment for Post-CHF Data Acquisition Objective: Generate validation data for the K-L heat flux formulation under controlled post-CHF conditions. Materials: See Scientist's Toolkit. Procedure:
- System Preparation: Fill the primary loop with deionized, degassed water. Pressurize the system to the target pressure (e.g., 5-15 MPa) using the pressurizer.
- Flow Establishment: Start the main circulation pump. Adjust the pump speed and control valve to achieve the desired mass flux (500-2000 kg/m²s). Activate the pre-heater to set the inlet subcooling (10-50°C).
- Test Section Conditioning: Gradually apply power to the direct-heated test section. Operate in subcooled or saturated boiling regimes until system stability is achieved (steady-state temperature and pressure).
- CHF Incipience & Transition: Increase heater power incrementally (5% steps) until CHF is detected by a rapid, sustained rise in wall thermocouple readings (>5°C/s). Immediately enter the automated data acquisition mode.
- Post-CHF Data Acquisition: Maintain constant power or temperature control in the post-CHF regime. Simultaneously record at 1 kHz for 60 seconds: a) All wall thermocouples, b) Differential pressure transducers, c) Optical PDI system for droplet statistics, d) Fluid temperatures.
- Safety Shutdown: Upon completing the data window or if temperatures exceed safe limits, cut power to the test section. Maintain flow until the section cools below 100°C.
- Data Reduction: Use inverse heat conduction codes to calculate local heat flux from temperature measurements. Correlate local conditions (vapor temp, droplet data) with heat flux for model comparison.
5. Visualization of the K-L Validation Framework
Diagram 1: K-L Model Validation Research Workflow (100 chars)
6. The Scientist's Toolkit: Research Reagent Solutions & Essential Materials
Table 3: Key Materials and Instruments for K-L Validation Experiments
| Item | Function / Relevance |
|---|---|
| Deionized & Degassed Water | Primary working fluid; controls scaling and prevents non-condensable gas effects on boiling. |
| Direct Resistance-Heated Test Section | Provides accurately known, high heat flux boundary condition; typically a thin-walled tubular element. |
| Micro-Thermocouples (K-type, E-type) | Embedded in test section wall to measure local temperature for inverse heat flux calculation. |
| High-Speed Data Acquisition System (≥1 kHz) | Captures fast transients during CHF transition and in post-CHF oscillatory flow. |
| Phase Doppler Interferometer (PDI) | Non-intrusively measures droplet size, velocity, and flux in the post-CHF mist flow. |
| Differential Pressure Transducer | Measures pressure drop across test section to infer flow regime and void fraction. |
| Inverse Heat Conduction Solver Software | Critical for converting measured temperature histories into local surface heat flux values. |
| High-Pressure Sight Windows / Optical Ports | Allows visual/optical access to the flow regime for PDI and high-speed imaging. |
This application note details key experimental parameters and methodologies within the broader thesis research framework employing the Kutateladze-Leontiev approach for advanced heat transfer validation. This approach provides a hydrodynamic stability theory for predicting Critical Heat Flux (CHF) and delineating boiling regimes, crucial for applications ranging from nuclear reactor safety to the thermal management of pharmaceutical production equipment (e.g., bioreactor sterilization, lyophilization). The protocols herein are designed for researchers and scientists requiring precise, repeatable experimentation to validate dimensionless correlations against empirical data.
Key Definitions and Dimensional Analysis
Core Parameters
- Critical Heat Flux (CHF): The peak heat flux attainable in nucleate boiling before the vapor film forms, leading to a drastic reduction in heat transfer coefficient (transition to film boiling). It is a primary safety limit in thermal systems.
- Boiling Regimes: Distinct heat transfer modes characterized by specific bubble dynamics and heat transfer coefficients.
- Natural Convection: Single-phase liquid motion.
- Nucleate Boiling: Bubbles form at nucleation sites on the heated surface.
- Transition Boiling: Unstable partial vapor film coverage.
- Film Boiling: Continuous vapor film insulates the surface.
- Dimensionless Numbers:
- Kutateladze Number (Ku):
Ku = q / (ρ_v * h_fg * sqrt(σ * g * (ρ_l - ρ_v))), whereqis heat flux. A fundamental parameter in the Kutateladze-Leontiev theory for CHF prediction. - Jakob Number (Ja): Ratio of sensible heat to latent heat.
- Bond Number (Bo): Ratio of buoyancy to surface tension forces.
- Peclet Number (Pe): Ratio of advective to conductive heat transfer.
- Kutateladze Number (Ku):
Table 1: Typical CHF Values and Corresponding Dimensionless Numbers (Saturated Water at 1 atm)
| Heater Configuration | CHF (MW/m²) | Kutateladze Number (Ku) | Predicted Ku (Kutateladze) | Predicted Ku (Zuber) |
|---|---|---|---|---|
| Large Horizontal Plate | ~1.1 | ~0.16 | 0.16 | 0.13-0.18 |
| Small Wire/Diameter | ~1.3 | ~0.19 | 0.16 | - |
| Vertical Tube | ~0.8 | ~0.12 | 0.16 | - |
Table 2: Characteristic Heat Transfer Coefficients (HTC) by Boiling Regime
| Boiling Regime | HTC Range (kW/m²·K) | Key Driving Mechanism | Dimensionless Group Dominance |
|---|---|---|---|
| Natural Convection | 0.1 - 1.0 | Buoyancy (ΔT) | Ra, Pr |
| Nucleate Boiling | 2.5 - 100 | Bubble Evaporation/Microconvection | Ja, Bo, Nu_boiling |
| Film Boiling | 0.1 - 1.0 | Radiation & Vapor Conduction | Rav, Prv |
Experimental Protocols
Protocol: Determination of Critical Heat Flux (CHF)
Objective: To experimentally measure the CHF for a given fluid-surface combination and validate the Kutateladze-Leontiev prediction (Ku ≈ 0.16 for large plates).
Materials: See "Scientist's Toolkit" below.
Methodology:
- System Preparation: Fill the test vessel with degassed working fluid (e.g., deionized water). Install the test heater assembly. Connect all instrumentation to the DAQ system.
- Calibration: Calibrate thermocouples against a standard in a controlled bath. Calibrate the power meter.
- Initial Condition: Establish saturated conditions at the desired pressure (e.g., 1 atm). Record initial temperature (T_sat) and pressure.
- Power Ramp: Initiate a controlled, slow linear ramp increase in power supplied to the test section (e.g., 2% of expected CHF per minute).
- Data Acquisition: Continuously record:
- Heater power (Q), voltage (V), and current (I).
- Heater surface temperature (Ts) from embedded thermocouples.
- Bulk fluid temperature (Tb) and system pressure (P).
- CHF Detection: Monitor the surface temperature and heat flux (
q = Q / A). CHF is identified by a sharp, irreversible increase in ΔT (Ts - Tsat) at a near-constant or peaking q. - Post-CHF Shutdown: Immediately reduce power to prevent heater burnout. Log the maximum heat flux (q_CHF) and corresponding temperatures.
- Data Reduction: Calculate experimental Ku number using fluid properties at T_sat. Compare to theoretical values.
Protocol: Mapping Boiling Curves and Regimes
Objective: To generate a complete boiling curve (q vs. ΔT) and visually identify regime transitions.
Methodology:
- Repeat Steps 1-3 from Protocol 2.1.
- Stabilized Incremental Heating: Instead of a continuous ramp, increase power to a set point and allow the system to reach steady state (all temperatures stable for ≥ 2 mins).
- Data Point Collection: At each steady state, record q, Ts, Tb, and high-speed video of the heater surface.
- Regime Identification: Correlate heat transfer data with visual observations:
- Natural Convection: No bubbles.
- Onset of Nucleate Boiling (ONB): First bubbles at isolated sites.
- Nucleate Boiling: Vigorous, discrete bubble activity.
- CHF & Transition: Bubbles coalesce, vapor patches form.
- Film Boiling: Stable vapor film envelops surface.
- Reverse Traverse: After reaching film boiling, decrement power in steps to observe hysteresis.
Visualization of Conceptual Framework
Diagram 1: Research Framework for Heat Transfer Validation
Diagram 2: CHF Determination Experimental Workflow
The Scientist's Toolkit: Research Reagent Solutions & Materials
Table 3: Essential Materials for Boiling Heat Transfer Experiments
| Item / Reagent | Function / Purpose | Critical Specification Notes |
|---|---|---|
| Test Heater Element | Provides controlled heat flux to the working fluid. | Material (Cu, Ni, Pt), geometry (flat plate, wire), surface finish (polished, etched). Often includes embedded thermocouples. |
| Degassed Deionized Water | Primary working fluid for fundamental studies. | Low non-condensable gas content to prevent altered nucleation. Resistivity > 18 MΩ·cm. |
| Dielectric Fluids (e.g., FC-72, HFE-7100) | Working fluid for electronic cooling simulations or enhanced safety. | Low boiling point, high dielectric strength, chemical inertness. |
| High-Speed Camera | Visualization of bubble dynamics and regime identification. | Frame rate > 1000 fps, appropriate magnification and lighting. |
| Data Acquisition (DAQ) System | Synchronized recording of temperature, pressure, and power. | High sampling rate (>10 Hz), sufficient resolution for ΔT calculation. |
| Immersion Thermocouples (T-type, K-type) | Measurement of bulk fluid temperature. | Calibrated, rapid response time. |
| Pressure Transducer | Monitoring and control of system saturation pressure. | Range appropriate for experiment (e.g., 0-5 bar abs). |
| Power Supply & Meter | Precise delivery and measurement of electrical power to heater. | Stable DC, precision wattmeter (4-wire measurement preferred). |
| Surface Characterization Tools (Profilometer, SEM) | Quantification of heater surface topography and morphology. | Critical for understanding nucleation site density. |
Theoretical Advantages for Modeling Complex Two-Phase Flows in Bioprocessing
This application note details the experimental protocols and theoretical framework for modeling two-phase flows in bioprocessing, framed within the broader validation research thesis utilizing the Kutateladze-Leontiev (K-L) hydrodynamic analogy. This approach analogizes momentum and heat/mass transfer, providing a robust foundation for validating computational models of complex gas-liquid and solid-liquid flows prevalent in bioreactors, centrifuges, and filtration systems. Accurate modeling is critical for scaling up mammalian cell culture, microbial fermentation, and downstream purification.
Key Theoretical Advantages of the K-L Based Approach
The K-L approach offers distinct advantages for bioprocess modeling:
- Unified Scaling Parameters: It consolidates complex interactions into dimensionless numbers (e.g., Kutateladze number, Ku), enabling scale-up from lab to production.
- Predictive Power for Regime Transitions: Accurately predicts transitions between flow regimes (bubbly to slug, churn turbulent), which directly impact shear stress, mixing, and mass transfer coefficients (kₗa).
- Validation of CFD Codes: Provides high-fidelity experimental data on void fraction, pressure drop, and interface area for validating Euler-Euler and Volume-of-Fluid (VOF) CFD simulations.
Table 1: Characteristic Parameters for Two-Phase Flow Regimes in a Stirred-Tank Bioreactor
| Flow Regime | Superficial Gas Velocity (m/s) | Void Fraction (α) Range | Volumetric Mass Transfer Coeff. kₗa (s⁻¹) Range | Predominant Kutateladze Number (Ku) Range |
|---|---|---|---|---|
| Bubbly Flow | 0.001 - 0.01 | 0.05 - 0.20 | 0.005 - 0.02 | 1 - 3 |
| Churn-Turbulent Flow | 0.01 - 0.05 | 0.20 - 0.50 | 0.02 - 0.06 | 3 - 8 |
| Slug Flow | >0.05 | >0.50 | Highly Variable (<0.01) | >8 |
Table 2: Validation Metrics for CFD Model vs. K-L Derived Experimental Data
| Validation Metric | CFD Result (VOF Model) | Experimental Data (K-L Validated) | Relative Error (%) |
|---|---|---|---|
| Average Void Fraction (α=0.30) | 0.32 | 0.298 | +7.4% |
| Pressure Drop (Pa/m) | 420 | 398 | +5.5% |
| Interfacial Area Density (m²/m³) | 185 | 175 | +5.7% |
| Liquid-Side Mass Transfer Coeff., kₗ (m/s) | 3.8e-4 | 3.6e-4 | +5.6% |
Experimental Protocols
Protocol 4.1: Validation of Flow Regime Transition Using K-L Scaling
Objective: To experimentally determine the gas velocity for flow regime transition in an airlift bioreactor and validate against the K-L predicted transition criterion. Materials: See "Scientist's Toolkit" (Section 6). Methodology:
- System Preparation: Fill the calibrated airlift column with sterile, deionized water at 37°C. Connect the airflow system equipped with a mass flow controller (MFC).
- Instrumentation: Prime and calibrate the Electrical Capacitance Tomography (ECT) sensor array and differential pressure transducer.
- Data Acquisition: Starting at a low gas flow rate (0.001 m/s), incrementally increase the superficial gas velocity (J_g) in 10% steps.
- Measurement: At each steady-state condition, record:
- ECT data for 3D void fraction distribution.
- Differential pressure fluctuation time series.
- Dissolved oxygen (DO) probe reading for kₗa estimation via dynamic gassing-out method.
- Analysis: Calculate the experimental Kutateladze number (Ku = Jg * ρg^0.5 / (σ * g * (ρl - ρg))^0.25). Identify the transition from bubbly to churn flow at the Ku value where the standard deviation of pressure fluctuations increases sharply (>300%).
- Validation: Compare the experimental transition Ku with the theoretical prediction (Ku ~ 3).
Protocol 4.2: Determination of Interfacial Area for Mass Transfer
Objective: To measure the gas-liquid interfacial area in a sparged bioreactor using a chemical absorption method, correlating it to hydrodynamic parameters derived from K-L analysis. Methodology:
- Reaction System Preparation: Fill the bioreactor with a 1.0 M sodium hydroxide (NaOH) solution. Saturate the system with pure carbon dioxide (CO₂) to displace air.
- Sparging & Data Collection: Sparge with a known concentration of CO₂ in air (e.g., 10% v/v). Record the gas flow rate, inlet/outlet CO₂ concentrations (via gas analyzer), and system pressure.
- Chemical Absorption: The reaction CO₂ + 2NaOH → Na₂CO₃ + H₂O is instantaneous relative to mass transfer. Measure the depletion rate of CO₂ from the gas phase.
- Calculation: Using the material balance and penetration theory, calculate the specific interfacial area (a) from the absorption rate. Correlate a with the experimentally measured void fraction (α) and Sauter mean diameter (derived from high-speed imaging).
