Decoding Battery Health: A Comprehensive Guide to EIS Equivalent Circuit Modeling for Researchers
This article provides a detailed, step-by-step exploration of Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling (ECM) for battery analysis.
Decoding Battery Health: A Comprehensive Guide to EIS Equivalent Circuit Modeling for Researchers
Abstract
This article provides a detailed, step-by-step exploration of Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling (ECM) for battery analysis. Targeted at researchers and scientists, it covers foundational principles, practical application methodology, strategies for troubleshooting and optimizing model fits, and rigorous validation techniques. By translating complex impedance spectra into intuitive circuit elements, this guide empowers professionals to accurately diagnose battery state-of-health (SOH), predict performance, and accelerate R&D cycles.
Understanding the Language of Batteries: Core Principles of EIS and Equivalent Circuits
Electrochemical Impedance Spectroscopy (EIS) is a non-destructive, frequency-domain technique pivotal for analyzing the complex electrical properties of electrochemical systems, such as batteries. Within the context of a broader thesis on EIS equivalent circuit modeling for battery research, understanding Nyquist and Bode plots is fundamental. These visual representations are not mere data outputs; they are critical diagnostic tools that enable researchers to deconvolute and quantify key battery phenomena—including charge transfer kinetics, mass transport limitations, and solid-electrolyte interphase (SEI) layer formation—by fitting data to physically meaningful equivalent electrical circuits.
Core Principles: Nyquist vs. Bode Plots
EIS measures a system's impedance (Z) as a function of frequency. The data is complex, comprising a real (Z') and an imaginary (-Z'') component.
- Nyquist Plot: Plots -Z'' vs. Z' across frequencies. Each point is a single frequency. It provides a concise visual "fingerprint" of the system. Semicircles and sloping lines correspond to specific physical processes (e.g., a depressed semicircle often represents a parallel combination of charge transfer resistance and double-layer capacitance).
- Bode Plot: Consists of two separate graphs: Log |Z| vs. Log(Frequency) and Phase Angle vs. Log(Frequency). It explicitly shows the frequency dependence of impedance magnitude and system time constants, making it superior for identifying processes with overlapping time constants that may be obscured in a Nyquist plot.
Why They Matter Together: The Nyquist plot offers intuitive, compact visualization of circuit elements, while the Bode plot provides clearer resolution of frequency-specific behavior. In battery research, correlating features in both plots is essential for validating an equivalent circuit model.
Key Quantitative Parameters from EIS for Batteries
The table below summarizes critical parameters extracted from Nyquist and Bode plots for a typical lithium-ion battery cell.
Table 1: Key Battery Parameters from EIS Analysis
| Parameter | Common Symbol | Typical Frequency Range | Physical Meaning in Batteries | Extracted Primarily From | ||
|---|---|---|---|---|---|---|
| Ohmic Resistance | RΩ, Rs | High (~10 kHz - 1 MHz) | Resistance from electrolyte, contacts, current collectors. | Nyquist: High-freq x-intercept. Bode: High-freq | Z | plateau. |
| Charge Transfer Resistance | Rct | Medium (~1 Hz - 1 kHz) | Kinetics of redox reaction at electrode/electrolyte interface. | Nyquist: Diameter of mid-freq semicircle. | ||
| Double Layer Capacitance | Cdl | Medium (~1 Hz - 1 kHz) | Capacitance at the electrode/electrolyte interface. | Fitted from semicircle in conjunction with Rct. | ||
| Warburg Impedance Coefficient | σw | Low (< 1 Hz) | Resistance related to solid-state or liquid-phase diffusion of ions. | Nyquist: Low-freq 45° line slope. | ||
| Interfacial Capacitance (SEI) | CSEI | Medium-High (~100 Hz - 10 kHz) | Capacitance of the passive Solid-Electrolyte Interphase layer. | Nyquist: Separate/merged semicircle at higher freq than Rct. |
Application Note: Protocol for EIS Measurement on Coin Cell Batteries
This protocol details a standard EIS experiment for a commercial Li-ion coin cell (e.g., CR2032) to characterize its impedance parameters.
Objective: To obtain impedance spectra for a fresh coin cell, fit the data to an equivalent circuit, and extract Rs, Rct, Cdl, and σw.
Experimental Workflow Diagram:
Title: EIS Measurement and Analysis Workflow for Coin Cells
Detailed Protocol:
- Cell Preparation & Mounting: In a glovebox, insert a freshly assembled or commercial coin cell into a suitable holder with spring-loaded contacts to ensure stable connection. Ensure the holder is connected to the potentiostat's working, counter, and reference leads (2 or 3-electrode configuration).
- Initial Conditioning (Optional but Recommended): Perform 2-3 low-rate (e.g., C/10) charge/discharge cycles on the cell using a battery cycler to stabilize the electrodes and SEI.
- State-of-Charge (SOC) Setting: Bring the cell to the desired SOC (e.g., 50%) using a controlled charge/discharge protocol. Allow the cell to rest at open-circuit voltage (OCV) for a minimum of 1 hour to reach equilibrium.
- Potentiostat Configuration:
- Set the DC bias voltage to the measured OCV of the cell.
- Set the AC perturbation amplitude to 10 mV (typical for linearity).
- Define the frequency range: 1 MHz to 10 mHz.
- Set the number of data points per frequency decade (e.g., 10).
- Select the appropriate integration time or measurement mode for low-frequency stability.
- Measurement Execution: Initiate the frequency sweep. The experiment may take from minutes to several hours depending on the low-frequency limit. Ensure the cell is in a temperature-controlled environment.
- Data Validation: Immediately check the data for stability (e.g., by comparing forward and reverse frequency sweeps) and linearity (Kramers-Kronig transform test is recommended).
- Data Visualization & Circuit Fitting:
- Plot the raw data in both Nyquist and Bode formats.
- Select an initial equivalent circuit model. A common model for a half-cell is: Rs(RctCdl)W (Randles Circuit with Warburg).
- Use non-linear least squares (NLLS) fitting software to fit the model to the data.
- Evaluate fit quality via chi-squared (χ²) values and residual errors.
The Scientist's Toolkit: Key Research Reagent Solutions & Materials Table 2: Essential Materials for EIS in Battery Research
| Item | Function & Importance |
|---|---|
| Biologic SP-300 Potentiostat/Galvanostat/EIS Analyzer | High-performance instrument capable of precise low-current measurement and wide-frequency EIS, crucial for high-impedance battery materials. |
| Faraday Cage | Metallic enclosure that shields the electrochemical cell from external electromagnetic interference (noise), essential for reliable low-frequency and high-impedance data. |
| Temperature-Controlled Chamber | Maintains cell at constant temperature (e.g., 25°C), as impedance parameters are highly temperature-sensitive. |
| High-Purity Argon Glovebox | Provides inert atmosphere for assembling moisture/oxygen-sensitive cells (e.g., Li-metal, sulfide electrolytes). |
| ZView or EquivalentCircuit (EC-Lab) Software | Specialized software for complex nonlinear least squares (CNLS) fitting of EIS data to equivalent circuit models. |
| Standard Coin Cell Hardware (CR2032) | Includes casing, spacers, springs, and gaskets for constructing reliable, reproducible test cells. |
| Lithium Hexafluorophosphate (LiPF6) in EC/DMC Electrolyte | Standard liquid electrolyte solution for Li-ion batteries. Composition and purity directly impact Rs and interfacial impedances. |
| Whatman Glass Microfiber Separators | Porous, inert separator material that holds electrolyte. Its thickness and wettability influence ohmic resistance. |
Equivalent Circuit Modeling: A Conceptual Diagram
The process of interpreting Nyquist/Bode plots involves mapping features to an electrical circuit composed of resistors, capacitors, and specialized elements like constant phase elements (CPE) and Warburg impedances.