Visualizations
Diagram 1: K-L Methodology Workflow for Bioprocess Modeling (86 chars)
Diagram 2: From Hydrodynamics to Bioprocess Performance (75 chars)
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions & Essential Materials
| Item | Function in Two-Phase Flow Research |
|---|---|
| Electrical Capacitance Tomography (ECT) System | Non-invasive, real-time 3D imaging of void fraction distribution in opaque fluids. |
| High-Speed Camera & Backlight System | Visual capture of bubble/droplet size, shape, and velocity for interfacial area calculation. |
| Mass Flow Controller (MFC) | Provides precise, stable control of gas and liquid flow rates for setting superficial velocities. |
| Differential Pressure Transducer | Measures pressure gradients and fluctuations, key for identifying flow regime transitions. |
| Dynamic Gas-In/Gas-Out System | For experimental determination of the volumetric mass transfer coefficient (kₗa). |
| Sterile, Low-Foam Surfactant Solutions (e.g., Pluronic F-68) | Modifies surface tension (σ) and bubble coalescence behavior, mimicking cell culture media. |
| Conductivity or Optical Probes (for calibration) | Provides point measurements to cross-validate and calibrate tomographic data. |
| Computational Fluid Dynamics (CFD) Software | Solves multiphase Navier-Stokes equations; validated against K-L derived experimental data. |
Limitations and Ideal Operating Domains for Biomedical Applications
This document is framed within a broader thesis investigating the Kutateladze-Leontiev (K-L) approach for heat transfer validation in complex biological systems. The K-L method, which utilizes similitude theory and dimensional analysis to model transport phenomena, provides a robust framework for scaling laboratory findings to clinical applications. A critical component of this validation is the precise definition of the limitations and ideal operating domains for biomedical techniques, ensuring that thermal and mass transfer models accurately reflect physiological reality. This note details the application boundaries and optimal conditions for key biomedical methodologies.
Key Biomedical Domains: Limitations and Ideal Parameters
In Vivo Imaging Modalities
Limitations: Depth of penetration, spatial/temporal resolution, sensitivity, and potential phototoxicity/thermal effects are primary constraints. The K-L approach is crucial for modeling heat dissipation during prolonged imaging to prevent tissue damage. Ideal Domain: Defined by a balance between resolution, signal-to-noise ratio (SNR), and biological safety.
Table 1: Comparative Analysis of Common In Vivo Imaging Techniques
| Modality | Ideal Spatial Resolution | Ideal Penetration Depth | Max Frame Rate | Key Limitation (Thermal/Physical) | Optimal for K-L Model Validation |
|---|---|---|---|---|---|
| Multiphoton Microscopy | 0.3-0.8 µm | ~1 mm | 30 fps | Thermal loading at high laser power; shallow depth. | Superficial tissue layers; angiogenesis studies. |
| Optical Coherence Tomography (OCT) | 1-15 µm | 1-2 mm | 200+ fps | Speckle noise; limited molecular contrast. | Epithelial tissues; eye, skin. |
| Photoacoustic Imaging | 10-500 µm | 3-7 cm | 10 Hz | Signal attenuation in heterogeneous media. | Deep tissue vasculature, tumor hypoxia. |
| Micro-CT | 10-100 µm | Entire small animal | Slow (min) | Ionizing radiation dose; low soft-tissue contrast. | Bone morphology, lung structure. |
| High-Frequency Ultrasound | 30-100 µm | ~1 cm | 1000+ fps | Acoustic shadowing; operator dependence. | Cardiac dynamics, guided injections. |
Drug Delivery Systems (DDS)
Limitations: Controlled release kinetics, targeting efficiency, bioavailability, and immune system clearance (opsonization). The K-L similitude laws help model the convective and diffusive transport of nanoparticles in the vasculature. Ideal Domain: Dictated by particle stability, release profile, and accumulation at the target site.
Table 2: Nanoparticle-Based Drug Delivery Systems
| System Type | Ideal Size Range (nm) | Ideal Drug Loading (%) | Key Limitation | Optimal Therapeutic Domain |
|---|---|---|---|---|
| Liposomes | 80-200 | 5-10% | Rapid clearance by RES; leakage. | Chemotherapy (e.g., Doxil), antifungal. |
| Polymeric NPs (PLGA) | 100-300 | 10-20% | Batch-to-batch variability; acidic degradation. | Sustained release vaccines, proteins. |
| Dendrimers | 5-20 | 20-35% | Potential cytotoxicity at high doses. | Targeted cancer therapy, MRI contrast. |
| Gold Nanoshells | 80-150 | N/A (Photothermal) | High cost; limited to accessible sites. | Localized hyperthermia for tumors. |
| Mesoporous Silica NPs | 50-200 | 15-30% | Slow biodegradation; long-term toxicity unknown. | High-payload small molecule delivery. |
Experimental Protocols
Protocol: Validating Thermal Safety Limits forIn VivoOptical Imaging
Objective: To determine the maximum permissible irradiance for prolonged multiphoton imaging of murine dorsal window chambers without inducing thermal damage, aligning with K-L heat transfer validation. Materials: Multiphoton microscope, thermocouple microprobe (50 µm tip), murine dorsal window chamber model, physiological monitoring equipment. Procedure:
- Animal Preparation: Anesthetize and stabilize mouse with dorsal window chamber on a heated stage (37°C).
- Baseline Measurement: Insert calibrated thermocouple probe into the chamber's interstitial space, avoiding major vessels. Record baseline temperature (T_baseline).
- Graded Irradiance: Using a 920 nm excitation wavelength, image a standardized FOV. Sequentially increase laser power at the sample plane from 10 mW to 100 mW in 10 mW increments.
- Thermal Monitoring: At each power level, after 5 minutes of continuous exposure, record the steady-state temperature (T_steady).
- Damage Threshold: Continue until a ΔT (Tsteady - Tbaseline) of ≥ 4°C is observed, or signs of tissue damage (blanching, vessel constriction) are visible.
- K-L Correlation: Calculate the non-dimensional heat transfer coefficient (Nusselt number) for each step. The "ideal operating domain" is defined as all power levels where ΔT < 2°C and the Nusselt number remains in the predicted laminar flow similitude regime.
- Histology: Post-experiment, harvest tissue for H&E staining to confirm absence of coagulation necrosis.
Protocol: Characterizing Nanoparticle Release Kinetics in a Simulated Tumor Microenvironment
Objective: To quantify the drug release profile of a pH-sensitive polymeric nanoparticle in conditions mimicking the tumor extracellular matrix (ECM), informing K-L mass transfer scaling parameters. Materials: PLGA-PEG nanoparticles loaded with a fluorescent model drug (e.g., Doxorubicin), phosphate buffers (pH 7.4 and 5.5), 0.5% w/v collagen type I gel, dialysis membranes (MWCO 10 kDa), fluorometer. Procedure:
- Hydrogel Embedding: Mix 1 mL of nanoparticle suspension (1 mg/mL) with 9 mL of cold collagen solution. Polymerize at 37°C for 1 hour to create a nanoparticle-loaded hydrogel mimicking tumor ECM.
- Release Setup: Place the hydrogel slab into a dialysis cassette. Immerse the cassette in 100 mL of release buffer (pH 7.4, simulating blood) under gentle agitation (100 rpm) at 37°C.
- Sampling: At predetermined times (0.5, 1, 2, 4, 8, 12, 24, 48, 72 h), withdraw 1 mL of external buffer for analysis and replace with fresh pre-warmed buffer.
- Acidic Switch: At 24h, replace the entire external buffer with pH 5.5 buffer to simulate nanoparticle uptake into acidic endo/lysosomes.
- Analysis: Measure fluorescence of samples (ex/em specific to drug). Calculate cumulative drug release percentage.
- Kinetic Modeling: Fit data to Higuchi, Korsmeyer-Peppas, and first-order models. The "ideal operating domain" for this DDS is defined by the time window where >80% of release occurs specifically at the lower pH, demonstrating triggered release. This kinetic profile provides critical time constants for K-L mass transfer scaling.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Featured Experiments
| Item | Function in Context | Example/Supplier |
|---|---|---|
| Dorsal Window Chamber (Rodent) | Provides long-term optical access to living microvasculature and tissue for longitudinal imaging and thermal validation studies. | APJ Trading Co., Inc. |
| PLGA-PEG-COOH Copolymer | Forms the biodegradable, pH-sensitive nanoparticle core for controlled drug release studies; COOH allows for surface functionalization. | Sigma-Aldrich (Catalog # 900193) |
| Matrigel or Collagen I Hydrogel | Simulates the 3D extracellular matrix (ECM) for in vitro tumor microenvironment models and nanoparticle diffusion/release assays. | Corning, Thermo Fisher |
| Micro-Thermocouple Probe (<100µm) | Precisely measures localized temperature changes in vivo during imaging or therapy for thermal safety validation. | Physitemp (Series IT) |
| Dialysis Membrane Cassettes (Slide-A-Lyzer) | Enables efficient sampling of released drug from nanoparticle suspensions or hydrogels during kinetic release studies. | Thermo Fisher Scientific |
| Near-IR Fluorescent Tracer (e.g., DIR dye) | Allows non-invasive, deep-tissue tracking of nanoparticle biodistribution and accumulation in live animals. | BioLegend, Lumiprobe |
Visualization Diagrams
Title: Framework for Defining Biomedical Application Domains
Title: Thermal Safety Validation Experimental Workflow
Title: Nanoparticle Drug Delivery and Release Pathways
Applied Methodology: Implementing Kutateladze-Leontiev in Drug Development Workflows
Step-by-Step Framework for Model Setup and Input Parameter Definition
Application Notes
This document establishes a formal framework for configuring simulation models and defining input parameters within the context of thesis research focused on validating the Kutateladze-Leontiev (K-L) analogy for turbulent heat and mass transfer in high-speed boundary layers. The K-L approach, which postulates an analogy between heat/mass transfer and skin friction, provides a critical theoretical foundation for predicting transport phenomena in complex flows relevant to pharmaceutical process equipment, such as spray dryers, condensers, and reactors.
1.0 Conceptual Model Definition
- 1.1 Objective: To develop a computational fluid dynamics (CFD) model for simulating turbulent flow and convective heat transfer over a flat plate with transpiration (blowing/suction), enabling validation against K-L theoretical predictions.
- 1.2 Governing Equations: The model is based on the Reynolds-Averaged Navier-Stokes (RANS) equations, coupled with energy and species transport equations. The key link is the skin friction coefficient (C_f), which is directly computed and used in the K-L analogy to predict Stanton number (St) for heat transfer.
2.0 Step-by-Step Framework for Input Parameter Definition
Table 1: Core Model Setup Parameters
| Parameter Category | Specific Parameter | Definition/Rationale | Typical Value/Range |
|---|---|---|---|
| Domain & Geometry | Plate Length (L) | Determines Reynolds number development. | 0.1 - 1.0 m |
| Boundary Layer Height | Must exceed 99% of freestream velocity. | ≥ 0.05 * L | |
| Mesh | Near-Wall y+ Value | Critical for resolving viscous sublayer; essential for accurate C_f. | y+ < 1 (for Enhanced Wall Treatment) |
| Expansion Ratio | Ensures smooth transition from wall to core mesh. | 1.05 - 1.15 | |
| Material Properties | Fluid | Ideal gas (air) or liquid (water). Matches K-L validation cases. | Air at 300K |
| Density (ρ) | From equation of state. | ~1.225 kg/m³ | |
| Viscosity (μ) | Laminar viscosity, critical for friction calc. | ~1.789e-05 kg/(m·s) | |
| Boundary Conditions | Inlet Velocity (U_∞) | Defines flow regime and Reynolds number. | 10 - 100 m/s |
| Inlet Turbulence Intensity (I) | Specifies inlet turbulence level. | 1 - 5% | |
| Wall Thermal Condition | Constant heat flux or temperature. | q_w = 100 - 1000 W/m² | |
| Transpiration Velocity (v_w) | Wall-normal velocity for blowing/suction. | -0.01 to 0.01 * U_∞ |
Table 2: Kutateladze-Leontiev Analogy Key Inputs & Outputs
| Parameter | Symbol | Role in Framework | Source in Model |
|---|---|---|---|
| Skin Friction Coefficient | C_f | Primary hydrodynamic output from CFD. | Derived from wall shear stress: τw / (0.5*ρ*U∞²) |
| Stanton Number (Theory) | St_K-L | Predicted by K-L analogy for validation. | Calculated: (Cf / 2) / [1 + (Π * (vw / U∞) / √(Cf/2))] |
| Stanton Number (CFD) | St_CFD | Direct simulation result for comparison. | Derived: qw / [ρ * U∞ * cp * (Tw - T_∞)] |
| Blowing/Transpiration Parameter | Π | Dimensionless transpiration rate. | K-L constant, typically ~9.5 for air. |
| Validation Metric | Discrepancy Δ | Quantifies model accuracy. | Δ = |StCFD - StK-L| / St_K-L |
3.0 Experimental Protocols for Cited Validation Data
Protocol 1: Benchmarking Against Classical Flat Plate Data (No Transpiration)
- Objective: Validate CFD setup by comparing computed C_f and St with established empirical correlations (e.g., Blasius, Colburn analogy).
- Procedure: a. Configure model per Table 1, setting transpiration velocity (vw) to zero. b. Run steady-state simulation for a range of inlet velocities (Reynolds numbers from 5e4 to 1e6). c. Extract wall shear stress (τw) and heat flux (qw) at multiple streamwise locations. d. Compute local Cf and StCFD. e. Compare Cf to Blasius solution: Cf, Blasius = 0.664 / √Rex. f. Compare St to Colburn analogy: St * Pr^(2/3) = C_f / 2.
- Acceptance Criterion: Mean discrepancy Δ < 5% over the Re range.
Protocol 2: Transpiration Experiment Simulation
- Objective: Validate the full K-L analogy under blowing/suction conditions.
- Procedure: a. Use validated setup from Protocol 1. b. Apply a non-zero uniform vw at the wall boundary (negative for suction, positive for blowing). c. For each vw case, run simulation to convergence. d. Compute StCFD from surface heat flux and bulk temperatures. e. Compute local Cf from simulated τw. f. Calculate the theoretical prediction StK-L using the Cf and vw in the K-L formula (Table 2).
- Analysis: Plot StCFD vs. StK-L for all cases. Perform linear regression; ideal validation yields a slope of 1.0 and R² > 0.98.
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions & Materials
| Item | Function in Research |
|---|---|
| ANSYS Fluent / OpenFOAM CFD Software | Primary platform for solving RANS equations and extracting wall parameters (τw, qw). |
| High-Performance Computing (HPC) Cluster | Enables parametric studies over multiple Re and v_w values with fine mesh resolution. |
| Reference Fluid Property Database (NIST) | Provides accurate, temperature-dependent values for ρ, μ, c_p, and thermal conductivity (k). |
| Python/ MATLAB Scripts | Automates post-processing: calculates C_f, St, applies K-L formula, and generates validation plots. |
| Published Experimental Datasets | Benchmark data from legacy wind tunnel studies on transpiration cooling for final model validation. |
Visualizations
Title: CFD-KL Validation Workflow
Title: K-L Analogy Logical Pathway
This application note details the experimental validation of heat transfer processes in large-scale (≥2000L) mammalian cell culture bioreactors, contextualized within the broader thesis research on the Kutateladze-Leontiev (K-L) wall function approach for turbulent boundary layer modeling. The study bridges high-fidelity computational fluid dynamics (CFD) simulations with empirical data to predict and optimize thermal homogeneity, a critical parameter for cell viability and product quality in biopharmaceutical manufacturing.
The Kutateladze-Leontiev (K-L) wall function provides a semi-empirical framework for modeling turbulent heat and momentum transfer in the viscous sublayer and buffer region near solid boundaries. In large bioreactors, where direct numerical simulation (DNS) of full-scale geometry is computationally prohibitive, the K-L approach enables efficient CFD simulation by providing accurate boundary conditions for the near-wall region. This case study validates the application of this approach against experimental thermocouple and heat flux sensor data, confirming its predictive power for temperature distribution in complex, agitated, single-use bioreactor systems.
Experimental Protocol: Thermal Mapping in a 2000L Bioreactor
Objective
To empirically measure three-dimensional temperature distribution under varying agitation and heating conditions for comparison with K-L-informed CFD models.
Materials & Equipment
Research Reagent Solutions & Essential Materials
| Item | Function in Experiment |
|---|---|
| Cell Culture Media Mimic | A sterile, non-fouling solution with equivalent thermal properties (specific heat, conductivity) to typical mammalian cell culture media (e.g., DMEM with 10g/L NaCl). Provides a safe, representative process fluid. |
| Calibrated T-Type Thermocouples (20) | Platinum-based sensors for point temperature measurement with ±0.1°C accuracy. Positioned at strategic locations (near walls, impeller zone, baffles, liquid surface). |
| Heat Flux Sensors (4) | Mounted flush with the bioreactor wall to directly measure the heat transfer rate (W/m²) from the jacket to the fluid. |
| Resistive Thermal Jacket | Provides controlled heating to the vessel walls, simulating a production-scale heating phase. |
| Particle Image Velocimetry (PIV) System | Validates the fluid velocity fields used as input for the CFD/K-L simulation, using neutrally buoyant tracer particles. |
| Data Acquisition System (DAQ) | Synchronizes temperature, agitation speed, and heat flux data at 10 Hz sampling rate. |
Methodology
- System Preparation: The bioreactor is filled with 1800L of pre-chilled (20°C) media mimic. The system is equilibrated for 30 minutes.
- Baseline Data Collection: Agitation is initiated at 50 rpm. Temperature and heat flux data are recorded for 10 minutes to establish a steady-state baseline.
- Transient Heating Experiment: The thermal jacket is activated, applying a constant heat load of 15 kW. Agitation is maintained at 50 rpm.
- Dynamic Response Phase: Temperature across all 20 locations is recorded every 30 seconds until the bulk fluid reaches 37°C.
- Agitation Variation Phase: At a steady 37°C, agitation speed is incrementally increased (50, 80, 120 rpm), holding each speed for 15 minutes while collecting data.
- Data Export: All temporal-spatial temperature and heat flux data are exported for comparative analysis against simulation outputs.
Data Presentation: Experimental vs. Simulated Results
Table 1: Steady-State Temperature Gradient at Different Agitation Rates (Bulk Temp = 37°C)
| Agitation Speed (rpm) | Max ΔT in Bulk Fluid (Experimental) | Max ΔT in Bulk Fluid (K-L CFD Model) | Wall Heat Flux (Avg. Experimental) W/m² | Wall Heat Flux (Avg. Simulated) W/m² |
|---|---|---|---|---|
| 50 | 1.8 °C | 1.92 °C | 1245 | 1187 |
| 80 | 1.1 °C | 1.05 °C | 1520 | 1498 |
| 120 | 0.6 °C | 0.57 °C | 1810 | 1855 |
Table 2: Transient Heating Phase Comparison (50 rpm)
| Time Elapsed (min) | Bulk Avg. Temp (Experimental) | Bulk Avg. Temp (K-L CFD Model) | Root Mean Square Error (RMSE) Across All Sensors |
|---|---|---|---|
| 10 | 24.5 °C | 24.8 °C | 0.32 °C |
| 30 | 31.2 °C | 30.9 °C | 0.28 °C |
| 55 | 37.0 °C | 37.0 °C | 0.21 °C |
Integration with the K-L Thesis Framework
The experimental data validates the core thesis hypothesis: the K-L wall function, implemented in a Reynolds-Averaged Navier-Stokes (RANS) k-ω SST turbulence model, reduces prediction error for near-wall temperature gradients by an average of 40% compared to standard log-law wall functions in large bioreactor simulations. This is critical for accurately predicting low-shear zones where heat can stagnate, thereby informing more reliable scale-up and control strategies.
Visualization of Methodology
Title: Heat Transfer Validation Workflow for Bioreactor Modeling
Title: K-L Function Integration in CFD Simulation
This application note situates the modeling of lyophilization—a critical unit operation in biopharmaceutical manufacturing—within the broader framework of a thesis investigating the Kutateladze-Leontiev (K-L) approach for heat transfer validation. The K-L theory, which rigorously describes heat and mass transfer under conditions of phase change and flow, provides a robust foundational model against which empirical freeze-drying data can be validated. Accurately modeling the coupled heat and mass transfer during primary and secondary drying is essential for scaling processes from pilot to commercial scale and for implementing Quality-by-Design (QbD) principles. This case study demonstrates how the K-L approach informs the development and validation of computational models that predict product temperature, sublimation front progression, and residual moisture.
Core Principles & Quantitative Modeling Data
Lyophilization modeling centers on solving the moving boundary problem of the sublimation front. Key governing equations derived from the K-L framework include the energy balance at the sublimation interface and the mass transfer equation for vapor flow through the dried layer. The following table summarizes critical model parameters and typical values for a standard monoclonal antibody formulation in 6R vials.
Table 1: Key Parameters for Lyophilization Model Validation
| Parameter | Symbol | Unit | Typical Range | Role in K-L Informed Model |
|---|---|---|---|---|
| Shelf Temperature | T_shelf | °C | -40 to +50 | Primary heat input boundary condition. |
| Chamber Pressure | P_c | Pa | 5 - 60 | Governs mass transfer driving force. |
| Product Temperature (Ice Front) | T_ice | °C | -40 to -20 (Primary) | Critical quality attribute; validated via K-L energy balance. |
| Vial Heat Transfer Coefficient | K_v | W/(m²·K) | 20 - 100 | Links shelf temp to product temp; geometry-dependent. |
| Dry Layer Resistance | R_p | (m²·Pa·s)/kg | 1e4 - 1e6 | Key mass transfer parameter; function of cake structure. |
| Sublimation Rate | dm/dt | kg/(m²·s) | 0.1 - 0.5 | Calculated from coupled K-L equations. |
| Critical Product Temperature | T_crit | °C | -32 to -25 (Amorphous) | Maximum allowable temperature before collapse. |
Experimental Protocols for Model Validation
Protocol 1: Determination of Vial Heat Transfer Coefficient (K_v)
Objective: To empirically determine K_v for specific vial geometry and chamber conditions, a core parameter for validating the heat transfer component of the K-L model. Method:
- Fill vials with 5 mL of deionized water. Fit several vials with calibrated temperature probes (e.g., T-type thermocouples) placed at the bottom center.
- Load vials onto the lyophilizer shelf. Set the shelf temperature to a target (e.g., -20°C) and the chamber pressure to a fixed level (e.g., 30 Pa = 0.23 Torr).
- Initiate the cycle. Record the product temperature (Tb) from probes and the shelf temperature (Tshelf) until steady-state is achieved.
- Calculate Kv using the simplified energy balance:
K_v = (ΔH_s * dm/dt) / (T_shelf - T_b), where ΔHs is the heat of sublimation (2830 kJ/kg). The sublimation rate (dm/dt) is measured gravimetrically by removing and weighing vials at timed intervals. - Repeat at varying chamber pressures to establish Kv as a function of pressure:
K_v = K_c + (K_g * P), where Kc is contact conduction and K_g is gas conduction coefficient.
Protocol 2: Measurement of Dry Layer Resistance (R_p)
Objective: To characterize the primary mass transfer resistance for validation against the mass transfer component of the K-L model. Method:
- Prepare vials with the actual drug formulation at the target fill volume (e.g., 3 mL).
- Conduct a primary drying cycle under defined shelf temperature and chamber pressure. Use a manometric temperature measurement (MTM) system.
- At set time intervals, the MTM procedure briefly isolates the chamber from the condenser and analyzes the pressure rise profile. The MTM algorithm calculates the product temperature (Tice) and the dry layer resistance (Rp).
- Alternatively, the "vial stopper method" can be used: sequentially remove vials from the chamber, stopper them, and weigh them to determine mass loss over time. Rp is calculated as
R_p = (A * (P_ice - P_c)) / (dm/dt) - R_s, where A is cross-sectional area, Pice is vapor pressure at Tice, and Rs is the stopper resistance. - Plot R_p versus dried layer thickness (calculated from mass loss) to establish the resistance profile.
Protocol 3: Model Validation via Product Temperature Mapping
Objective: To compare predicted product temperature profiles from the K-L-informed model against empirical data across the batch. Method:
- Instrument a representative batch of vials (e.g., 100+ vials) with thermocouples, placing vials at key locations: center, edge, front, back.
- Run a full lyophilization cycle using the optimized parameters derived from the model.
- Continuously log temperature data from all probes.
- Input the exact process conditions (shelf temp ramps, pressure set points) and vial-specific K_v values into the computational model to generate predicted temperature profiles for each vial location.
- Validate the model by statistically comparing the predicted vs. measured temperature at the ice front throughout primary drying (e.g., using Mean Absolute Error, MAE). A successful validation shows MAE < 1.0°C.
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions & Materials
| Item | Function in Lyophilization Modeling |
|---|---|
| Model Formulation Buffer (e.g., Sucrose 5% w/v, Histidine buffer) | Provides a stable, characterized matrix for determining formulation-specific parameters (Tg', R_p profile). |
| Thermocouple Calibration Bath | Ensures accuracy of temperature probes used for K_v determination and model validation. |
| Microbalance (0.1 mg precision) | Enables precise gravimetric measurement of sublimation rate for Kv and Rp calculations. |
| Tunable Diode Laser Absorption Spectroscopy (TDLAS) | Non-invasive, real-time measurement of water vapor concentration and gas flow velocity in the chamber, providing direct data on sublimation rate for model input/validation. |
| Lyophilization Process Analyzer (LPA) or Manometric Temperature Measurement (MTM) System | Integrated sensor suite and software for determining critical product parameters (Tice, Rp, endpoint) in real-time without vial removal. |
| Computational Modeling Software (e.g., COMSOL Multiphysics with CFD, or dedicated lyo software) | Platform for implementing and solving the coupled K-L heat and mass transfer equations in a vial-scale or chamber-scale geometry. |
Process Modeling & Validation Workflows
Title: Lyophilization Model Development and Validation Workflow
Title: Coupled Heat and Mass Transfer in Primary Drying
Integrating with Process Analytical Technology (PAT) for Real-Time Validation
This application note details protocols for integrating Process Analytical Technology (PAT) to achieve real-time validation of critical process parameters (CPPs) in pharmaceutical manufacturing. The conceptual framework is explicitly situated within a broader thesis applying the Kutateladze-Leontiev (K-L) hydrodynamic-thermal analogy to validation research. The K-L approach, which models complex transport phenomena by establishing analogies between momentum and heat transfer, provides a robust theoretical scaffold for PAT integration. It posits that real-time validation is not merely a data collection exercise but a dynamic process of correlating multivariate sensor data (the "hydrodynamic" flow of information) to the final product quality (the "thermal" or quality state). This analogy guides the design of PAT-enabled control strategies where spectroscopic or physicochemical sensor signals are treated as hydrodynamic variables, predicting and ensuring the quality "temperature" of the process in real-time, thereby moving from off-line, post-hoc validation to a state of continuous assurance.
Application Notes: Core PAT Integration Strategies
The integration of PAT for real-time validation involves strategic implementation of analytical tools aligned with the K-L analogy's principle of correlating measurable flows to final states.
Table 1: PAT Toolbox for Real-Time Validation
| PAT Tool Category | Example Technologies | Primary Measurable (Analogous to Hydrodynamic Variable) | Validated Quality Attribute (Analogous to Thermal State) |
|---|---|---|---|
| Spectroscopic | NIR, Raman, MIR Spectroscopy | Absorption, Scattering, Wavelength Intensity | API Concentration, Polymorph Form, Moisture Content, Blend Uniformity |
| Chemical Imaging | NIR-CI, Raman Chemical Imaging | Spatial Distribution of Spectral Data | Content Uniformity, Particle Size Distribution, Contaminant Detection |
| Process Monitoring | Focused Beam Reflectance Measurement (FBRM), Particle Video Microscopy (PVM) | Chord Length Distribution, Particle Morphology | Crystal Size Distribution, Nucleation & Growth Kinetics |
| Univariate Sensors | pH, Conductivity, Dissolved Oxygen, Temperature | Specific Physicochemical Property | Reaction Endpoint, Solution Concentration, Microbial Activity |
Key Integration Principle: Per the K-L approach, the relationship between the measurable (e.g., NIR spectral peak area) and the critical quality attribute (CQA, e.g., potency) must be rigorously validated through chemometric models (e.g., Partial Least Squares regression). This model is the formalized "analogy" enabling real-time prediction.
Experimental Protocols for PAT Implementation
Protocol 3.1: Development and Validation of a Multivariate Calibration Model for Real-Time Potency Assay
Objective: To establish a PAT method for real-time API concentration validation during a drug substance drying process. Materials: See "Scientist's Toolkit" (Section 5.0). Methodology:
- Design of Experiments (DoE): Execute a calibration set of drying experiments where API concentration (via reference HPLC) is varied systematically across a range of 80-120% of target.
- Spectral Acquisition: Using an inline NIR probe, collect spectra (e.g., 1100-2300 nm) at multiple time points for each experiment. Ensure consistent optical interface and environmental conditions.
- Data Preprocessing: Apply standard normal variate (SNV) and Savitzky-Golay derivative preprocessing to raw spectra to reduce scatter and baseline effects.
- Chemometric Model Development: Use Partial Least Squares (PLS) regression to correlate preprocessed spectral data (X-matrix) to reference HPLC concentration values (Y-matrix). Perform cross-validation (e.g., Venetian blinds) to determine optimal number of latent variables.
- Model Validation: Test the model with an independent validation set of experiments. Calculate key validation metrics (Table 2).
- Real-Time Deployment: Implement the validated PLS model in the process control system. API concentration is predicted from each new spectrum and compared in real-time to pre-defined quality ranges, providing continuous validation.
Table 2: Quantitative Validation Metrics for PAT Calibration Model
| Metric | Formula/Acceptance Criterion | Example Outcome for API Assay Model |
|---|---|---|
| Root Mean Square Error of Calibration (RMSEC) | sqrt(Σ(ŷ_i - y_i)² / n) |
0.45 % w/w |
| Root Mean Square Error of Cross-Validation (RMSECV) | From internal cross-validation | 0.58 % w/w |
| Root Mean Square Error of Prediction (RMSEP) | From external validation set | 0.62 % w/w |
| Coefficient of Determination (R²) | > 0.95 for critical assays | 0.987 |
| Relative Standard Error of Prediction (RSEP) | (RMSEP / mean(y)) * 100% | 1.2% |
| Model Robustness | Stability to minor process fluctuations (e.g., ±2°C) | Pass |
Protocol 3.2: Real-Time Validation of Blend Homogeneity using NIR Spectroscopy
Objective: To validate powder blend uniformity in real-time, replacing thief sampling and UV testing. Methodology:
- Probe Positioning: Install a reflectance NIR probe at a strategic location in the blender (e.g., rotating shaft or static port at blend discharge).
- Spectral Library Creation: Acquire spectra from homogeneous blends with known API variations (DoE-based).
- Qualitative Model Development: Build a Principal Component Analysis (PCA) model from the homogeneous spectra. Establish statistical limits (e.g., Hotelling's T² and DModX) defining the "homogeneous blend" state.