Diagram: From EIS Data to Battery Insights via Equivalent Circuit
Title: Mapping EIS Data to Battery Properties via Circuit Models
Nyquist and Bode plots are the essential languages of EIS. For battery researchers developing equivalent circuit models, mastery of both plots allows for robust diagnosis of state-of-health, degradation mechanisms, and kinetic limitations. The structured protocol and conceptual framework provided here serve as a foundation for applying EIS as a powerful, quantitative tool in advanced energy storage research.
Philosophical & Methodological Foundation
Equivalent Circuit Modeling (ECM) for Electrochemical Impedance Spectroscopy (EIS) is a form of phenomenological model-building. Its philosophical basis lies in translating the continuous, frequency-dependent information of an impedance spectrum (Nyquist or Bode plots) into a discrete, intuitively understandable network of electrical components (resistors, capacitors, constant phase elements, Warburg elements). This process is an exercise in representational pragmatism. The model is not a literal depiction of the physical battery but a functional representation that captures key dynamic behaviors (charge transfer, diffusion, interfacial phenomena) for a specific purpose: state estimation, degradation diagnosis, or simulation.
Core Logical Flow of ECM Development:
Title: The Iterative Process of EIS Model Development
Core ECM Elements & Quantitative Correlates
The table below maps common EIS spectral features to ECM components and their hypothesized physical/chemical origins in a Li-ion battery.
Table 1: Mapping EIS Features to ECM Components & Physical Origins
| Spectral Feature (Nyquist) | Primary ECM Component(s) | Typical Parameter Range (Example Li-ion) | Postulated Physical/Chemical Origin |
|---|---|---|---|
| High-Freq Intercept with Real Axis | Ohmic Resistance, Rs (Ω) | 5 – 50 mΩ | Electrolyte ionic resistance, cell geometry, current collector & contact resistances. |
| High-Freq Semicircle | Parallel Rct//CPEdl | Rct: 10 – 200 mΩ; CPEdl-Y0: 0.1 – 10 F·s(α-1) | Charge-transfer kinetics at electrode-electrolyte interface (Rct) & double-layer capacitance with surface inhomogeneity (CPEdl). |
| Mid-Freq Semicircle | Parallel RSEI//CPESEI | RSEI: 5 – 100 mΩ; CPESEI-Y0: 0.001 – 0.1 F·s(α-1) | Solid-Electrolyte Interphase (SEI) layer resistance and capacitance. |
| Low-Freq ~45° Line | Warburg Element (Ws or Wo) | σ (Warburg coeff.): 1 – 50 Ω·s−0.5 | Semi-infinite linear diffusion of Li+ ions in the active material. |
| Low-Freq Upturn (Finite-Length) | Finite-Length Warburg (O) or Capacitance | Cb (Bulk Capacitance): 0.1 – 10 F | Bulk storage (intercalation) or finite diffusion length effects. |
Experimental Protocols for EIS-Based ECM Development
Protocol 3.1: Acquisition of High-Fidelity EIS Data for ECM
Objective: To obtain a clean, stable impedance spectrum suitable for robust ECM fitting.
Materials & Equipment:
- Potentiostat/Galvanostat with FRA: (e.g., Biologic VMP-300, Solartron 1470E/1455A).
- Environmental Chamber: For temperature control (±0.5°C).
- Li-ion Cell (Coin, Pouch, or Cylindrical): In desired state-of-charge (SOC).
- Cabling: Low-inductance, shielded cables with 4-wire Kelvin connection.
- Software: EC-Lab, ZPlot, or equivalent.
Procedure:
- Cell Conditioning: Cycle the cell 2-3 times at C/10 rate to ensure stable SEI. Bring to target SOC (e.g., 50%) and hold at constant voltage until current decays below C/100.
- Thermal Equilibration: Place cell in temperature chamber (e.g., 25°C) for minimum 2 hours.
- Instrument Setup: Connect cell using 4-wire setup. Set DC bias voltage to the cell's open-circuit voltage (OCV) at that SOC.
- EIS Parameters: Apply a sinusoidal perturbation amplitude of 5-10 mV (rms). Ensure linearity by verifying results are amplitude-invariant. Sweep frequency from 100 kHz to 10 mHz (or lower), with 5-10 points per decade. Use a logarithmic sweep.
- Data Collection: Perform at least 3 sequential measurements. The spectra should be superimposable, confirming stationarity.
- Validation: Apply Kramers-Kronig transform checks to validate data causality, linearity, and stability.
Protocol 3.2: Systematic ECM Synthesis & Fitting Protocol
Objective: To transform an EIS spectrum into a validated equivalent circuit model.
Materials & Equipment:
- EIS Data File: From Protocol 3.1.
- Fitting Software: ZView, equivalent circuit (EC-Lab), or Python SciPy lmfit library.
- Physical Priors: Known cell chemistry, electrode architecture.
Procedure:
- Data Pre-processing: Remove obvious outliers. Select relevant frequency range (often omit the highest freq. inductive tail).
- Circuit Hypothesis (Model Selection):
a. Inspect Nyquist Plot: Identify number of depressed semicircles and low-frequency slope.
b. Propose Initial Circuit: Start with a simple model (e.g., R(RC)(RW)).
c. Incorporate Physical Knowledge: For a fresh Li-ion cell, a model like
R_s(R_ct CPE_dl)(R_SEI CPE_SEI)W_smay be appropriate. - Initial Parameter Estimation: a. Use software's "Fit by Dragging" or manual estimation: Rs from high-freq real intercept, semicircle diameters for R's.
- Non-Linear Least Squares (NLLS) Fitting: a. Input initial estimates and perform complex non-linear fitting (both Z' and Z''). b. Use weighting if appropriate (often modulus weighting is a good start).
- Goodness-of-Fit Assessment: a. Calculate χ² value. Aim for χ² < 10⁻³. b. Critical Step: Plot weighted residuals vs. frequency. They should be randomly distributed around zero. Structured residuals indicate an inadequate model.
- Model Validation & Parsimony: a. Apply F-test or Akaike Information Criterion (AIC) if comparing nested/non-nested models. b. The simplest model that yields random residuals and a low χ² should be accepted.
Title: Protocol for Model Selection & Validation
The Scientist's Toolkit: Key Research Reagent Solutions & Materials
Table 2: Essential Tools for EIS-ECM Research in Batteries
| Item | Function & Rationale |
|---|---|
| Potentiostat with High-Precision FRA | Applies precise DC bias with superposed AC perturbation and measures phase-sensitive current/voltage response. Essential for data generation. |
| 4-Wire Kelvin Probe Cables | Eliminates cable and contact resistance from the high-frequency measurement of Rs. Critical for accuracy. |
| Temperature-Controlled Chamber | Electrochemical kinetics are strongly Arrhenius-dependent. Isolating temperature effects is key for identifying physical processes. |
| Reference Electrode (3-Electrode Cell) | Enables deconvolution of anode and cathode contributions to the full-cell spectrum, guiding more physically accurate ECM. |
| Non-Linear Least Squares Fitting Software (e.g., ZView, pyEIS) | Performs the complex optimization to extract ECM parameters from spectral data. The core analytical engine. |
| Kramers-Kronig Validation Tool | Checks data quality before fitting. A model can only be as good as the data it fits. |
| CPE (Constant Phase Element) Component in ECM Software | Real-world capacitors are rarely ideal. CPE (Z=1/(Y₀(jω)^α)) models depressed semicircles from surface roughness or inhomogeneity. |
| Warburg Circuit Elements (Finite/Infinite) | Models diffusion processes. Selecting the correct type (semi-infinite, finite-length, bounded) is critical for low-frequency accuracy. |
Application Notes & Protocols for EIS Battery Research
This section provides a glossary of fundamental circuit elements used in Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling for battery research. Understanding these elements is critical for deconvoluting complex electrochemical processes, such as charge transfer, double-layer formation, and mass transport, which are pivotal in assessing battery health, state-of-charge (SOC), state-of-health (SOH), and degradation mechanisms.
Glossary of Core Elements
Resistor (R)
- Symbol: R
- Physical Origin: Represents pure ohmic resistance to current flow. In a battery, this includes electrolyte resistance, electrode material bulk resistance, and contact resistances.
- Impedance Function: Z(ω) = R
- Nyquist Plot: A single point on the real (Z') axis.
- Phase Angle: 0° at all frequencies.
- Key Application in Batteries: Quantifying the overall internal resistance (IR drop), which impacts power capability and efficiency. A rise in ohmic resistance often indicates electrolyte drying, loss of conductive additives, or poor contact.
Capacitor (C)
- Symbol: C
- Physical Origin: Represents an ideal, frequency-independent charge storage element. In batteries, it models the ideal double-layer capacitance at the electrode-electrolyte interface.
- Impedance Function: Z(ω) = 1 / (jωC)
- Nyquist Plot: A straight line with a -90° slope (vertical line along the -Z'' axis).
- Phase Angle: -90° at all frequencies.
- Key Application in Batteries: Rarely ideal in real systems. Used as a conceptual building block for understanding the non-ideal capacitive behavior typically modeled by a CPE.
Constant Phase Element (CPE or Q)
- Symbol: Q
- Physical Origin: Models non-ideal, distributed capacitive behavior due to surface roughness, porosity, or inhomogeneous current distribution. Ubiquitous in real battery electrode interfaces.
- Impedance Function: Z(ω) = 1 / [Q (jω)^n]
- Parameters: Q (CPE constant, units of S·sⁿ), and n (CPE exponent, 0 ≤ n ≤ 1). For n=1, CPE is an ideal capacitor (C); for n=0, it is an ideal resistor (R); for n=0.5, it resembles a Warburg element.
- Nyquist Plot: A depressed semicircle or a straight line with a phase angle of -(90*n)°.
- Key Application in Batteries: Modeling the double-layer capacitance at porous composite electrodes (e.g., Li-ion anodes/cathodes). The exponent
nis a diagnostic for surface homogeneity; a decrease often correlates with electrode degradation.
Warburg Impedance (W)
- Symbol: W
- Physical Origin: Models semi-infinite linear diffusion of ions in an electrolyte or active material. Assumes diffusion into a bulk medium.
- Impedance Function: Z(ω) = (σ / √ω) - j(σ / √ω) (for semi-infinite Warburg)
- Parameters: σ (Warburg coefficient, Ω·s⁻⁰·⁵).
- Nyquist Plot: A diagonal line with a 45° slope at low frequencies.
- Phase Angle: -45° at all frequencies for the ideal case.
- Key Application in Batteries: Characterizing solid-state diffusion of lithium in active material particles (e.g., in graphite, NMC) or diffusion in the electrolyte. The transition from a 45° line to a vertical line indicates a finite diffusion length (e.g., bounded by particle size).
Table 1: Characteristic Parameters and Signatures of Basic EIS Circuit Elements.
| Element | Symbol | Impedance Formula Z(ω) | Key Parameters | Nyquist Signature | Typical Phase | Primary Battery Process Modeled |
|---|---|---|---|---|---|---|
| Resistor | R | R | R (Ω) | Point on Z' axis | 0° | Ohmic losses (electrolyte, contacts) |
| Capacitor | C | 1/(jωC) | C (F) | Vertical line along -Z'' | -90° | Ideal double-layer charging |
| Constant Phase Element | Q | 1/[Q (jω)^n] | Q (S·sⁿ), n (0-1) | Depressed semicircle/line | -(90*n)° | Non-ideal double-layer (porous electrodes) |
| Warburg (Semi-infinite) | W | σω⁻⁰·⁵(1-j) | σ (Ω·s⁻⁰·⁵) | 45° line at low freq | -45° | Bulk material diffusion |
Experimental Protocol: EIS Measurement for Battery Half-Cell
Objective: To acquire impedance spectra of a Li-ion battery half-cell (e.g., Li-metal vs. NMC cathode) to parameterize an equivalent circuit model.
Materials & Reagents (Scientist's Toolkit): Table 2: Essential Research Reagent Solutions & Materials for EIS in Battery Research.
| Item | Function/Explanation |
|---|---|
| Potentiostat/Galvanostat with EIS Module | Primary instrument for applying a small AC perturbation and measuring the current/voltage response across a wide frequency range. |
| Environmental Chamber | Provides precise temperature control (e.g., 25°C ± 0.5°C) to ensure reproducible electrochemical kinetics and transport properties. |
| CR2032 Coin Cell Hardware | Standard housing for laboratory-scale battery testing, including cases, spacers, springs, and gaskets. |
| Cathode Electrode (e.g., NMC on Al foil) | Working electrode. Active material mass loading and porosity are critical variables. |
| Li-metal Foil (Anode & Reference) | Serves as both counter and reference electrode due to its stable potential. |
| Microporous Polyolefin Separator | Electrically isolates electrodes while allowing ionic conduction (e.g., Celgard 2325). |
| Li-ion Electrolyte (e.g., 1M LiPF6 in EC:EMC) | Provides ionic conduction medium. Composition (salt, solvents, additives) is a key research variable. |
| Argon-filled Glovebox (O2/H2O < 0.1 ppm) | Essential environment for assembling cells with moisture- and oxygen-sensitive materials (Li-metal, charged electrodes, Li-salts). |
| Electrochemical Cell Holder | Fixture to make stable electrical connections to the coin cell during testing. |
Procedure:
- Cell Assembly: Inside an argon glovebox, assemble a CR2032 coin cell in the sequence: cathode case (positive), NMC cathode electrode, separator (saturated with 80 µL electrolyte), Li-metal foil, spacer, spring, and anode case (negative). Crimp the cell using a hydraulic crimper.
- Conditioning & Rest: After assembly, transfer the cell to the temperature-controlled chamber set to 25°C. Allow the cell to rest for a minimum of 2 hours to ensure complete separator wetting and voltage stabilization.
- Instrument Connection: Connect the cell to the potentiostat using the cell holder. The working (red) cable connects to the cathode case, while the counter and reference (both white) cables connect to the anode (Li-metal) case.
- Open Circuit Potential (OCP) Check: Measure and record the OCP. Ensure it is stable (< 1 mV change over 5 minutes) before proceeding.
- EIS Measurement Setup: In the instrument software, configure the EIS experiment.
- DC Bias: Set to the measured OCP.
- AC Amplitude: Typically 5-10 mV RMS. Ensure linearity (current response should be sinusoidal).
- Frequency Range: A broad range from high to low (e.g., 200 kHz to 10 mHz).
- Points per Decade: Minimum 10 points for adequate resolution.
- Data Acquisition: Initiate the scan. The instrument will apply a sinusoidal voltage at each frequency and measure the amplitude and phase shift of the current response.
- Validation: After the scan, check data quality using Kramers-Kronig transforms or by examining the consistency of replicate measurements.
- Circuit Fitting: Use specialized software (e.g., ZView, EC-Lab) to fit the acquired spectrum (Nyquist plot) to a candidate equivalent circuit (e.g., R(QR)(QRW)) and extract parameter values (R, Q, n, σ).
Visualized Relationships
Diagram 1: EIS Elements and Battery Processes (69 chars)
Diagram 2: EIS Experimental Workflow (48 chars)
Within the broader thesis of Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling for advanced battery research, this application note serves as a critical bridge. It maps the abstract electrical components of a typical Randles circuit to the fundamental physical and electrochemical processes occurring within a lithium-ion battery. This mapping is essential for researchers, including those in drug development who utilize similar electrochemical sensing platforms, to translate impedance data into meaningful insights about kinetic limitations, mass transport, and interfacial evolution. Accurate interpretation guides material design, electrolyte formulation, and diagnostic protocols.
Circuit-Process Mapping: Theoretical Framework
The simplified Randles circuit is the foundational model for a single electrode-electrolyte interface. Each component corresponds to a specific physical domain.