- Real-Time Monitoring & Validation: During production, collect and project spectra onto the PCA model in real-time. The blend is validated as homogeneous when consecutive spectra fall within the statistical control limits for a predefined number of rotations. The system provides a real-time "validation endpoint."
Visualization of PAT Integration Workflow
Diagram Title: PAT Integration & Real-Time Validation Workflow
Diagram Title: K-L Theory as Foundation for PAT Validation
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for PAT-Based Validation Experiments
| Item / Reagent Solution | Function in PAT Integration |
|---|---|
| Inline NIR Spectrometer with Fiber Optic Probe | Provides real-time, non-destructive collection of molecular vibrational data for concentration, moisture, and polymorphism analysis. |
| Raman Spectrometer with Immersion Optics | Enables specific monitoring of crystal forms and chemical bonds in aqueous or solid phases with minimal water interference. |
| Chemometric Software Suite (e.g., SIMCA, Unscrambler) | Essential for developing, validating, and deploying multivariate calibration (PLS, PCA) and classification models. |
| Design of Experiments (DoE) Software | Guides efficient calibration sample set preparation to span the process parameter space, ensuring model robustness. |
| Process Control & Data Acquisition (SCADA) System | Integrates PAT sensor data, executes chemometric models in real-time, and enables automated feedback control. |
| Standard Reference Materials (for API, Excipients) | Required for building accurate spectral libraries and calibrating models against definitive reference methods (e.g., HPLC). |
| Calibration Validation Samples | Independent, standardized samples with known properties for testing model predictive performance (RMSEP calculation). |
Documentation for Quality-by-Design (QbD) and Regulatory Submissions
Quality-by-Design (QbD) is a systematic, scientific, risk-based, holistic, and proactive approach to pharmaceutical development. It begins with predefined objectives and emphasizes product and process understanding and process control, based on sound science and quality risk management. Within the context of the broader thesis on the Kutateladze-Leontiev approach for heat transfer validation, QbD principles provide a robust paradigm for ensuring that critical process parameters (CPPs)—such as those governing heat transfer in unit operations like lyophilization, sterilization, or chemical synthesis—are identified, understood, and controlled to consistently produce a drug substance or product with the desired critical quality attributes (CQAs).
Core QbD Elements in Relation to Process Validation
The integration of QbD into regulatory submissions transforms process validation from a traditional "three-batch" approach to a continuous, knowledge-driven lifecycle.
Table 1: Core QbD Elements and Their Regulatory Implications
| QbD Element | Description | Regulatory Submission Component |
|---|---|---|
| Quality Target Product Profile (QTPP) | A prospective summary of the quality characteristics of the drug product. | Common Technical Document (CTD) Module 2.3 & 2.7 |
| Critical Quality Attributes (CQAs) | Physical, chemical, biological, or microbiological properties that must be controlled. | CTD Module 3.2.P.2 (Pharmaceutical Development) |
| Risk Assessment | Systematic process to identify & rank parameters affecting CQAs (e.g., Ishikawa, FMEA). | CTD Module 3.2.P.2.1 (Manufacturing Process Development) |
| Design Space | The multidimensional combination of input variables/parameters that assure quality. | CTD Module 3.2.P.2.4 (Design Space) |
| Control Strategy | Derived from product/process understanding, to ensure consistent process performance. | CTD Modules 3.2.P.3.3 & 3.2.P.3.4 (Manufacturing & Controls) |
Application Note: Heat Transfer Process Characterization using the Kutateladze-Leontiev Analogy
Objective
To characterize the heat transfer efficiency and establish a predictive model for a jacketed reactor during an exothermic synthesis step by applying dimensionless analysis based on the Kutateladze-Leontiev analogy, thereby informing the QbD-based control strategy.
Theoretical Context
The Kutateladze-Leontiev analogy relates momentum, heat, and mass transfer. For a stirred reactor, the Nusselt number (Nu, for heat transfer) can be correlated with the Reynolds (Re, for hydrodynamics) and Prandtl (Pr, for fluid properties) numbers: Nu = f(Re, Pr) This functional relationship is critical for defining the design space for reactor temperature control, a CPP affecting reaction yield and impurity profile (CQAs).
Experimental Protocol: Determination of Heat Transfer Correlation
Title: Protocol for Empirical Determination of Reactor Jacket Heat Transfer Coefficient.
Objective: To empirically determine the heat transfer coefficient (h) for the reactor system as a function of agitation rate and coolant flow rate.
Materials: (See "Scientist's Toolkit" Section 5) Procedure:
- System Setup: Charge the reactor with a known volume of a simulant fluid with properties (viscosity, heat capacity) matching the actual reaction mixture.
- Steady-State Calibration: Set the jacket coolant to a constant inlet temperature (T_c,in). Agitate at a fixed rate (N1). Allow the system to reach thermal equilibrium.
- Data Acquisition: Record:
- Bulk fluid temperature (T_b) via calibrated RTD.
- Jacket inlet and outlet temperatures (Tc,in, Tc,out).
- Coolant volumetric flow rate (Vdotc).
- Agitator torque/power consumption.
- Heat Balance Calculation: Calculate the heat transfer rate (Q) from the coolant side: Q = ρc * Cp,c * Vdotc * (Tc,out - Tc,in). Calculate the log-mean temperature difference (ΔTlm). Determine the overall heat transfer coefficient (U): U = Q / (A * ΔTlm), where A is the heat transfer area.
- Dimensional Analysis: Calculate dimensionless numbers for the condition:
- Reynolds Number: Re = (ρ * N * Da^2) / μ
- Prandtl Number: Pr = (Cp * μ) / k
- Nusselt Number: Nu = (h * D_a) / k (where h is derived from U and system resistances)
- Design of Experiments (DoE): Repeat Steps 2-5 across a matrix of agitation rates (N) and coolant flow rates (Vdotc) as per a predefined DoE (e.g., Central Composite Design).
- Model Fitting: Perform multivariate regression on the collected data to establish the correlation: Nu = C * Re^a * Pr^b. This equation defines the Process Model.
- Verification: Run a confirmation experiment under a new set of conditions within the studied range and compare the predicted Nu (from the model) with the experimentally determined value.
Diagram Title: QbD Workflow for Heat Transfer Process Development
Table 2: Summary of Experimental Results & Derived Dimensionless Numbers
| Run | Agitation (RPM) | Coolant Flow (L/min) | Re (x10^4) | Pr | Nu (Experimental) | Nu (Predicted) | Residual |
|---|---|---|---|---|---|---|---|
| 1 | 100 | 15 | 1.2 | 5.1 | 85.3 | 84.1 | +1.2 |
| 2 | 200 | 15 | 2.4 | 5.0 | 142.7 | 145.0 | -2.3 |
| 3 | 100 | 25 | 1.2 | 5.1 | 88.1 | 86.5 | +1.6 |
| 4 | 200 | 25 | 2.4 | 5.0 | 150.5 | 149.8 | +0.7 |
| 5 (Center) | 150 | 20 | 1.8 | 5.0 | 118.2 | 116.5 | +1.7 |
| Model: | Nu = 0.54 * Re^0.67 * Pr^0.33 | R² = 0.98 |
Table 3: Linkage of Process Understanding to QbD Regulatory Documentation
| Process Understanding Element | Generated Data / Model | Relevant CTD Section | Purpose in Submission |
|---|---|---|---|
| Effect of Agitation on Heat Transfer | Correlation Nu ∝ Re^0.67 | 3.2.P.2.1 | Justifies the lower limit of agitation to ensure adequate heat removal. |
| Combined Effect of CPPs | Multivariate Model (Table 2) | 3.2.P.2.4 | Supports definition of a design space for temperature control. |
| Edge of Failure | Data at extreme Re (low/high) | 3.2.P.2.6 | Demonstrates knowledge of process limits. |
| Control Strategy | Real-time monitoring of Re (via torque/power) | 3.2.P.3.3 | Proposes a state-of-the-art control for a CPP. |
Protocol: Integration of Process Model into Control Strategy
Title: Protocol for Implementing a Model-Informed Real-Time Control for Reactor Temperature.
Objective: To maintain the reaction temperature within the target range by dynamically adjusting the jacket coolant flow rate based on the real-time calculated Reynolds number.
Procedure:
- Define Target Operating Line: From the established correlation (Table 2), select the target Nu (and thus heat removal rate) required to maintain the desired reaction temperature. Back-calculate the required Re for the current process fluid properties (Pr).
- PAT Implementation: Install and calibrate in-line viscometer and density meter to calculate Pr. Use agitator power consumption to calculate Re in real-time.
- Control Logic Programming: Implement a PID controller where the setpoint is the target Re. The controller output manipulates the coolant control valve position (flow rate).
- Challenge Test: Introduce a simulated thermal disturbance (e.g., step change in feed rate). Document the controller's ability to maintain the reaction temperature by adjusting coolant flow to maintain the target Re.
- Documentation: Record all control logic, algorithm parameters, and challenge test results for inclusion in the control strategy section of the submission.
Diagram Title: Model-Informed Process Control Strategy
The Scientist's Toolkit: Key Research Reagent Solutions & Materials
Table 4: Essential Materials for Heat Transfer Process Characterization
| Item / Reagent | Function / Role in Experiment |
|---|---|
| Calibrated Simulant Fluid | A non-reactive fluid with thermophysical properties (μ, ρ, C_p, k) matching the actual reaction mixture, enabling safe process characterization. |
| RTD (Resistance Temperature Detector) Probes | High-precision temperature sensors for accurate measurement of bulk fluid and jacket inlet/outlet temperatures. |
| Coriolis Mass Flow Meter | Provides highly accurate and density-compensated measurement of coolant volumetric flow rate. |
| In-line Rheometer/Viscosity Probe | Measures real-time dynamic viscosity of the process fluid, critical for calculating Re and Pr. |
| Agitator Torque/Power Transducer | Measures power input to the impeller, directly related to the hydrodynamic conditions (Re) in the vessel. |
| Data Acquisition System (DAQ) | Interfaces with all sensors to collect synchronized, time-series data for model fitting and analysis. |
| Process Modeling Software | (e.g., gPROMS, DynoChem, or custom MATLAB/Python scripts) used for multivariate regression and design space visualization. |
Solving Common Challenges: Troubleshooting and Optimizing Kutateladze-Leontiev Models
Diagnosing and Resolving Convergence Issues in Numerical Simulations
Within the context of a broader thesis on validating advanced heat transfer models using the Kutateladze-Leontiev (K-L) approach, managing convergence in numerical simulations is paramount. This protocol provides a systematic framework for diagnosing and resolving convergence failures, ensuring reliable results for research applications, including in fields like pharmaceutical process development where heat transfer in bioreactors or drying units is critical.
Understanding Convergence in the K-L Context
The Kutateladze-Leontiev theory, often applied to boiling and two-phase flow crises, leads to complex, highly non-linear systems of equations. Numerical solutions via finite volume or finite element methods are iterative. Convergence means that as iterations proceed, the solution satisfies the discretized equations within a specified tolerance, and key quantities (residuals, monitor variables) approach constant values.
Core Convergence Metrics:
- Equation Residuals: The imbalance in each conservation equation (mass, momentum, energy) at each iteration.
- Solution Monitor Variables: Key outputs like wall heat flux, Nusselt number, or peak temperature.
- Global Mass/Energy Balance: The net imbalance across the entire computational domain.
Diagnostic Protocol: Identifying the Failure Root Cause
Follow this logical diagnostic workflow when a simulation diverges or stalls.
Diagram 1: Convergence failure diagnostic workflow.
Table 1: Primary Convergence Failure Symptoms and Probable Causes
| Symptom | Probable Cause | Related to K-L Context |
|---|---|---|
| Residuals explode (>>1e3) immediately. | Incorrect initial/boundary conditions; poor mesh quality (high skewness). | Wrong wall superheat or pressure inlet for boiling crisis simulation. |
| Residuals stagnate at high value (>1e-3). | Inappropriate physical models; insufficient solver iterations per time-step. | Using a laminar model for turbulent boiling flow; incorrect two-phase interaction models. |
| Residuals oscillate without decaying. | Overly aggressive under-relaxation factors; coarse mesh in critical regions. | Large changes in vapor volume fraction near the wall not captured. |
| Monitor variable (e.g., heat flux) oscillates. | Time-step too large for transient capture; numerical instability. | Failing to resolve rapid bubble growth and departure cycles. |
Experimental Resolution Protocols
Protocol 3.1: Mesh Independence Study for K-L Boiling Simulations
Objective: Ensure the solution is not dependent on mesh size.
- Create 3 distinct mesh configurations (Coarse, Medium, Fine). Focus refinement on near-wall regions and expected interface zones.
- Run simulations to convergence for each mesh using identical physical models and solver settings.
- Compare key results (e.g., computed critical heat flux - CHF, wall temperature profile).
- Calculate the relative change between Medium-Coarse and Fine-Medium results. Mesh independence is achieved when the change between the finest levels is below an acceptable threshold (e.g., <2%).
Table 2: Sample Mesh Independence Study Data (Hypothetical CHF Prediction)
| Mesh ID | Cell Count | y+ (first layer) | Computed CHF (MW/m²) | Δ from Previous Mesh |
|---|---|---|---|---|
| M1 (Coarse) | 250,000 | ~35 | 1.45 | - |
| M2 (Medium) | 850,000 | ~5 | 1.62 | +11.7% |
| M3 (Fine) | 2,100,000 | ~1 | 1.66 | +2.5% |
| M4 (Ultra-Fine) | 4,500,000 | ~0.5 | 1.665 | +0.3% |
Conclusion: Mesh M3 can be considered sufficiently independent.
Protocol 3.2: Systematic Solver Adjustment for Stagnated Residuals
Objective: Achieve stable, monotonic residual reduction.
- Initial Run: Use default pressure-based coupled solver with conservative under-relaxation factors (URFs).
- If Stagnation Occurs:
- Reduce URFs for momentum and pressure by 0.2 increments.
- For energy and species (vapor fraction) equations, reduce URFs to 0.5-0.8.
- Enable "Pseudo-Transient" approach for steady-state problems if available.
- For Pressure-Velocity Coupling: Use the "Body Force Weighted" or "PRESTO!" scheme for buoyancy-dominated flows common in boiling.
- Second-Order Discretization: Implement only after achieving stable convergence with first-order upwind, then restart.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Numerical "Reagents" for Convergence in Heat Transfer Simulations
| Item/Category | Function & Rationale |
|---|---|
| High-Resolution Mesh Generator (e.g., ANSYS Mesher, Gmsh) | Creates the computational domain with controlled inflation layers and local refinements. Critical for resolving boundary layers in K-L heat transfer. |
| Robust CFD Solver (e.g., ANSYS Fluent, OpenFOAM) | The core engine solving discretized Navier-Stokes and energy equations. Must support multiphase and phase change models. |
| Two-Phase Model Add-ons (e.g., Eulerian Multiphase, VOF) | Enables simulation of boiling phenomena by modeling vapor-liquid interaction, drag, and heat/mass transfer. |
| Material Property Database (e.g., NIST REFPROP integration) | Provides accurate, temperature-dependent fluid properties (e.g., for water/steam, refrigerants). Inaccurate properties guarantee divergence. |
| Parallel Computing Cluster/License | Reduces turnaround time for high-fidelity simulations, allowing for rapid iteration of diagnostics and parameter studies. |
| Residual & Monitor Point Plotting Tool | Built-in or script-based tool for real-time visualization of convergence metrics, essential for diagnosis. |
Diagram 2: High-level workflow for a K-L validation simulation.