Table 1: Mapping of Equivalent Circuit Components to Physical Battery Processes
| Circuit Element | Symbol | Primary Physical Process | Governing Equation / Relationship | Key Influencing Factors |
|---|---|---|---|---|
| Solution Resistance | Rs | Ionic conduction in bulk electrolyte. | Rs = ρ (L/A); ρ = electrolyte resistivity. | Electrolyte conductivity, electrode porosity & geometry, temperature. |
| Charge Transfer Resistance | Rct | Kinetics of Faradaic reaction at electrode surface (e.g., Li+ + e- + Mat Li-Mat). | Butler-Volmer Equation. Rct ∝ 1/(exchange current density, i0). | Temperature, overpotential, electrode catalytic activity, state of charge. |
| Double Layer Capacitance | Cdl | Non-Faradaic charge separation at electrode-electrolyte interface (Helmholtz & diffuse layers). | Cdl = εrε0A / d. | Electrode surface area, electrolyte dielectric constant, SEI properties. |
| Warburg Element | ZW | Semi-infinite linear diffusion of ions in electrolyte/active material. | ZW = σ ω-1/2 (1-j); σ = Warburg coefficient. | Diffusion coefficient (D), concentration gradient, particle geometry. |
| Constant Phase Element | CPE | Non-ideal capacitive behavior from surface heterogeneity, roughness, or porous diffusion. | ZCPE = 1 / [Q (jω)n]; 0 < n ≤ 1. | Electrode surface morphology, inhomogeneous current distribution, SEI layer. |
| SEI Layer Resistance | RSEI | Ionic conduction through the Solid-Electrolyte Interphase layer. | Often in series with Rct. | SEI thickness, composition, uniformity, mechanical stability. |
Experimental Protocols for Parameter Deconvolution
Protocol 3.1: EIS Measurement for Full-Cell Battery Characterization
Objective: To collect impedance spectra for mapping circuit parameters to physical states.
- Equipment Setup: Connect a potentiostat/galvanostat with EIS capability to a climate-controlled battery test chamber. Use a 3-electrode cell setup (if possible) or a 2-electrode full cell.
- Cell Conditioning: Cycle the battery 3 times at C/10 rate to stabilize the SEI. Bring the cell to a specific State of Charge (SOC) (e.g., 50%) and allow it to rest at open-circuit voltage (OCV) for 2 hours to reach equilibrium.
- EIS Acquisition: Apply a sinusoidal voltage perturbation with amplitude of 5-10 mV (rms) over a frequency range from 100 kHz to 10 mHz. Log 10 points per frequency decade. Perform measurements at multiple SOCs (e.g., 20%, 50%, 80%) and temperatures (e.g., 25°C, 10°C, 45°C).
- Data Validation: Ensure Kramers-Kronig transform compliance to verify data linearity, stability, and causality.
Protocol 3.2: Galvanostatic Intermittent Titration Technique (GITT) for Diffusion Coefficient (D) Estimation
Objective: To quantify the lithium-ion diffusion coefficient, informing the Warburg element parameter (σ).
- Procedure: At a fixed SOC, apply a constant current pulse (C/20) for a duration τ (e.g., 1800 s).
- Measurement: Monitor the voltage transient. Follow the pulse with a long rest period (e.g., 7200 s) until the voltage stabilizes to a new steady-state (ΔEs).
- Calculation: Calculate the apparent chemical diffusion coefficient (D) using the simplified equation for short times: [ D = \frac{4}{\pi\tau} \left( \frac{nm Vm}{A} \frac{\Delta Es}{\Delta Et} \right)^2 ] where nm, Vm, A are molar number, molar volume, and electrode area. ΔEt is the voltage change during the constant current pulse, excluding IR drop.
Protocol 3.3: Potentiostatic Hold for SEI Growth Analysis
Objective: To probe SEI layer dynamics and correlate with RSEI evolution.
- Procedure: Hold the electrode at a potential slightly above the Li plating potential (e.g., 50 mV vs. Li/Li+) for a prolonged period (e.g., 24-72 hours) in a 3-electrode cell.
- In-situ EIS Monitoring: Record EIS spectra at predefined intervals (e.g., every 6 hours). Use a equivalent circuit with a RSEI-CPESEI branch in series with the charge transfer (Rct-Cdl) branch.
- Post-Mortem Analysis: Correlate the increase in RSEI and the decrease in CPE exponent 'n' with ex-situ analysis (XPS, SEM) of the harvested electrode to link electrical parameters to physical SEI thickness/composition.
Visualization of Relationships and Workflows
Title: EIS Data to Battery Insight Workflow
Title: Circuit Elements Mapped to Battery Layers
The Scientist's Toolkit: Key Research Reagent Solutions
Table 2: Essential Materials for EIS-Based Battery Interface Studies
| Item / Reagent | Function & Relevance to Circuit-Process Mapping |
|---|---|
| Lithium Hexafluorophosphate (LiPF6) in Carbonate Blends (e.g., EC:DMC) | Standard electrolyte. Its conductivity directly defines Rs; its reduction kinetics and products govern Rct and RSEI/CPE. |
| Vinylene Carbonate (VC) or Fluoroethylene Carbonate (FEC) | Electrolyte additives. Form a stable, low-resistance SEI, directly modifying the RSEI and CPE parameters in the model. |
| Reference Electrode (e.g., Li Metal Foil in Swagelok-type cells) | Enables half-cell or pseudo-3-electrode EIS. Critical for deconvoluting anode and cathode contributions to the full-cell circuit model. |
| Standard Battery Cyclers with EIS Modules (e.g., Biologic, Gamry) | Provides precise potentiostatic control and accurate phase-sensitive impedance measurement across wide frequency ranges. |
| Non-Linear Least Squares (NLLS) Fitting Software (e.g., ZView, EC-Lab) | Essential for robust fitting of EIS data to equivalent circuit models to extract component values. |
| Ex-situ Surface Analysis (XPS, ToF-SIMS) | Correlates fitted parameters (e.g., increasing RSEI) with physical changes in SEI chemistry and thickness. |
| Temperature-Controlled Test Chamber | Allows study of Arrhenius behavior of Rct and D, confirming the kinetic and diffusion-linked nature of these components. |
Within the broader thesis on Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling for battery research, the accurate determination of State-of-Charge (SOC), State-of-Health (SOH), and Internal Resistance is paramount. These parameters are not directly measurable but must be inferred from electrochemical and physical measurements. EIS, coupled with equivalent circuit model (ECM) fitting, serves as a critical tool for deconvoluting the contributions of various internal resistive and capacitive processes to the overall cell impedance, which in turn informs SOC and SOH estimation. This document outlines application notes and experimental protocols for researchers.
Table 1: Key Parameter Definitions, Significance, and Typical Ranges
| Parameter | Definition | Significance in Research & Development | Typical Range (Li-ion Example) | Primary EIS Correlation |
|---|---|---|---|---|
| State-of-Charge (SOC) | The available capacity (%) relative to the current maximum capacity. | Critical for Battery Management Systems (BMS), performance prediction, and cycle life testing. | 0% (Fully Discharged) to 100% (Fully Charged). | Strongly influences charge transfer resistance (semi-circle in mid-frequency range) and diffusion Warburg element (low-frequency slope). |
| State-of-Health (SOH) | A measure of battery aging, expressed as current maximum capacity (%) relative to nominal capacity. | Predicts remaining useful life (RUL), essential for warranty and performance guarantees. | 100% (New) to ~70-80% (End of Life). | Correlates with increase in ohmic internal resistance (high-frequency intercept) and growth of Solid Electrolyte Interphase (SEI) layer resistance (first semi-circle). |
| Internal Resistance | The total opposition to current flow within a cell. Often broken into Ohmic (Rs) and Polarization (Rp) components. | Determines power capability, efficiency, and heat generation. A key indicator of degradation. | Ohmic Rs: < 50 mΩ for 18650 cells. Polarization Rp: Varies with SOC and temperature. | Directly extracted from ECM fitting: Rs (high-frequency Z'), Sum of Rct + RSEI (mid-frequency diameters). |
Table 2: Common Equivalent Circuit Model Elements for Parameter Extraction
| ECM Element | Physical Origin | EIS Signature | Linked Key Parameter |
|---|---|---|---|
| Resistor (Rs) | Electrolyte, separator, current collector resistance. | High-frequency real-axis intercept. | Internal Resistance (Ohmic), SOH. |
| Constant Phase Element (CPESEI) | Non-ideal capacitance of the Solid Electrolyte Interphase layer. | Depressed semi-circle at high-mid frequencies. | SOH (growth over cycles). |
| Resistor (RSEI) | Ionic resistance of the SEI layer. | Diameter of first semi-circle. | SOH, Internal Resistance. |
| Constant Phase Element (CPEdl) | Non-ideal double-layer capacitance at electrode-electrolyte interface. | Depressed semi-circle at mid frequencies. | SOC, active surface area (SOH). |
| Resistor (Rct) | Charge transfer resistance of the electrochemical reaction. | Diameter of second/mid-frequency semi-circle. | SOC (strong function), temperature. |
| Warburg Element (W) | Semi-infinite linear diffusion of lithium ions. | 45° line at low frequencies. | SOC, diffusion limitations. |
Experimental Protocols
Protocol 1: EIS Measurement for SOC and Internal Resistance Profiling
Objective: To obtain a signature EIS Nyquist plot across a range of SOCs for model parameterization. Materials: See "The Scientist's Toolkit" below. Procedure:
- Cell Conditioning: Place the test cell in a temperature-controlled chamber (e.g., 25°C). Perform 3 full formation cycles per manufacturer specs.
- SOC Setting: Charge the cell to 100% SOC using a standard Constant Current-Constant Voltage (CC-CV) protocol. Allow a 2-hour rest period for voltage stabilization.
- EIS Measurement: Configure the potentiostat/galvanostat-EIS. Apply a sinusoidal voltage perturbation with amplitude of 5-10 mV (RMS) over a frequency range from 100 kHz to 10 mHz. Record the impedance spectrum.
- SOC Increment: Discharge the cell by a fixed capacity increment (e.g., 10% SOC) using a constant current. Rest for 1 hour.
- Repeat: Repeat steps 3-4 until reaching 0% SOC. Optionally, repeat during the charge phase.
- Data Analysis: Fit each EIS spectrum (per SOC point) to a pre-defined ECM (e.g., R(CR)(CR)(W)) using non-linear least squares fitting software. Extract Rs, RSEI, Rct, and CPE parameters.
Protocol 2: Cycle Aging for SOH and Internal Resistance Tracking
Objective: To monitor the evolution of SOH and internal resistance components over the battery's lifetime. Materials: As in Protocol 1, with multiple identical cells for statistical relevance. Procedure:
- Baseline Testing (Cycle 0): For each cell, record its initial capacity (C0) via a full discharge from 100% SOC. Perform a reference EIS measurement at 50% SOC (Protocol 1).
- Aging Protocol: Subject cells to repeated charge/discharge cycles under accelerated aging conditions (e.g., 1C rate, 45°C ambient temperature, 100% Depth-of-Discharge).
- Periodic Checkpoint Testing: Every 25-50 cycles, pause the aging protocol. a. Perform a full capacity check (Cn) to calculate SOH: SOH = (Cn / C0) * 100%. b. Perform EIS at 50% SOC and 25°C (standardized condition). c. Resume aging cycles.
- Termination: Continue until cells reach 80% SOH (or other defined End-of-Life criteria).
- Post-Mortem Analysis: Correlate the trajectory of ECM-fitted parameters (especially Rs and RSEI) with the measured SOH decay to build a predictive model.
Protocol 3: Hybrid Pulse Power Characterization (HPPC) for DC Internal Resistance
Objective: To measure the DC internal resistance (pulse power capability) as a function of SOC, complementing EIS AC resistance data. Procedure:
- SOC Setting: Bring the cell to a target SOC (e.g., 90%) and rest for 1 hour.
- Discharge Pulse: Apply a high-current discharge pulse (e.g., 1C or 5C) for 10 seconds. Record the immediate voltage drop ΔVdischarge.
- Rest: Allow a 40-second rest.
- Charge Pulse: Apply a high-current charge pulse for 10 seconds. Record the immediate voltage rise ΔVcharge.
- Resistance Calculation: Calculate DC resistance for each pulse: RDC = |ΔV| / I.
- SOC Increment: Discharge the cell to the next SOC point (e.g., 80%). Rest for 1 hour.
- Repeat: Repeat steps 2-6 across the full SOC window.
Visualization: EIS-ECM Workflow for Parameter Extraction
Title: Workflow for Battery Parameter Extraction via EIS and ECM Fitting
Title: Logical Relationship from EIS Data to SOC and SOH
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions & Essential Materials
| Item | Function/Description | Example Supplier/Product Type |
|---|---|---|
| Potentiostat/Galvanostat with EIS Module | Applies controlled voltage/current perturbations and measures the electrochemical impedance response. | Biologic SP-300, Metrohm Autolab PGSTAT, GAMRY Interface. |
| Battery Cycler | Precisely controls charge/discharge cycles for aging studies and SOC adjustment. | Arbin Instruments BT-5HC, Bio-Logic BCS-800, MACCOR Series 4000. |
| Thermal Chamber | Provides a constant, controlled temperature environment for reproducible aging and testing. | ESPEC BTL-433, Tenney T10C. |
| Electrochemical Cell (Test Cell) | Holds the battery material (coin cell, pouch cell, or full cell) with appropriate fixtures. | EL-CELL PAT-Core, Coin Cell Crimper, custom pouch cell fixtures. |
| Equivalent Circuit Modeling Software | Performs complex non-linear fitting of EIS data to user-defined circuit models. | ZView (Scribner), EC-Lab (Bio-Logic), Relaxed (Freeware). |
| Reference Electrolyte & Solvent | High-purity, battery-grade electrolytes for consistent experimental conditions. | BASF, Sigma-Aldrich Battery Grade EC/DEC with 1M LiPF6. |
| Active Electrode Materials | Well-characterized materials for reproducible electrode fabrication (e.g., NMC622, Graphite). | Targray, MSE Supplies. |
| Separator | High-quality porous membrane to prevent electrical shorting (e.g., Celgard 2325). | Celgard LLC. |
| High-Precision Glovebox | Maintains an inert (Argon) atmosphere with low H2O/O2 (<1 ppm) for safe handling of air-sensitive materials. | MBRAUN, VAC. |
From Theory to Lab Bench: A Step-by-Step Guide to Building and Fitting ECMs
Within the broader thesis on electrochemical impedance spectroscopy (EIS) equivalent circuit modeling (ECM) for battery research, the acquisition of high-quality, reproducible data is the critical foundation. Reliable ECM fitting and subsequent physicochemical interpretation are impossible without data integrity. This application note provides detailed protocols and best practices for the experimental community, emphasizing the stringent control of variables influencing battery impedance.
Prerequisite Cell Conditioning and Stabilization
A battery's impedance is highly sensitive to its state-of-charge (SOC), temperature, and electrochemical history. Inadequate stabilization is the leading cause of irreproducible data.
Protocol 1.1: SOC Conditioning and Open-Circuit Voltage (OCV) Stabilization
- SOC Setting: Bring the cell to the target SOC using a low, constant current (C/20 or lower) charge or discharge, followed by a potentiostatic hold at the voltage corresponding to the target SOC until the current decays below C/100.
- Thermal Equilibration: Transfer the conditioned cell to the temperature-controlled EIS measurement station. Allow for a minimum stabilization period (see Table 1).
- OCV Stability Criterion: Monitor OCV. Proceed only when the voltage drift is less than 0.1 mV per minute over a 30-minute period. For some high-precision studies, a criterion of 0.02 mV/min may be required.