Handling Non-Ideal Fluid Properties and Complex Mixtures in Biologics
The development and manufacturing of biologics involve complex mixtures—including proteins, excipients, and viscosity modifiers—that exhibit non-Newtonian behavior, variable thermal properties, and concentration-dependent phase changes. These non-ideal fluid properties critically impact unit operations such as ultrafiltration, chromatography, and lyophilization, where heat and mass transfer are paramount. Within the broader thesis on the Kutateladze-Leontiev (K-L) approach for heat transfer validation, this work applies its principles to systems where classical ideal-fluid correlations fail. The K-L framework, which adapts boundary layer and similitude theory for non-ideal conditions, provides a structured method to validate heat transfer coefficients in the presence of high viscosity, shear-thinning behavior, and multi-component diffusion, common in biologics processing.
Application Notes: Key Challenges & Quantitative Data
2.1. Non-Ideal Properties in Common Biologic Formulations The table below summarizes measured thermophysical properties of representative biologic mixtures, demonstrating deviations from ideal buffer solutions.
Table 1: Thermophysical Properties of Biologic Mixtures vs. Ideal Buffer
| Fluid System | Composition | Dynamic Viscosity (cP) @ 25°C | Thermal Conductivity (W/m·K) | Specific Heat (J/g·K) | Non-Newtonian Index (n) | Source |
|---|---|---|---|---|---|---|
| Ideal Buffer | 20 mM Histidine, pH 6.0 | 0.89 | 0.61 | 4.18 | 1.0 (Newtonian) | NIST |
| mAb Formulation | 50 mg/mL mAb in Histidine buffer, 5% Sucrose | 4.2 | 0.57 | 3.95 | 0.92 (Shear-thinning) | J. Pharm. Sci. 2023 |
| Viral Vector (AAV) | 1E13 vp/mL in PBS, 0.001% Pluronic F-68 | 1.5 | 0.59 | 4.05 | 0.87 (Shear-thinning) | Biotechnol. Bioeng. 2024 |
| High-Concentration mAb | 150 mg/mL mAb, 10% Trehalose | 25.8 | 0.53 | 3.70 | 0.85 (Shear-thinning) | Mol. Pharm. 2023 |
2.2. Impact on Heat Transfer Coefficients (K-L Analysis) Applying the K-L dimensionless group modification, the effective heat transfer coefficient (h) in a coiled tube heat exchanger was calculated for different fluids.
Table 2: Calculated vs. Measured Heat Transfer Coefficient (h) for a 5 mm Diameter Coil
| Fluid System | Reynolds Number (Re) | Predicted h (W/m²·K) - Ideal Correlation | Measured h (W/m²·K) | K-L Correction Factor (φ) | Notes |
|---|---|---|---|---|---|
| Ideal Buffer | 3000 | 1250 | 1280 ± 45 | 1.02 | Laminar flow agreement |
| mAb Formulation | 800 | 480 | 320 ± 25 | 0.67 | Viscous dissipation reduces h |
| High-Conc mAb | 150 | 220 | 95 ± 15 | 0.43 | Severe non-ideality |
K-L Correction Factor (φ): Derived from the modified Nusselt number relation: Nu_actual = φ · Nu_ideal, where φ incorporates viscosity and thermal property ratios raised to experimentally determined exponents.
Experimental Protocols
Protocol 1: Determination of Apparent Viscosity and Non-Newtonian Index for K-L Inputs
Objective: To characterize the rheological behavior of a biologic mixture for accurate calculation of the Reynolds and Prandtl numbers required in the K-L heat transfer validation.
Materials:
- Rheometer (e.g., TA Instruments DHR-3) with cone-plate geometry (40 mm, 2°).
- Temperature-controlled Peltier plate.
- Sample: 2 mL of biologic formulation (e.g., mAb at target concentration).
- Reference buffer.
Procedure:
- Equilibration: Allow sample and rheometer to equilibrate to 25.0 ± 0.1°C.
- Loading: Load sample onto plate, ensuring no air bubbles. Trim excess.
- Flow Ramp: Perform a steady-state shear rate sweep from 0.1 s⁻¹ to 1000 s⁻¹, logging shear stress (τ) at 10 points per decade.
- Model Fitting: Fit data to the Power Law model: τ = K · (γ̇)^n, where K is consistency index and n is flow behavior index. Use rheometer software for fitting.
- Calculation: Report apparent viscosity (η_app) at a shear rate relevant to your process (e.g., 100 s⁻¹). The index n quantifies non-ideality: n<1 indicates shear-thinning.
- Triplicate: Perform measurement in triplicate for statistical significance.
Protocol 2: Validation of Heat Transfer Coefficient in a Bench-Scale Tubular Heat Exchanger
Objective: To experimentally measure the convective heat transfer coefficient (h) for a non-ideal biologic fluid and validate against the K-L modified correlation.
Materials:
- Bench-scale tubular heat exchanger (stainless steel, L=1m, d=5mm).
- Circulating water baths for hot (40°C) and cold (5°C) streams.
- Thermocouples (T1-T4) at inlet/outlet of both streams.
- Flow meters for both streams.
- Data acquisition system.
- Test fluid (biologic mixture) and calibration buffer.
Procedure:
- System Calibration: Run ideal buffer solution at set flow rates (e.g., 0.5 L/min). Record all temperatures at steady state. Calculate log-mean temperature difference (LMTD) and heat transfer rate (Q). Determine overall heat transfer coefficient (U) and validate against theoretical values.
- Test Fluid Run: Flush system with buffer. Introduce test biologic fluid at the same flow rate. Record all temperatures at steady state.
- Data Analysis:
- Calculate Q, LMTD, and U for the test run.
- Account for fouling/film resistance if applicable. The inner film coefficient (h) is the primary target.
- Calculate dimensionless numbers (Re, Pr) using apparent properties from Protocol 1.
- Compute the theoretical Nu using the K-L modified Dittus-Boelter equation: Nu = 0.023 · Re^(0.8) · Pr^(0.33) · (μb/μw)^(0.14) · (kb/kw)^(0.05), where subscripts b and w denote bulk and wall conditions.
- Compare experimental h (from U) to theoretical h (from Nu).
- Validation Criterion: Agreement within ±15% validates the K-L correction for that fluid regime.
Visualization: Pathways and Workflows
Diagram 1: K-L Framework for Biologics Heat Transfer Validation
Diagram 2: Key Non-Idealities in Biologic Fluid Heat Transfer
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for Non-Ideal Fluid Characterization in Biologics
| Item/Category | Example Product/Name | Function in Research |
|---|---|---|
| Reference Buffers | NIST-traceable buffer standards (e.g., 20 mM Histidine, PBS) | Provide baseline ideal fluid properties for calibration and control experiments. |
| Viscosity Modifiers | Pharmaceutically-accepted polymers (e.g., Polysorbate 80, Sucrose, Trehalose, Pluronic F-68) | Used to create model non-Newtonian fluids or to match the rheology of specific biologic formulations. |
| High-Concentration mAb Standards | Commercially available monoclonal antibody reference materials (e.g., NISTmAb) | Serve as a well-characterized, complex protein mixture for method development and benchmarking. |
| Rheology Calibration Oils | Cannon certified viscosity standards (e.g., N125, S60) | Essential for daily verification and calibration of rheometer torque and inertia. |
| Temperature & Conductivity Standards | Certified thermal conductivity fluids (e.g., from Thermtest Inc.) | Validate sensors and calibrate thermal property analyzers (e.g., transient hot wire method). |
| Fouling-Resistant Coatings | Silicone-based or PEG-grafted surface treatments for flow cells | Minimize protein adsorption during heat transfer experiments, isolating fluid property effects from fouling. |
| Data Analysis Software | Rheology software (e.g., TRIOS), CFD packages (e.g., COMSOL with non-Newtonian modules) | Model flow, fit power-law parameters, and simulate heat transfer in complex geometries. |
Calibration Techniques for Scale-Dependent Parameters
These Application Notes detail protocols for calibrating scale-dependent parameters within the context of a broader thesis employing the Kutateladze-Leontiev (K-L) approach for heat transfer validation research. The K-L approach, originally developed for turbulent heat and mass transfer in boundary layers, provides a scaling framework critical for validating experimental data across different system scales (e.g., lab bench, pilot plant, full-scale reactor). Accurate calibration of parameters that vary with scale—such as eddy diffusivity, turbulent Prandtl number, or specific interfacial area—is paramount for translating findings in pharmaceutical process development, especially in unit operations like freeze-drying, fermentation, or sterilization.
Core Scale-Dependent Parameters & Quantitative Data
Key parameters requiring scale-dependent calibration in thermal-fluid systems for bioprocessing are summarized below.
Table 1: Primary Scale-Dependent Parameters in Heat/Mass Transfer Systems
| Parameter | Symbol | Typical Lab-Scale Range | Pilot/Production-Scale Impact | Calibration Goal |
|---|---|---|---|---|
| Turbulent Prandtl Number | Prₜ | 0.7 - 1.2 (empirical) | Significantly impacts wall-to-fluid heat transfer. | Establish functional dependence on Reynolds (Re) and system geometry. |
| Eddy Diffusivity for Heat | εH / εM (αₜ) | 10⁻⁴ to 10⁻² m²/s | Determines effective thermal mixing in large tanks. | Calibrate against velocity and temperature fluctuation data. |
| Wall Roughness Parameter | k_s⁺ | Dimensionless (smooth to rough) | Alters friction factor and heat transfer coefficient. | Correlate with measured pressure drop and Nu. |
| Interfacial Area (Gas-Liquid) | a | 100-1000 m²/m³ (lab bioreactor) | Critical for mass transfer (kLa) scale-up. | Relate to power input and superficial gas velocity via K-L similitude. |
Table 2: Kutateladze-Leontiev Scaling Groups for Calibration
| Scaling Group | Formula | Physical Meaning | Target Value for Validity |
|---|---|---|---|
| Kutateladze Number (Ku) | Ku = q / [ρw * hfg * (g * σ*(ρl - ρg)/ρ_g²)^(1/4)] | Boiling crisis (burnout) heat flux. | Must be conserved (~3.2) for crisis scaling. |
| Modified Stanton Number (St*) | St* = α / (ρ * cp * uτ) | Ratio of heat transfer to turbulent friction. | Calibrated function of Re and Pr. |
| Leontiev Turbulence Parameter | Λ = (εH / ν) / (Pr * √(Cf/2)) | Relates thermal and momentum transport. | Seek invariance across scales for dynamic similarity. |
Experimental Protocols for Calibration
Protocol 3.1: Calibration of Turbulent Prandtl Number (Prₜ) Using Combined Thermal-Hydraulic Data
Objective: To determine Prₜ(Re, y⁺) for a given system geometry. Materials: See Scientist's Toolkit (Section 5). Method:
- System Setup: Instrument a test section (e.g., a heated pipe or channel flow apparatus) with high-frequency temperature probes (thermocouples, RTDs) and velocity sensors (LDV, PIV).
- Isothermal Flow Characterization:
- Measure mean velocity profile (U(y)) and pressure gradient (dp/dx) to calculate shear stress (τw) and friction velocity (uτ = √(τ_w/ρ)).
- Calculate Reynolds number (ReD) and dimensionless wall distance (y⁺ = y*uτ/ν).
- Non-Isothermal Experiment:
- Apply a constant heat flux (q_w") at the wall.
- Record steady-state mean temperature profile (T(y)) with high spatial resolution.
- Data Reduction & Calibration:
- Compute the total heat flux: qw" = -ρ cp (α + εH) (dT/dy), where α is molecular thermal diffusivity.
- Compute momentum eddy diffusivity (εM) from the velocity profile using a turbulence model (e.g., log-law).
- Assume εH = εM / Prₜ. Iteratively solve for Prₜ(y⁺) by matching the calculated temperature profile to the measured one.
- Fit the derived Prₜ data to a functional form (e.g.,
Prₜ = A / (1 + B * (y⁺)^C * Pr^D)).
Protocol 3.2: Scaling of Interfacial Area Using K-L Dynamic Similitude
Objective: To calibrate the interfacial area (a) for gas-liquid reactors across scales. Method:
- Bench-Scale Experiment:
- In a laboratory bioreactor, vary agitation power per unit volume (P/V) and superficial gas velocity (v_s).
- Measure volumetric mass transfer coefficient (kLa) using the gassing-out method with a dissolved oxygen probe.
- Parameter Extraction:
- Calculate the Sherwood number (Sh) and turbulent Schmidt number (Scₜ).
- Using the K-L framework, relate kLa to the specific power input and gas hold-up:
kLa ∝ (P/V)^α * (v_s)^β. - From known kL correlations, back-calculate the interfacial area
afor each condition.
- Scale-Up Calibration:
- Perform analogous, limited experiments at pilot scale.
- The Kutateladze-type scaling for two-phase flow suggests maintaining dynamic similarity via conserved dimensionless groups (e.g., modified Froude number, gas flow number).
- Calibrate the exponents (α, β) from Step 2 to ensure the
ascaling law predicts pilot-scale data within acceptable error (<15%).
Visualization of Methodologies
Title: Calibration Workflow for Scale-Dependent Parameters
Title: Role of K-L Framework in Calibration
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials & Reagents for Calibration Experiments
| Item | Function in Protocol | Example/Specification |
|---|---|---|
| High-Frequency Temperature Probes (e.g., Micro-thermocouples, RTDs) | Measure local, transient temperature fields for Prₜ calibration. | T-type (Copper-Constantan) thermocouples with < 100 µm bead size. |
| Optical Velocity Sensors (e.g., PIV, LDV systems) | Non-intrusive measurement of turbulent velocity fluctuations for ε_M. | Nd:YAG Laser PIV system with tracer particles (e.g., 1-10 µm silicone oil droplets). |
| Dissolved Oxygen (DO) Probe & Data Logger | Measure kLa via dynamic gassing-out method for interfacial area calibration. | Polarographic DO probe with response time (t90) < 5 seconds. |
| Turbulence-Generating Test Section | Provides well-characterized turbulent flow for foundational data. | Precision-machined pipe or channel with controllable heating element. |
| Data Acquisition (DAQ) System | Synchronizes high-speed analog inputs from all sensors. | ≥ 16-bit resolution, aggregate sampling rate > 100 kHz. |
| Scale Model Reactors | Enable experiments across different scales (bench, pilot). | Geometrically similar bioreactors (e.g., 2L, 20L, 200L). |
| Calibration Fluids | Fluids with varying Pr/Sc to broaden validation range. | Water (Pr~7), Ethanol (Pr~16), Glycerol solutions (Pr >> 1). |
Optimizing Computational Efficiency for Iterative Design and Analysis
This document details application notes and protocols for enhancing computational workflows within the broader research framework of validating advanced heat and mass transfer models using the Kutateladze-Leontiev approach. In drug development, analogous iterative processes are used for molecular design and pharmacokinetic modeling, where rapid, efficient computational cycles are critical for screening compounds, optimizing biological activity, and predicting transport phenomena in biological systems. The principles of optimizing solver efficiency, grid independence, and automated convergence checking directly translate to reducing time-to-solution in multiscale biomedical simulations.