Table 1: Minimum Stabilization Times Post-Conditioning
| Cell Format | Temperature | Minimum Stabilization Time | Key Rationale |
|---|---|---|---|
| Coin Cell (Lab-scale) | 25°C | 2 hours | Homogenization of Li-ion concentration gradients. |
| Pouch Cell | 25°C | 4-6 hours | Larger geometric scale requires longer relaxation. |
| Cylindrical (e.g., 18650) | 25°C | 6-12 hours | Complex, wound geometry extends relaxation time. |
| Any Format | 0°C to 15°C | Multiply above time by 2x | Slower ion diffusion and kinetics. |
| Any Format | 35°C to 50°C | Multiply above time by 0.5x | Faster ion diffusion and kinetics. |
EIS Measurement Parameters and Instrument Configuration
Incorrect perturbation amplitude or frequency parameters can induce non-linear response or insufficient data density for modeling.
Protocol 2.1: Linear Region Verification and Frequency Sweep
- Perturbation Amplitude Selection:
- Perform a preliminary "amplitude sweep" at a middle frequency (e.g., 100 Hz).
- Apply sinusoidal voltage perturbations from 1 mV to 20 mV (peak-to-peak or RMS, as per instrument specification).
- Plot log(amplitude) vs. current response. The linear region is where the response is proportional.
- Typical Amplitude: For most Li-ion batteries, a 5-10 mV (RMS) perturbation is optimal. For highly resistive solid-state cells, 10-50 mV may be necessary.
- Frequency Range and Density:
- Standard Range: 100 kHz to 10 mHz. High-frequency limit is constrained by system inductance; low-frequency limit by cell stability.
- Points per Decade: Minimum 10 points per decade. For detailed analysis of distributed processes (e.g., solid-state diffusion), use 15-20 points per decade.
- Integration Time/Number of Cycles: Configure to ensure sufficient signal-to-noise ratio, especially at low frequencies (e.g., 3-5 cycles per measurement at lowest frequencies).
Title: EIS Measurement Parameter Optimization Workflow
Critical Environmental and Hardware Controls
Uncontrolled variables introduce significant artifacts and scatter.
Protocol 3.1: Temperature Control and Connection Setup
- Temperature: Use an environmental chamber or precision oven. Allow cell temperature to equilibrate, verified by a thermocouple attached to the cell casing. Temperature stability of ±0.5°C is essential; ±0.1°C is ideal.
- Connections & Cabling:
- Use 4-terminal (Kelvin) connections to eliminate cable and contact resistance.
- Use shielded, low-noise cables.
- Keep cables short and geometry consistent between experiments to minimize variable inductance.
- Employ a Faraday cage if measuring very low impedance cells or in electrically noisy environments.
- Hardware Validation: Regularly perform a "dummy cell" calibration measurement using a known resistor-capacitor network to validate instrument and cable performance.
Data Validation and Reproducibility Assessment
A single spectrum is insufficient. Statistical confidence is required for modeling.
Protocol 4.1: Triplicate Measurement and Kramers-Kronig Validation
- Triplicate Acquisition: At the defined SOC and temperature, perform three consecutive EIS measurements without moving the cell or connections.
- Kramers-Kronig (KK) Test: Apply a KK residuals check to each spectrum. This test evaluates if the data is causal, linear, and stable—the fundamental requirements for valid EIS.
- Many modern potentiostats include this software function.
- A "fit error" or residual > 1-2% at any frequency suggests a violation of assumptions (e.g., cell drifting, perturbation too large).
- Acceptance Criteria:
- KK residuals for all three spectra must be within the acceptable threshold.
- The Nyquist plots of the three spectra must overlay visually.
- Quantitative reproducibility: Key features (e.g., high-frequency real axis intercept, diameter of semicircles) should have a coefficient of variation (CV) < 2%.
Table 2: Key Validation Metrics for Reproducible EIS Data
| Metric | Target Value | Measurement Protocol | Implication of Deviation | ||
|---|---|---|---|---|---|
| OCV Drift Rate | < 0.1 mV/min | Protocol 1.1 | Cell is not at equilibrium; impedance will drift. | ||
| KK Residual Max | < 1% (absolute) | Protocol 4.1 | Data violates EIS assumptions; unfit for modeling. | ||
| HF Intercept CV* | < 1% | Protocol 4.1 (Triplicate) | Poor contact resistance or cable stability. | ||
| Semicircle Diameter CV* | < 2% | Protocol 4.1 (Triplicate) | Process instability (e.g., SEI growth, temperature drift). | ||
| Low-Freq Noise | < 5% of | Z | Increased cycles per measurement at low freq. | Poor signal-to-noise; difficult to fit diffusion elements. |
*CV = Coefficient of Variation (standard deviation / mean).
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials and Reagents for Battery EIS Experiments
| Item | Function & Rationale | Key Specifications / Notes |
|---|---|---|
| Biologic VSP-300 (or equivalent) | Potentiostat/Galvanostat with EIS. | High current capability, low current noise, integrated frequency response analyzer (FBA). |
| Environmental Chamber (e.g., Binder) | Precise temperature control. | Stability of ±0.1°C to ±0.5°C; uniform air flow. |
| 4-Terminal Cell Holder | Eliminates lead/contact resistance. | Gold-plated contacts; compatible with coin, pouch, or cylindrical cell formats. |
| Shielded Low-Noise Cables | Minimizes electromagnetic interference. | Coaxial, with high-quality connectors (BNC, alligator clips for contacts). |
| Known RC Dummy Cell | Validates instrument and cable performance. | Typically a 100 Ω resistor in parallel with a 10 µF capacitor. |
| Electrolyte (e.g., 1M LiPF6 in EC:EMC) | Standard battery operation medium. | Battery-grade, ultra-dry (<20 ppm H₂O); composition must be reported. |
| Reference Electrode (e.g., Li-metal ring) | For 3-electrode cell measurements. | Enables deconvolution of anode and cathode impedances. Critical for mechanistic studies. |
| KF Titrator | Measures electrolyte moisture. | Ensures consistency of electrolyte quality, a major variable. |
| KK Validation Software | Checks data quality and validity. | Often built into instrument software (e.g., EC-Lab, Ivium). |
Title: Reliable ECM Depends on Controlled Variables & Validation
The path to meaningful equivalent circuit modeling in battery research begins with meticulous experimental practice. By rigorously applying the protocols for cell conditioning, parameter optimization, environmental control, and statistical validation outlined herein, researchers can generate EIS data of sufficient quality and reproducibility to build robust, physically interpretable models. This disciplined approach is non-negotiable for advancing the fundamental understanding of battery kinetics and degradation mechanisms.
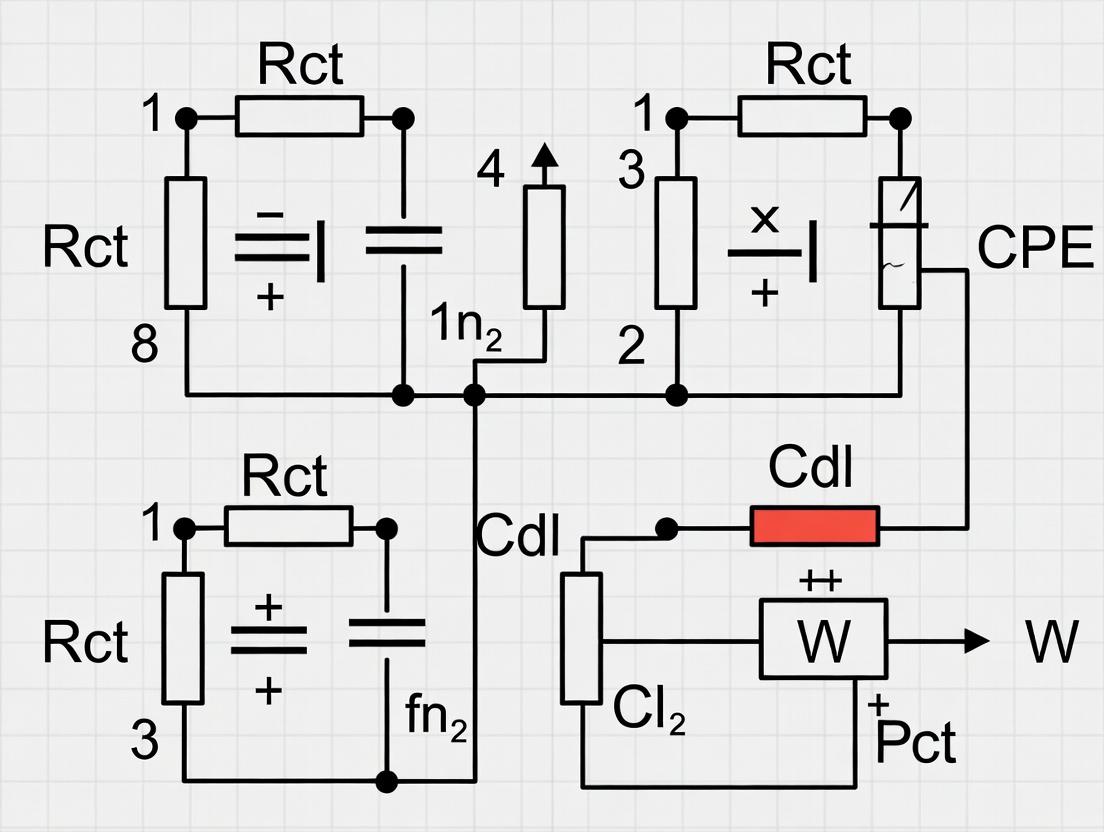
Within the broader research on Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling (ECM) for batteries, selecting an appropriate starting circuit topology is a critical first step. The choice is heavily dependent on the battery's chemistry, as different materials and mechanisms manifest distinct features in the Nyquist and Bode plots. This application note provides a structured guide to common ECM topologies, protocols for initial model selection and validation, and essential research tools for scientists and engineers in battery development.
Common ECM Topologies and Their Corresponding Chemistries
The table below summarizes the recommended starting ECM topologies for various battery chemistries, based on characteristic electrochemical signatures.
Table 1: Starting ECM Topologies for Common Battery Chemistries
| Battery Chemistry | Recommended Starting ECM Topology | Key Physicochemical Elements Represented | Typical Frequency Range of Key Features |
|---|---|---|---|
| Commercial Li-ion (NMC/LFP-Graphite) | [Re(RctQdl)(Qdiff)] | Re: Bulk electrolyte/contact resistance. Rct/Qdl: Charge transfer at electrode-electrolyte interface. Qdiff: Solid-state diffusion in bulk electrode. | 10 kHz (Re), 1 Hz - 100 Hz (Semicircle), <0.1 Hz (Warburg tail) |
| Solid-State Battery (Lithium Metal Anode) | Re(RintQint)(RctQdl) | Rint/Qint: Interfacial resistance/CPE due to passivation layer (SEI). Rct/Qdl: Charge transfer across the solid-solid interface. | 10 kHz (Re), 1 kHz - 10 Hz (2nd Semicircle: interface), 10 Hz - 0.1 Hz (1st Semicircle: charge transfer) |
| Lithium-Sulfur (Li-S) | Re(RctQdlpsQps)] | Rps/Qps: Polysulfide diffusion/shuttling-related resistance and capacitance. Often requires multiple (RQ) parallels for complex precipitation/dissolution. | ~1 kHz (Re), 100-1 Hz (Main semicircle), <1 Hz (Low-frequency diffusion/processes) |
| Lithium Titanate Oxide (LTO) Anode-based | Re(RctQdl) | "Zero-strain" material often shows minimal SEI growth. Circuit is simpler, dominated by charge transfer. Bulk diffusion feature may be absent. | 10 kHz (Re), 100 Hz - 1 Hz (Single dominant semicircle) |
| Nickel-Metal Hydride (NiMH) | Re(R1Q1)(R2Q2)QH | Two (RQ) parallels for processes at nickel hydroxide cathode and metal hydride anode. QH: Hydrogen adsorption/absorption capacitance. | Two overlapping semicircles (mHz - kHz), Very low frequency capacitive line |
Note: Q elements represent Constant Phase Elements (CPE), used instead of ideal capacitors to model depressed semicircles and distributed time constants. The exponent 'n' of the CPE provides insight into surface heterogeneity.
Experimental Protocol for Initial ECM Selection and Validation
Protocol 1: Systematic EIS Measurement for ECM Fitting
Objective: To acquire high-fidelity EIS data for reliable ECM topology selection and parameter extraction.
Materials & Setup:
- Potentiostat/Galvanostat with FRA capability.
- Environmental chamber for temperature control.
- Battery cell (coin, pouch, or cylindrical) at known State of Charge (SOC).
- Data acquisition software.
Procedure:
- Cell Stabilization: Hold the cell at the target SOC for a minimum of 2 hours (or until OCV drift < 0.1 mV/min) to achieve quasi-equilibrium.
- Parameter Setting:
- Signal Amplitude: Apply a sinusoidal voltage perturbation of 5-10 mV RMS. For high-power cells, a current perturbation corresponding to ±5% of C-rate may be used.
- Frequency Range: Sweep from a high frequency (e.g., 100 kHz or instrument maximum) to a low frequency (typically 10 mHz or lower). Use 5-10 points per decade for initial scans.
- Temperature: Conduct measurements at a controlled, constant temperature (e.g., 25°C ± 0.5°C).
- Data Acquisition: Perform the frequency sweep. Record the real (Z') and imaginary (-Z'') impedance components.
- Quality Check: Immediately plot Nyquist and Bode representations. Verify data meets Kramers-Kronig relations or inspect for obvious noise/instability. Repeat if necessary.
Protocol 2: ECM Topology Identification and Fitting Workflow
Objective: To logically progress from EIS data to a validated equivalent circuit model.
Procedure:
- Visual Feature Mapping: Plot the acquired EIS data on a Nyquist plot. Identify distinct features: high-frequency intercept (Re), semicircles (time constants), and low-frequency slopes (diffusion).
- Initial Model Selection: Based on the features identified in Step 1 and the chemistry of the cell (refer to Table 1), choose a starting ECM topology.
- Non-Linear Least Squares (NLLS) Fitting:
- Input the starting ECM and initial parameter estimates into fitting software (e.g., ZView, RelaxIS, or Python
lmfit). - Perform the fit, weighting data appropriately (often by modulus).
- Record the chi-squared (χ²) value and residuals.
- Input the starting ECM and initial parameter estimates into fitting software (e.g., ZView, RelaxIS, or Python
- Model Validation:
- Residual Analysis: Check if residuals are randomly distributed. Non-random patterns suggest a poor model.
- Physical Plausibility: Assess if fitted parameters (R, Q, n) are physically sensible (e.g., positive values, 0 < n <= 1).
- Complexity Increase: If fit is poor, judiciously add elements (e.g., an extra (RQ) parallel branch) and re-fit. Use the F-test or Akaike Information Criterion (AIC) to justify the increased complexity.
- SOC/Temperature Validation: Test if the model structure remains consistent across multiple SOCs and temperatures. A robust model should be broadly applicable.