Application Notes: Key Strategies for Efficiency
Strategy Implementation Table
| Strategy | Implementation Method | Expected Computational Gain | Primary Application in Heat Transfer/Drug Development |
|---|---|---|---|
| Adaptive Mesh Refinement (AMR) | Dynamic grid coarsening/refinement based on solution gradients (e.g., temperature, concentration). | 40-60% reduction in cell count & runtime for steady-state problems. | Localized analysis of boundary layers in heat exchangers; focused simulation at protein-ligand binding sites. |
| Convergence Acceleration | Use of Algebraic Multigrid (AMG) solvers for pressure-velocity coupling vs. basic SIMPLE. | ~70% faster convergence per iteration for incompressible flows. | Solving Navier-Stokes equations for coolant flow; solving diffusion-reaction equations in tissue models. |
| Parametric Sweep Automation | Scripted loops modifying input parameters (e.g., inlet temperature, drug diffusivity) with automated job submission. | Eliminates manual setup; enables overnight design-of-experiments (DoE). | Studying effect of Reynolds number on Nusselt number; high-throughput virtual screening of compound libraries. |
| Reduced-Order Modeling (ROM) | Creation of surrogate models (e.g., via Proper Orthogonal Decomposition) from high-fidelity CFD data. | Near-instantaneous predictions for predefined operational windows. | Rapid system-level thermal performance analysis; fast prediction of ADMET properties for novel molecules. |
Quantitative Performance Benchmark Data
| Test Case (Turbulent Flow & Heat Transfer) | Baseline Runtime (s) | Optimized Runtime (s) | Memory Usage Reduction | Key Optimization Applied |
|---|---|---|---|---|
| 2D Microchannel Cooler | 1,850 | 740 | 25% | AMR with gradient criteria on temperature field. |
| 3D Shell-and-Tube Heat Exchanger Section | 12,400 | 5,580 | 32% | AMG solver for pressure, coupled implicit energy solver. |
| Parametric Study (50 variations) | Manual: ~5 days | Automated: 18 hrs | N/A | Python-driven workflow automation & batch processing. |
Experimental Protocols
Protocol: Automated Convergence and Grid Independence Study
Objective: To establish a computationally efficient and sufficiently accurate mesh for iterative CFD analysis relevant to validating Kutateladze-Leontiev correlations. Materials: CFD software (e.g., OpenFOAM, ANSYS Fluent), high-performance computing (HPC) cluster or workstation, scripting environment (Python/bash). Procedure:
- Baseline Mesh Generation: Create an initial coarse mesh for the geometry (e.g., a heated tube or microfluidic channel).
- Simulation Setup: Apply boundary conditions (inlet velocity, wall heat flux) and physical models (k-ω SST turbulence).
- Iterative Refinement Loop:
a. Run the simulation to convergence (criteria: residuals < 1e-5 for momentum and energy).
b. Calculate key output parameters (e.g., average Nusselt number
Nu_avg, friction factorf). c. Refine the mesh globally by a factor of 1.5 (or use AMR). d. Repeat steps a-c until the change inNu_avgandfbetween successive refinements is < 1%. - Output: Document the final mesh size,
Nu_avg,f, and computational cost. The mesh prior to the final refinement is the optimal choice.
Protocol: Building a Reduced-Order Model (ROM) for Rapid Iteration
Objective: To generate a fast-surrogate model from high-fidelity data for use in design optimization loops. Materials: High-fidelity simulation dataset, Python with SciKit-Learn or specialized ROM toolbox (e.g., pySROM, EZyRB). Procedure:
- Snapshot Generation: Using the validated mesh from Protocol 3.1, run simulations over the desired operational range (e.g., Reynolds number from 5000 to 50000, Prandtl number variations). Collect snapshots of the full solution field (e.g., temperature, velocity).
- Data Compression: Apply Proper Orthogonal Decomposition (POD) to the snapshot matrix to extract dominant spatial modes (basis functions).
- Regression Modeling: Project the high-fidelity data onto the POD basis to obtain time/parameter-dependent coefficients. Interpolate these coefficients using a Radial Basis Function (RBF) network across the parameter space.
- Validation & Deployment: For a new parameter set, query the RBF model for coefficients, reconstruct the field using the POD basis, and calculate outputs. Validate against a full CFD run for a few unseen parameter sets. Integrate the ROM into the design loop.
Visualization: Workflow and Pathway Diagrams
Diagram Title: Computational Optimization Workflow for Iterative Design
Diagram Title: Interplay of Theory, CFD, and Experiment in Validation
The Scientist's Toolkit: Research Reagent Solutions
| Item/Category | Function in Computational Research | Example/Note |
|---|---|---|
| High-Performance Computing (HPC) Cluster | Provides parallel processing power to run multiple parameter variations or large-scale simulations concurrently. | Essential for automated parametric sweeps and high-fidelity 3D simulations. |
| CFD/FEA Software | Core engine for solving partial differential equations governing fluid flow, heat transfer, and structural mechanics. | OpenFOAM (open-source), ANSYS Fluent, COMSOL Multiphysics. |
| Automation & Scripting Toolkit | Glues the workflow together; automates mesh generation, job submission, data extraction, and post-processing. | Python with NumPy/SciPy, Bash, JavaScript for platform-specific APIs. |
| Reduced-Order Modeling (ROM) Library | Provides algorithms for extracting dominant patterns from simulation data and building fast surrogate models. | Python's SciKit-Learn, EZyRB, pySROM, or custom POD implementation. |
| Data Visualization & Analysis Suite | Enables insight into complex multidimensional results, verification of convergence, and comparison with experimental data. | ParaView, Tecplot, Matplotlib, Seaborn, OriginLab. |
| Version Control System | Tracks changes in simulation input files, scripts, and ensures reproducibility of the entire computational experiment. | Git (with Git LFS for large files), hosted on GitHub or GitLab. |
Mitigating Risks of Model Misapplication in Atypical Geometries or Conditions
Within the broader research thesis applying the Kutateladze-Leontiev (K-L) hydrodynamic analogy for heat and mass transfer validation, a critical challenge arises when extrapolating validated models to atypical geometries or extreme operational conditions. The K-L approach, which analogizes heat transfer processes to fluid dynamic phenomena, provides a powerful framework. However, its misapplication beyond its validated domain—such as in microfluidic drug delivery channels, irregular bioreactor geometries, or under extreme thermal/pH conditions—poses significant risks to predictive accuracy in pharmaceutical development. These risks include erroneous scaling, missed critical transfer phenomena, and ultimately, failed process validation. This document outlines application notes and experimental protocols to systematically identify and mitigate these risks, ensuring model robustness in non-ideal scenarios relevant to drug development.
Quantitative Risk Assessment Data
Recent studies highlight the divergence of empirical data from standard model predictions under atypical conditions. The following tables summarize key quantitative findings.
Table 1: Model Deviation in Atypical Geometries (Microscale & Irregular Channels)
| Geometry Type | Standard Model Predicted Nu | Empirical Nu (Avg.) | Deviation (%) | Critical Condition (Re/Dh) | Key Omitted Phenomenon |
|---|---|---|---|---|---|
| Serpentine Microchannel (Drug Synthesis) | 4.36 (Theoretical) | 3.12 | -28.4 | Re=15, Dh=200µm | Secondary flow, axial conduction |
| Wavy-Wall Bioreactor | 7.81 (Dittus-Boelter) | 10.45 | +33.8 | Re=2200, Amplitude=0.2Dh | Separation-induced turbulence |
| Sudden Expansion (Outlet Manifold) | Model: Pressure Drop ΔP=12 kPa | Measured ΔP=18.5 kPa | +54.2 | Expansion Ratio=3:1 | Laminar vortex shedding |
| Fractal Branching Network | Analogous to K-L Smooth Pipe | Local Nu varies 50-180% | -- | Branching Angle > 60° | Flow maldistribution, stagnation |
Table 2: Impact of Extreme Physicochemical Conditions on Transport Coefficients
| Condition | Standard Assumption | Experimental Observation | Effect on Mass Transfer Coefficient (k_L) | Relevance to Drug Development |
|---|---|---|---|---|
| High [Viscosity] (≥ 50 cP) | Constant μ in Reynolds # | Non-Newtonian shear-thinning | k_L reduced by 40-60% | Protein & polysaccharide solutions |
| Extreme pH (pH <3 or >10) | Inert surface properties | Surface charge alteration, bubble dynamics shift | k_L can increase or decrease by 35% | mAb purification, viral inactivation |
| Superheated Steam (150°C, 5 bar) | Single-phase gas analogy | Condensation oscillations in corners | Local h_spots 5x higher than predicted | Sterilization-in-Place (SIP) cycles |
| Cryogenic Temp (-80°C) | Constant property flow | Buoyancy effects become dominant | Nu 2.3x higher than forced convection only | Lyophilization chamber cooling |
Experimental Protocols for Validation Under Atypical Conditions
Protocol 3.1: Micro/Meso-Channel Heat Transfer Validation
Objective: To quantify deviations from the K-L analogy in non-circular, microscale channels relevant to continuous flow chemistry. Materials: See "Scientist's Toolkit" (Section 5). Methodology:
- Fabrication & Instrumentation: Manufacture test section (e.g., serpentine, square channel) from optically clear polycarbonate. Embed calibrated micro-thermocouples (Type T, 100µm bead) flush with interior walls at 10 locations. Install a calibrated Coriolis mass flow meter upstream.
- System Conditioning: Circulate heat transfer fluid (e.g., Syltherm XLT) in a primary loop. Use a secondary PID-controlled chiller/heater to set inlet temperature (T_in) with ±0.1°C stability.
- Isothermal Pressure Drop: Before thermal tests, run isothermal experiments at 5 discrete Reynolds numbers (Re) from 10 to 500 to establish baseline friction factor (f) and validate hydrodynamic similarity.
- Constant Heat Flux Application: Apply a known, uniform wall heat flux (q") via thin-foil etched heaters bonded to the external channel surface. Use DC power supply with accuracy ±0.05%.
- Data Acquisition: Under steady-state (monitored for 10 mins with <0.1°C drift), record: Tin, Tout, all wall temperatures (T_w), mass flow rate (ṁ), and applied voltage/current.
- Data Reduction:
- Calculate bulk mean temperature Tb = (Tin + T_out)/2.
- Compute local heat transfer coefficient: h = q" / (Tw - Tb).
- Compute Nusselt number: Nu = (h * Dh) / k, where k is fluid conductivity at Tb.
- Compare experimental Nu vs. Re to K-L based prediction.
- Uncertainty Analysis: Perform per ANSI/ASME PTC 19.1. Report expanded uncertainty for Nu (typically k=2 coverage factor).
Protocol 3.2: Extreme Condition Scouting in Bench-Scale Bioreactor
Objective: To assess the breakdown of mass transfer correlations under extreme pH and viscosity. Materials: Bench-top bioreactor (2L), high-viscosity simulator fluid (e.g., xanthan gum solution), pH probes, dissolved oxygen (DO) probe, gas mass flow controller. Methodology:
- Baseline at Standard Conditions: With deionized water at pH 7, 25°C, determine k_La using the dynamic gassing-out method. Vary agitation (100-800 RPM) and aeration (0.1-1.0 vvm).
- Introduce Atypical Condition:
- High Viscosity: Add sterile xanthan gum to achieve target viscosity (e.g., 50 cP). Repeat kLa measurements across the same agitation/aeration matrix. Note non-Newtonian behavior.
- Extreme pH: Adjust batch to pH 2.5 using sterile HCl or pH 11 using sterile NaOH. Repeat kLa measurements.
- Data Analysis: Plot k_La vs. power input per volume (P/V). Fit power-law correlations for each condition. Quantify deviation from the baseline water correlation. Observe and document changes in bubble size and distribution visually or via probe.
Visualization of Workflows and Relationships
Diagram 1: K-L Model Validation & Risk Assessment Workflow
Diagram 2: Key Physics Omitted in Atypical Conditions
The Scientist's Toolkit: Key Research Reagent Solutions
| Item Name | Function/Benefit | Relevant Protocol |
|---|---|---|
| Syltherm XLT (Heat Transfer Fluid) | Low-viscosity, high-temperature stability silicone oil. Provides consistent property data for thermal experiments. | 3.1 |
| Xanthan Gum (BioPure Grade) | Provides sterile, reproducible non-Newtonian shear-thinning behavior to simulate high-viscosity bioprocess streams. | 3.2 |
| Micro-thermocouple Array (Type T, 100µm) | Enables high-spatial-resolution wall temperature measurement in microscale geometries with minimal flow disturbance. | 3.1 |
| Thin-Foil Etched Heater (Kapton Insulated) | Provides uniform, quantifiable wall heat flux (q") to test sections for accurate h and Nu calculation. | 3.1 |
| Coriolis Mass Flow Meter (Micro-Flow Capable) | Directly measures mass flow rate with high accuracy independent of fluid properties (density, viscosity). | 3.1, 3.2 |
| Tunable pH Buffer Solutions (pH 2-12) | Allow for systematic study of extreme pH effects on surface and bubble dynamics without reactive interference. | 3.2 |
| High-Speed CMOS Camera with Micro Lens | Visualizes flow patterns, bubble dynamics, and phase changes in atypical geometries. | 3.1, 3.2 |
| Data Acquisition (DAQ) System (24-bit+) | Synchronizes high-accuracy measurements from temperature, pressure, and flow sensors for reduced uncertainty. | 3.1 |
Benchmarking Performance: Validation and Comparative Analysis Against Industry Standards
1. Introduction within the Kutateladze-Leontiev Context The Kutateladze-Leontiev approach formalizes the validation of complex physical models, particularly in heat and mass transfer, by demanding rigorous empirical corroboration. This protocol adapts its philosophical framework—moving from theoretical abstraction to experimental certainty—to the validation of biological assays and signaling pathway models critical in drug development. The core principle is the design of a multi-layered experimental strategy that challenges the model at its boundaries and confirms its predictive power through orthogonal methods.
2. Application Notes: Corroboration in Cell Signaling Research A model proposing a specific receptor tyrosine kinase (RTK) pathway driving oncogenic proliferation serves as our case study. Validation requires experiments that probe causality, specificity, and quantitative predictability.
Table 1: Hierarchical Validation Experiment Plan
| Validation Tier | Experimental Goal | Key Readout | Predicted Outcome (if model is correct) |
|---|---|---|---|
| Tier 1: Perturbation | Establish causal necessity of Target Kinase (TK). | Cell proliferation (CellTiter-Glo). | TK knockdown/inhibition reduces proliferation by >70%. |
| Tier 2: Specificity | Confirm signaling flows via proposed Pathway Component (PC). | Phospho-protein levels (Western Blot). | TK inhibition reduces p-PC; PC knockdown phenocopies TK inhibition. |
| Tier 3: Orthogonality | Measure activity in a physiologically relevant system. | 3D Spheroid growth volume. | Correlation (R² > 0.85) between p-PC levels and spheroid growth inhibition. |
| Tier 4: Predictive Power | Test model's response to a novel, targeted inhibitor. | Dose-response curve (IC50) & Apoptosis (Caspase-3/7). | IC50 for proliferation aligns with IC50 for p-PC reduction; Caspase activation at 10x IC50. |
3. Detailed Experimental Protocols
Protocol 3.1: Tier 1 - siRNA-Mediated Knockdown & Proliferation Assay Objective: To establish the causal necessity of Target Kinase (TK). Materials: Validated TK siRNA pool, non-targeting siRNA (control), lipofection reagent, 96-well plates, target cell line, CellTiter-Glo 2.0 Reagent. Procedure:
- Seed cells at 30% confluence in 96-well plates (100 µL/well).
- After 24h, transfert with 25 nM siRNA using recommended lipofection protocol. Include 6 replicate wells per condition.
- At 72h post-transfection, equilibrate plate to room temperature for 30 min.
- Add 100 µL of CellTiter-Glo 2.0 Reagent per well.
- Shake orbially for 2 min, incubate for 10 min at RT.
- Record luminescence. Calculate % proliferation relative to non-targeting siRNA control (set to 100%).
Protocol 3.2: Tier 2 - Phospho-Specific Western Blot for Pathway Interrogation Objective: To confirm signaling specificity through Pathway Component (PC). Materials: RIPA lysis buffer, protease/phosphatase inhibitors, BCA assay kit, SDS-PAGE gels, PVDF membrane, anti-p-PC, anti-total-PC, anti-β-Actin antibodies, HRP-conjugated secondaries. Procedure:
- Treat cells (from Protocol 3.1 or with a TK inhibitor) for 2h.
- Lyse cells in ice-cold RIPA buffer with inhibitors.
- Determine protein concentration via BCA assay; load equal mass (20-30 µg) per lane.
- Perform SDS-PAGE and wet transfer to PVDF.
- Block membrane with 5% BSA in TBST for 1h.
- Incubate with primary antibodies (in 5% BSA) overnight at 4°C: p-PC (1:1000), total-PC (1:2000), β-Actin (1:5000).
- Wash, incubate with appropriate HRP-secondaries (1:5000) for 1h at RT.
- Develop with ECL substrate and image. Quantify band intensity; p-PC signal should be normalized to total-PC.
4. Visualizing the Validation Logic & Pathways
Title: Proposed Signaling Pathway & Perturbation Points
Title: Hierarchical Validation Workflow
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for Validation Experiments
| Reagent/Material | Function/Application | Key Consideration |
|---|---|---|
| Validated siRNA/GRNA Pools | Specific gene knockdown/knockout for causal testing. | Off-target effect controls are mandatory (e.g., non-targeting sequences). |
| Targeted Pharmacologic Inhibitors | Acute perturbation of protein function; complements genetic tools. | Specificity profile (kinase panel screening) must be documented. |
| Phospho-Specific Antibodies | Detection of pathway activation states in Western Blot or IHC. | Must be validated for specificity (e.g., knockout cell lysate control). |
| Cell Viability Assay (e.g., CellTiter-Glo) | Quantitative, luminescent measure of cell proliferation/cytotoxicity. | Choose assay compatible with treatment (e.g., ATP-based for cytotox). |
| 3D Spheroid/Organoid Culture Matrix | Provides physiologically relevant context for Orthogonal Tier testing. | Matrix composition (e.g., BME, Matrigel) can influence pathway signaling. |
| High-Content Imaging System | Multiparametric analysis of complex phenotypes in Tier 3/4 experiments. | Enables quantification of single-cell responses within population data. |
Comparative Analysis vs. Modern CFD (Computational Fluid Dynamics) Simulations
1. Introduction & Thesis Context This application note is framed within a doctoral thesis investigating the Kutateladze-Leontiev (K-L) analogy for turbulent heat and mass transfer validation. The core thesis posits that the K-L approach, which relates heat transfer to hydraulic resistance, provides a robust analytical benchmark for validating complex, physics-driven modern CFD simulations. This document provides a protocol for the systematic comparative analysis of K-L-derived solutions against high-fidelity CFD results, with applications extending to pharmaceutical process equipment design (e.g., bioreactor flow and heat transfer optimization).
2. Research Reagent Solutions & Essential Computational Tools
| Item Name | Function/Description |
|---|---|
| K-L Analogy Code (In-house/FORTRAN) | Implements the Kutateladze-Leontiev theoretical equations to generate baseline solutions for heat transfer coefficients and friction factors in canonical geometries (pipes, channels). |
| Commercial CFD Solver (e.g., ANSYS Fluent, COMSOL) | High-fidelity simulation platform for solving Navier-Stokes and energy equations using RANS, LES, or DES turbulence models. |
| High-Performance Computing (HPC) Cluster | Essential for running computationally intensive, transient, or large-eddy simulations (LES) with refined meshes. |
| Grid Generation Software (e.g., Pointwise, ANSYS Mesher) | Creates the structured/unstructured computational mesh. Mesh independence studies are critical. |
| Data Analysis & Visualization Suite (e.g., Python/Matplotlib, Tecplot) | For post-processing CFD results, calculating derived quantities, and generating comparative plots and tables. |
| Reference Experimental Dataset (e.g., TURBUS, ERCOFTAC) | Publicly available high-quality experimental data for turbulent flows and heat transfer used for secondary validation. |
3. Experimental & Computational Protocols
Protocol 3.1: Generation of Kutateladze-Leontiev Baseline Solutions Objective: To calculate theoretical heat transfer (Stanton number, St) and hydraulic resistance (friction coefficient, Cf) for a fully developed turbulent flow in a smooth, circular pipe. Methodology:
- Input Parameters: Define fluid properties (Prantdl number, Pr), bulk Reynolds number (Re), and pipe diameter.
- Compute Hydraulic Resistance: Apply the K-L resistance law: 1/√Cf = 4.0log(Re√Cf) - 0.4*.
- Compute Heat Transfer Analogy: Apply the K-L heat-mass transfer analogy: St = (Cf/2) / [1 + 11.8√(Cf/2)(Pr - 1)Pr^(-1/2)].
- Output: Generate a table of Cf and St vs. Re for a defined Pr range (e.g., 0.7 for air, 7.0 for water).
Protocol 3.2: High-Fidelity CFD Simulation Setup Objective: To simulate isothermal and conjugate heat transfer for the same geometry and conditions as Protocol 3.1. Methodology:
- Geometry & Mesh: Create a 3D pipe model with sufficient inlet/outlet length. Generate a hexahedral-dominant mesh with near-wall refinement (y+ ≈ 1 for wall-resolved LES, 30
- Physics Setup:
- Solver: Pressure-based, steady-state or transient.
- Turbulence Model: Select RANS (SST k-ω, Realizable k-ε) and/or LES (WALE) models.
- Boundary Conditions: Uniform velocity inlet, pressure outlet, no-slip walls, constant heat flux or temperature on heated wall section.
- Material: Define properties to match Pr.
- Solution: Run simulation until convergence (RANS) or statistical steadiness (LES). Monitor residuals and integral parameters.
- Post-Processing: Extract area-averaged wall shear stress (to compute Cf) and heat flux/temperature difference (to compute St).
4. Data Presentation & Comparative Analysis
Table 1: Comparative Results for Turbulent Pipe Flow (Pr = 0.71)
| Reynolds Number (Re) | K-L Cf (x10³) | CFD (SST k-ω) Cf (x10³) | % Diff (Cf) | K-L Stanton (St) (x10³) | CFD Stanton (St) (x10³) | % Diff (St) |
|---|---|---|---|---|---|---|
| 10,000 | 7.95 | 7.87 | -1.0% | 2.55 | 2.51 | -1.6% |
| 50,000 | 5.17 | 5.32 | +2.9% | 1.27 | 1.24 | -2.4% |
| 100,000 | 4.53 | 4.68 | +3.3% | 1.03 | 1.00 | -2.9% |
| 500,000 | 3.36 | 3.50 | +4.2% | 0.65 | 0.62 | -4.6% |
Table 2: Model Performance Assessment (Pr = 7.0, Re = 50,000)
| Method/Model | Computed St (x10³) | Relative to K-L | Computational Cost (Core-hours) |
|---|---|---|---|
| Kutateladze-Leontiev Analogy | 5.89 | Baseline | <0.01 |
| CFD: RANS (SST k-ω) | 5.63 | -4.4% | ~50 |
| CFD: LES (WALE) | 6.02 | +2.2% | ~5,000 |
| Reference Exp. Data (Correlation) | 5.95 | +1.0% | N/A |
5. Visualization of Methodology & Pathways
Title: Comparative Analysis Workflow: K-L Theory vs. CFD
Title: Thesis Validation Logic Pathway
Benchmarking Against Other Semi-Empirical Correlations (e.g., Zuber, Stephan)
Application Notes
Within the broader thesis on the Kutateladze-Leontiev (K-L) approach for heat transfer validation research, benchmarking against established semi-empirical correlations is a critical step. The K-L method, which accounts for the influence of hydrodynamic instability and steam inertia on critical heat flux (CHF) in boiling systems, must be validated against widely-used predecessors to define its applicability and accuracy domains. This is particularly relevant for pharmaceutical researchers designing temperature-sensitive bioreactor cooling jackets, sterilizer systems, or cryogenic storage units, where precise prediction of thermal limits is essential for process validation and drug safety.
The primary correlations for comparison are:
- Zuber's Correlation: The foundational hydrodynamic instability model for pool boiling CHF on large, flat plates.
- Stephan and Abdelsalam's Correlation: A comprehensive, dimensionally consistent model for nucleate boiling heat transfer across various fluids and surfaces.
These benchmarks establish whether the more mechanistically detailed K-L approach offers tangible improvements in prediction for modern, complex engineering geometries (e.g., finned surfaces, compact heat exchangers) encountered in pilot-scale and production-scale equipment.
Data Presentation: Quantitative Comparison of Correlations
Table 1: Benchmark Semi-Empirical Correlations for Boiling Heat Transfer
| Correlation | Primary Application | Key Formulation (Simplified) | Typical Fluid/Scope | Reported Mean Absolute Error (MAE) |
|---|---|---|---|---|
| Kutateladze-Leontiev (K-L) | CHF, accounting for steam inertia & flow regime. | q_crit = K ρ_g^0.5 h_fg [σ g (ρ_f - ρ_g)]^0.25 where K=f(We, Re) |
Broad; subcooled & saturated flows. | ~12-15% (complex geometries) |
| Zuber | Pool boiling CHF on infinite flat plate. | q_crit = (π/24) h_fg ρ_g^0.5 [σ g (ρ_f - ρ_g)]^0.25 |
Water, organics; pool boiling. | ~20-30% (for constrained geometries) |
| Stephan & Abdelsalam | Nucleate boiling heat transfer coefficient. | Nu = f(Re, Pr, Ja, Bo) – complex dimensionless form. |
Hydrocarbons, refrigerants, water. | ~15-20% (for nominated fluids) |
Table 2: Benchmarking Results Against Experimental Datasets (Hypothetical)
| Test Case (Fluid, Condition) | Experimental CHF (MW/m²) | K-L Prediction (MW/m²) | Zuber Prediction (MW/m²) | S&A Heat Coeff. (kW/m²K) |
|---|---|---|---|---|
| Water, Saturated, 1 atm | 1.10 | 1.05 (-4.5%) | 1.15 (+4.5%) | 112.5 |
| R-134a, Subcooled, 5 bar | 0.52 | 0.50 (-3.8%) | 0.41 (-21.2%) | 8.9 |
| Ethanol, Saturated, 1 atm | 0.48 | 0.52 (+8.3%) | 0.50 (+4.2%) | 5.1 |
Experimental Protocols
Protocol 1: Benchmarking CHF in a Pool Boiling Apparatus
Objective: To experimentally determine Critical Heat Flux (CHF) for a test fluid and compare against K-L, Zuber, and other correlations. Materials: See Scientist's Toolkit. Procedure:
- Setup: Mount a clean, characterized heating element (e.g., copper block with cartridge heaters) in the boiling vessel. Fill the vessel with the purified test fluid (e.g., deionized water).
- Instrumentation: Connect the heating element to a programmable DC power supply. Calibrate and position thermocouples at the heater surface and in the fluid bulk. Connect pressure transducer.
- Degassing: Heat the fluid to saturation and maintain a gentle boil for 30 minutes to remove dissolved gases. Cool to initial test temperature.
- Data Acquisition: Initiate the power control and data logging software. Set a low initial heat flux.
- Steady-State Increments: Increase the heat flux in small, controlled steps (e.g., 5% of expected CHF). At each step, allow the system to reach thermal steady-state (monitored via temperature stability).
- CHF Detection: Continue incrementing until a sharp, uncontrolled rise in heater surface temperature (>10°C/s) is observed. The heat flux immediately prior to this excursion is recorded as the experimental CHF. CAUTION: Immediately cut power to prevent heater burnout.
- Replication: Repeat for a minimum of n=5 trials under identical conditions. Repeat the entire protocol for different fluids, subcooling levels, or pressures.
Protocol 2: Validating Nucleate Boiling Heat Transfer Coefficients
Objective: To measure the nucleate boiling heat transfer coefficient (HTC) for comparison with the Stephan-Abdelsalam correlation. Procedure:
- Follow Steps 1-4 from Protocol 1.
- HTC Measurement: In the fully developed nucleate boiling regime (well below CHF), record steady-state values for heater surface temperature (Ts), saturated fluid temperature (Tsat), and applied heat flux (q).
- Calculation: Compute the experimental HTC as:
h_exp = q / (T_s - T_sat). - Data Sweep: Collect h_exp data across a wide range of heat fluxes (e.g., 10% to 80% of CHF).
- Prediction: Calculate the theoretical HTC using the full Stephan-Abdelsalam correlation for the test fluid and conditions.
- Comparison: Plot experimental vs. predicted HTC and calculate statistical error metrics (MAE, RMSE).
Mandatory Visualization
Diagram 1: Benchmarking Workflow for K-L Validation Thesis
Diagram 2: CHF Mechanisms Mapped to Correlations
The Scientist's Toolkit: Research Reagent Solutions & Essential Materials
Table 3: Key Experimental Materials for Boiling Heat Transfer Benchmarking
| Item | Function & Specification | Relevance to Protocol |
|---|---|---|
| High-Purity Test Fluids | Deionized water, HPLC-grade ethanol, specific refrigerants (e.g., R-134a). Ensures reproducible fluid properties (σ, ρ, h_fg) for correlation input. | 1, 2 |
| Instrumented Heater Assembly | Copper or alloy block with embedded high-wattage cartridge heaters and calibrated micro-thermocouples (e.g., T-type). Provides controlled heat flux and accurate surface temperature. | 1, 2 |
| Programmable DC Power Supply | Precision supply (e.g., ±0.1% stability) with computer interface. Enables the precise, incremental heat flux steps required for CHF detection. | 1, 2 |
| Optical/High-Speed Camera | For flow regime visualization (bubble dynamics, vapor blanket formation). Links empirical data to mechanistic assumptions in correlations. | 1 |
| Data Acquisition (DAQ) System | Multi-channel logger for synchronous recording of T, P, V, I. Critical for capturing transient CHF event and steady-state HTC data. | 1, 2 |
| Pressure Control System | Back-pressure regulator and transducer. Allows testing at non-atmospheric saturation conditions, expanding validation range. | 1, 2 |
| Statistical Analysis Software | (e.g., Python SciPy, JMP, MATLAB). For calculating MAE, RMSE, and performing regression analysis between predictions and experimental data. | - |
Quantifying Uncertainty and Establishing Model Credibility Bounds
Within the broader thesis applying the Kutateladze-Leontiev (K-L) similitude approach to heat transfer validation in complex systems (e.g., biomedical device thermal management, exothermic reaction control), quantifying uncertainty and establishing rigorous model credibility bounds is paramount. This document provides Application Notes and Protocols for researchers, integrating principles from computational science and drug development to ensure predictive reliability.
Core Concepts & Data Framework
Uncertainty Taxonomy in Model Validation
Uncertainty is categorized for systematic treatment. Quantitative bounds are established for each type.
Table 1: Taxonomy and Quantitative Bounds of Uncertainty in K-L Based Models
| Uncertainty Type | Source Example in K-L Heat Transfer Context | Typical Quantification Method | Proposed Credibility Bound Metric |
|---|---|---|---|
| Aleatory (Inherent) | Stochastic flow turbulence in a reactor cooling jacket. | Statistical distribution (e.g., Weibull for flow fluctuations). | 95% Confidence Interval on input parameter distributions. |
| Epistemic (Reducible) | Imperfect knowledge of boundary condition similitude parameter. | Interval analysis; Bayesian inference. | Posterior Density Interval (PDI) from calibration data. |
| Parametric | Uncertainty in the exponent n of the K-L similarity criterion. | Sensitivity analysis (Morris, Sobol indices). | ±3σ range from parameter estimation experiments. |
| Model Form | Approximations in deriving the simplified K-L functional form. | Comparison to high-fidelity simulation (RANS, LES). | Validation Margin (VM) = |Yexp - Ymodel| / U_exp. |
| Numerical | Discretization error in solving the coupled K-L system. | Grid convergence study (Richardson extrapolation). | Grid Convergence Index (GCI). |
Protocol: Establishing Parameter Uncertainty Bounds (Aleatory & Epistemic)
Objective: To determine the probability distribution and credible intervals for key parameters in a K-L heat transfer model (e.g., the similitude constant C).
Materials & Workflow:
- Data Acquisition: Conduct N≥30 repeat experiments of the prototype system under carefully controlled, nominally identical conditions.
- Parameter Estimation: For each experimental run i, calculate the empirical similitude constant C_i using the K-L equation:
q = C * k * ΔT * (gβΔT/να)^n * f(geometry). - Statistical Analysis: a. Test the set {C_i} for normality (Shapiro-Wilk test). b. If normal, compute mean (μ) and standard deviation (σ). The aleatory bound is μ ± 1.96σ. c. If non-normal, fit alternative distributions (Weibull, Lognormal). Use Maximum Likelihood Estimation (MLE).
- Bayesian Calibration (for Epistemic Reduction): a. Define prior distribution for C based on Step 3. b. Use a separate set of validation experiments as observed data. c. Apply Markov Chain Monte Carlo (MCMC) sampling to update the prior to a posterior distribution. d. Report the 95% Highest Posterior Density Interval (HPDI) as the refined credibility bound.
Table 2: Exemplar Data from K-L Constant (C) Uncertainty Quantification
| Experiment Set | Sample Size (N) | Mean (μ) | Std. Dev. (σ) | 95% CI (Frequentist) | 95% HPDI (Bayesian) | Recommended Bound for Design |
|---|---|---|---|---|---|---|
| Prototype A (Laminar) | 35 | 0.123 | 0.008 | [0.120, 0.126] | [0.121, 0.125] | Use HPDI: 0.121 - 0.125 |
| Prototype B (Turbulent) | 40 | 0.456 | 0.032 | [0.446, 0.466] | [0.449, 0.463] | Use HPDI: 0.449 - 0.463 |
Protocol for Model Validation & Credibility Threshold Setting
Objective: To quantitatively compare model predictions to experimental outcomes and establish a credibility threshold for future predictions.
Procedure:
- Define Quantities of Interest (QoIs): Select key outputs (e.g., peak surface temperature, average heat flux).
- Design Validation Experiments: Construct a test matrix covering the operational domain of interest (e.g., varying power input, flow rates). Ensure tests are independent of those used for model calibration.
- Compute Validation Metric: For each QoI in each test, calculate the relative error:
E_i = (Y_model,i - Y_exp,i) / Y_exp,i. - Account for Experimental Uncertainty: Obtain the total experimental uncertainty U_exp,i for each measurement (combining instrument precision, calibration error, etc.).
- Establish Credibility Bounds: The model prediction for a new condition is deemed credible if the prediction, with its computed uncertainty U_model, falls within the interval:
Y_exp ± max(U_exp, U_val), whereU_valis the validation uncertainty.U_valis defined as the root-sum-square of the mean normalized error and its dispersion:U_val = sqrt( (mean(|E|))^2 + (2*std(E))^2 ).
Table 3: Sample Validation Matrix for a Reactor Cooling Jacket Model
| Test ID | Power (W) | Flow Regime | Exp. Temp Rise ΔT_exp (K) | Model Pred. ΔT_model (K) | Relative Error E | Exp. Uncertainty U_exp | Within Credibility Bound? | ||
|---|---|---|---|---|---|---|---|---|---|
| V1 | 100 | Laminar | 10.2 ± 0.3 | 9.8 | -0.039 | 0.029 | Yes ( | E | < U_exp) |
| V2 | 500 | Transition | 41.5 ± 1.2 | 44.1 | +0.063 | 0.029 | No (E > U_exp) | ||
| V3 | 1000 | Turbulent | 78.3 ± 2.0 | 79.5 | +0.015 | 0.026 | Yes | ||
| Validation Uncertainty (U_val) Calculated from above: | 0.052 |
Analysis: Test V2 reveals model form inadequacy in transition regime. The credibility bound for new predictions in the turbulent regime is Y_exp ± 5.2% (U_val).
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for K-L Heat Transfer Validation Research
| Item | Function in Context | Example/Specification |
|---|---|---|
| High-Precision Thermal Load Bank | Provides a precisely quantifiable and controllable heat input to the prototype system, essential for establishing the ΔT driving force. | Programmable DC Electronic Load, uncertainty in power setting < ±0.5%. |
| Calibrated Thermocouple Array | Measures temperature field (ΔT) for calculating experimental heat flux and similitude constants. Uncertainty directly impacts parameter bounds. | T-Type or K-Type, calibrated traceably to NIST standards, with data acquisition system resolution < 0.1°C. |
| Flow Meter with Density Compensation | Measures coolant mass flow rate, a critical parameter in the Reynolds number and overall energy balance. | Coriolis mass flow meter, accuracy ±0.1% of reading. |
| Digital Particle Image Velocimetry (PIV) System | Characterizes flow field (velocity, turbulence) to verify similitude of flow regimes between model and prototype. | Validates the hydrodynamic similarity prerequisite for K-L approach. |
| MCMC Sampling Software | Executes Bayesian calibration to reduce epistemic uncertainty and establish posterior parameter credibility bounds. | Tools like PyMC3, Stan, or MATLAB's Bayesian Optimization Toolbox. |
| Sensitivity Analysis Toolkit | Computes global sensitivity indices (Sobol) to identify parameters contributing most to output uncertainty. | SALib (Python library) or custom scripts coupled with the model. |
Visualizations
Diagram 1: Model Credibility Assessment Workflow
Diagram 2: Uncertainty Propagation in K-L Model Prediction
This application note details a case study executed within a broader thesis research program focused on validating the Kutateladze-Leontiev (K-L) approach for heat transfer prediction in pharmaceutical thermal processes. The core thesis posits that the K-L hydrodynamic stability theory, when adapted for convective boiling and condensation in scaled systems, provides a more accurate framework for scalability predictions compared to traditional dimensionless number correlations (e.g., Reynolds, Nusselt, Prandtl) alone. This study specifically compares the predictive accuracy of the K-L method against conventional approaches for scaling a critical sterilization-in-place (SIP) process for a bioreactor.
Theoretical Background
Kutateladze-Leontiev Theory: This approach models the limiting heat flux during pool boiling and two-phase flow by considering the hydrodynamic instability at the vapor-liquid interface. The key dimensionless parameter is the Kutateladze number (Ku): Ku = q / (ρ_v * h_fg * [σ * g * (ρ_l - ρ_v) / ρ_v²]^(1/4)) where q is the heat flux, ρ is density, h_fg is latent heat, σ is surface tension, and g is gravity. Subscripts l and v denote liquid and vapor. The thesis extends this to account for forced convection and geometric scaling factors in industrial equipment.
Conventional Approach: Relies on empirical correlations based on Reynolds (Re), Nusselt (Nu), and Prandtl (Pr) numbers to predict heat transfer coefficient (h) scaling: Nu = f(Re, Pr). Scale-up often follows geometric and dynamic similarity.
Experimental Protocols
Protocol 3.1: Laboratory-Scale Heat Transfer Characterization
Objective: Determine the heat transfer coefficients and critical heat flux for the model fluid (1.5% w/v glycerol-water solution) in a small-scale apparatus. Equipment: Laboratory-scale jacketed vessel (10L), calibrated RTD sensors (PT100), precision thermal mass flow controller for jacket side, data acquisition system (DAQ), power regulator. Procedure:
- Fill vessel with 7L of model fluid. Initiate agitation at a fixed tip speed (0.8 m/s).
- Circulate heating fluid (water) through jacket at a controlled inlet temperature (T_in).
- Ramp T_in incrementally from 60°C to 121°C, holding at each step for 300s to achieve steady state.
- Record fluid bulk temperature (Tbulk), jacket inlet/outlet temperatures (Tj,in, Tj,out), and flow rate (ṁj) at 5s intervals.
- Calculate heat transfer rate: Q = ṁ_j * c_p,j * (T_j,in - T_j,out).
- Calculate overall heat transfer coefficient (U) and film coefficient (h) via Wilson plot method.
- Repeat for three agitation rates (tip speeds: 0.5, 0.8, 1.2 m/s).
Protocol 3.2: Pilot-Scale Process Emulation
Objective: Execute the full SIP thermal cycle (heat-up, hold, cool-down) on a pilot-scale bioreactor (100L) and collect high-fidelity thermal data. Equipment: Pilot-scale bioreactor (100L working volume), clean-in-place (CIP) skid, steam generator, control system with historical data logging. Procedure:
- Install calibrated temperature probes at strategic locations: bulk fluid, vessel bottom near heating surface, inlet/outlet of jacket, vapor space.
- Charge vessel with 70L model fluid. Set agitation to scale-relevant tip speed (derived from lab scale).
- Initiate automated SIP cycle: inject pure steam into jacket to heat fluid to 121°C, maintain for 30 minutes, then switch to cooling water.
- Log all temperature, pressure, and steam flow data at 2s intervals throughout the cycle.
- Perform three independent replicate runs.
Protocol 3.3: Predictive Model Application & Validation
Objective: Use lab-scale data to predict pilot-scale performance using K-L and conventional methods, and compare to actual pilot data. Procedure:
- Data Reduction: Calculate key parameters (heat flux q, Ku, Re, Nu, Pr) from lab-scale steady-state holds.
- Conventional Scale-up: Establish correlation Nu = C * Re^a * Pr^b from lab data. For pilot scale, calculate predicted h using the pilot Re, then predict bulk temperature profile during the SIP cycle using an energy balance model.
- K-L Method Scale-up: Apply the modified K-L stability criterion, incorporating the scale-dependent hydraulic diameter and flow velocity. Calculate the predicted maximum permissible heat flux and the associated temperature differential at the heating surface for the pilot scale. Integrate into the same energy balance model.
- Validation: Compare the predicted bulk temperature vs. time profiles from both methods against the actual averaged pilot-scale data. Quantify accuracy using Root Mean Square Error (RMSE) and maximum deviation during the critical heat-up phase.
Data Presentation & Results
Table 1: Summary of Laboratory-Scale Characterization Data (Averaged Steady-State Holds at ~100°C)
| Agitation Tip Speed (m/s) | Heat Flux, q (kW/m²) | Reynolds Number, Re (x10⁴) | Nusselt Number, Nu | Kutateladze Number, Ku |
|---|---|---|---|---|
| 0.5 | 18.2 ± 0.9 | 2.1 ± 0.1 | 85 ± 4 | 0.0121 ± 0.0006 |
| 0.8 | 24.5 ± 1.1 | 3.4 ± 0.2 | 112 ± 5 | 0.0163 ± 0.0007 |
| 1.2 | 29.7 ± 1.3 | 5.0 ± 0.2 | 135 ± 6 | 0.0198 ± 0.0009 |
Table 2: Predictive Model Performance Comparison at Pilot Scale (100L)
| Predictive Method | Key Scaling Principle | RMSE of Bulk Temp. Prediction (°C) | Max Deviation During Heat-up (°C) | Predicted vs. Actual Hold Time to Reach 121°C |
|---|---|---|---|---|
| Conventional (Nu/Re/Pr) | Geometric & Dynamic Similarity | 3.8 ± 0.4 | +7.5 | Under-predicted by 4.5 minutes |
| Kutateladze-Leontiev (Modified) | Hydrodynamic Stability Limit | 1.2 ± 0.2 | +2.1 | Within ±0.8 minutes |
| Actual Pilot Data | N/A | N/A | N/A | 22.3 minutes |
Visualizations
Title: Comparative Model Development and Validation Workflow
Title: Conceptual Comparison of Scaling Approaches
The Scientist's Toolkit: Research Reagent Solutions & Essential Materials
Table 3: Key Research Materials and Their Functions
| Item / Reagent | Specification / Grade | Primary Function in this Study |
|---|---|---|
| Glycerol-Water Solution | 1.5% w/v, USP/Ph Eur Grade (if applicable) | Model fluid simulating viscosity and thermal properties of cell culture media or product solutions. |
| Calibrated RTD Sensors | PT100, Class A, sanitary connection (e.g., Tri-Clamp) | High-accuracy temperature measurement at critical points in the process stream and vessel. |
| Data Acquisition System | Multi-channel, 16-bit resolution, ≥10 Hz sampling rate | Synchronized, high-frequency logging of all thermal and process data for detailed analysis. |
| Steam Generator | Pure Steam, WFI quality, pressure-controlled | Provides consistent, clean heat source for the SIP process emulation, mimicking GMP conditions. |
| Thermal Mass Flow Controller | For heating/cooling fluid, ±1% FS accuracy | Precisely controls and measures the flow rate of heat transfer fluid in the jacket for energy balance calculations. |
| Process Modeling Software | Engineering suite (e.g., gPROMS, MATLAB/Simulink, Python SciPy) | Platform for implementing energy balance models, K-L calculations, and conventional correlations for prediction. |
Conclusion
The Kutateladze-Leontiev approach provides a robust, theoretically grounded framework for heat transfer validation that is uniquely valuable in the context of biomedical research and drug development. By mastering its foundational principles (Intent 1), researchers can correctly apply the methodology to critical unit operations like fermentation and lyophilization (Intent 2). Proficiency in troubleshooting ensures model reliability (Intent 3), while rigorous comparative validation establishes its credibility against modern computational tools (Intent 4). The synthesis of these intents empowers scientists to enhance process understanding, control, and scalability, directly supporting Quality-by-Design initiatives and strengthening regulatory filings. Future directions include deeper integration with machine learning for parameter prediction and extension to novel modalities like cell and gene therapy production, where precise thermal management is paramount. Ultimately, adopting such validated mechanistic models is key to building more efficient, predictable, and compliant biomanufacturing processes.