Diagram Title: Workflow for ECM Topology Identification & Fitting
The Scientist's Toolkit: Essential Research Reagent Solutions & Materials
Table 2: Key Materials and Reagents for Battery EIS/ECM Research
| Item Name | Function/Description | Example/Catalog Consideration |
|---|---|---|
| Reference Electrode | Enables separation of anode and cathode impedances in 3-electrode cell setups. Critical for assigning ECM elements to specific electrodes. | Li-metal ring reference for lab-scale pouch cells; Li-In alloy for SSBs. |
| Galvanostatic EIS Software Module | Controls the potentiostat to apply a sinusoidal current perturbation and measure voltage response. Preferred for very low impedance cells. | Included with Bio-Logic EC-Lab, Gamry Framework, or Solartron Multistat. |
| Non-Linear Fitting Software | Performs complex least-squares fitting of ECM to EIS data, providing parameter values and confidence intervals. | Commercial: ZView (Scribner), RelaxIS (RHD). Open-source: lmfit (Python). |
| Constant Phase Element (CPE) | A non-ideal circuit component used in place of a capacitor to model depressed semicircles, where impedance ZCPE = 1/(Q(jω)n). | A fundamental element in all ECM fitting software. The 'n' parameter indicates surface disorder. |
| Electrolyte/Electrolyte Components | The ionic conductor. Its composition directly impacts Re and interfacial kinetics (Rct). | 1M LiPF6 in EC:EMC (3:7) for Li-ion; LPSCI powder for solid-state cells. |
| Stabilized Lithium Metal Powder (SLMP) | Used to pre-lithiate anodes or create reproducible Li-metal surfaces for reference electrodes, improving interfacial consistency. | Sold by companies like FMC (Albemarle). |
| Environmental Test Chamber | Maintains precise temperature control during EIS measurement, as kinetics and transport are highly temperature-sensitive. | Tenney, Espec, or Binder series chambers. |
Diagram Title: Core Elements of the ECM Fitting Process
Selecting an appropriate starting ECM is not arbitrary but a hypothesis-driven step based on the known electrochemistry of the system. The protocols outlined here provide a reproducible method for model development and validation. Within thesis research on EIS for batteries, this structured approach ensures that subsequent parameter tracking and degradation mode analysis are built upon a physically justified foundation, directly linking circuit elements to underlying chemical and physical processes in Li-ion, solid-state, and next-generation batteries.
Application Notes
Electrochemical Impedance Spectroscopy (EIS) is a cornerstone technique in battery research, enabling the characterization of kinetic and transport phenomena. Equivalent Circuit Modeling (ECM) is the primary method for interpreting EIS data, translating complex spectra into physical and chemical parameters. The choice of software for ECM fitting critically impacts the accuracy, efficiency, and reproducibility of research.
ZView (Scribner Associates) is a dedicated commercial software offering a user-friendly graphical interface for EIS analysis. Its primary strength lies in its robust, iterative fitting algorithm and an extensive, customizable library of circuit elements. It excels in batch processing and provides clear error estimation for fitted parameters. However, its closed-source nature limits customization and automation for high-throughput research pipelines.
EC-Lab (BioLogic Sciences Instruments) is an integrated software suite, often bundled with BioLogic potentiostats. It provides seamless hardware control, real-time data acquisition, and advanced analysis modules, including nonlinear least squares fitting for EIS. Its native integration ensures optimal data fidelity and supports complex protocols like galvanostatic intermittent titration technique (GITT) coupled with EIS. The software's cost and vendor lock-in are primary considerations.
Python/Open-Source Alternatives (e.g., impedance.py, eis-fitting, Relaxation-Time-Analysis) represent a flexible, transparent, and cost-free paradigm. Leveraging libraries such as SciPy, NumPy, and lmfit, they allow for complete control over the fitting algorithm, statistical weighting, and custom model definitions. This facilitates advanced data processing, machine learning integration, and reproducible workflows via scripts. The steep learning curve and need for code verification are notable barriers.
Table 1: Comparative Summary of ECM Fitting Software
| Feature | ZView | EC-Lab (Analysis Module) | Python/Open-Source |
|---|---|---|---|
| Primary Use | Post-acquisition EIS analysis | Integrated acquisition & analysis | Programmable analysis & modeling |
| Cost | ~$2,000 (single license) | Bundled with hardware or separate module | Free |
| Fitting Algorithm | Complex Nonlinear Least Squares | Levenberg-Marquardt | User-selectable (e.g., Levenberg-Marquardt) |
| Custom Circuit Definition | Yes, graphical & scripted | Yes, graphical | Fully programmable via code |
| Automation & Scripting | Limited internal scripting | Basic macro functionality | Full programmability (Python scripts) |
| Error Analysis | Confidence intervals, Chi-squared | Parameter uncertainty | User-defined, extensive statistical libraries |
| Best For | Standardized analysis, quick fitting | Turnkey solution for BioLogic users | Custom models, high-throughput, reproducibility |
Table 2: Example Fitting Performance on a Standard Li-ion Battery Model (R(CR)(RW))
| Software Tool | Typical Fit Time (1000 pts) | Chi-squared (Typical) | Ease of Constraint Application |
|---|---|---|---|
| ZView 3.5 | < 5 s | ~2e-4 | Very Easy (GUI sliders) |
| EC-Lab V11 | < 3 s | ~3e-4 | Easy (GUI input) |
Python (lmfit) |
1-10 s (hardware dependent) | ~1e-4 - 1e-3 | Programmatically Flexible |
Experimental Protocols
Protocol 1: Standardized ECM Fitting Workflow for Battery EIS Data
This protocol outlines a general method for fitting EIS data to an equivalent circuit, applicable across software tools.
Materials & Reagents:
- EIS Data File: Typically in .txt, .csv, .mpr (EC-Lab), or .z (ZView) format.
- Initial Circuit Model: A hypothesis-driven circuit (e.g., Re(RctCPEdl)(Ws)).
- Software Tool: As detailed above.
Procedure:
- Data Import: Load the EIS data file containing frequency (Hz), Z' (Ω), Z'' (Ω), and optionally phase or modulus.
- Data Validation: Inspect the Nyquist and Bode plots for consistency (e.g., no negative frequencies, anomalous spikes). Exclude obvious outliers.
- Circuit Selection: Define the equivalent circuit based on the electrochemical system.
- For a standard Li-ion battery, a common model is:
R1(R2-CPE1)(W1)where:- R1: Ohmic resistance (electrolyte, contacts).
- R2: Charge-transfer resistance.
- CPE1: Constant Phase Element representing double-layer capacitance.
- W1: Warburg element for semi-infinite linear diffusion.
- For a standard Li-ion battery, a common model is:
- Initial Parameter Estimation: Use software tools to seed initial values.
R1: Intercept of high-frequency real axis.R2: Diameter of high-frequency semicircle.CPE-T, CPE-P: Estimate from the semicircle's shape (P often 0.8-1 for batteries).W-R, W-T: Estimate from low-frequency 45° line slope.
- Fitting Execution:
- Apply appropriate weighting (often
modulusorproportionalfor battery data). - Set constraints if necessary (e.g., 0.5 < CPE-P < 1.0).
- Run the iterative complex nonlinear least squares (CNLS) fitting algorithm.
- Apply appropriate weighting (often
- Goodness-of-Fit Assessment:
- Evaluate χ², error sum of squares.
- Examine residuals plot (real and imaginary) for random distribution.
- Visually compare simulated and experimental data on Nyquist/Bode plots.
- Physical Interpretation: Convert fitted parameters (e.g., CPE to effective capacitance) and correlate with battery state (SOC, SOH, temperature).
Protocol 2: High-Throughput EIS Analysis Using Python
This protocol enables automated fitting of multiple EIS spectra for parameter trend analysis.
Materials & Reagents:
- Python Environment: Installation of Anaconda with
numpy,scipy,matplotlib,pandas,impedance.py, andlmfit. - Data Directory: Folder containing multiple EIS files from a cycling experiment.
Procedure:
- Script Setup: Create a new Jupyter Notebook or Python script.
- Import Libraries:
Batch Data Loading:
Define Circuit and Fit:
Output and Visualization: Compile results into a DataFrame and plot parameter evolution vs. cycle number.
Visualizations
Title: Standard ECM Fitting Workflow for Battery EIS
Title: High-Throughput Python Batch Fitting Process
The Scientist's Toolkit: Essential Research Reagents & Materials
Table 3: Key Materials for EIS-based Battery ECM Research
| Item | Function in Research |
|---|---|
| Electrochemical Cell (Swagelok, PEEK) | Provides a stable, sealed environment for the battery materials (cathode, anode, separator) during testing. |
| Reference Electrode (Li-metal foil) | Enables precise potential measurement of the working electrode in 3-electrode cell configurations, critical for deconvoluting anode/cathode contributions. |
| High-Purity Electrolyte (e.g., 1M LiPF6 in EC:DMC) | Standardized ion conduction medium; purity minimizes side reactions that distort EIS measurements. |
| Glass Fiber Separator (Whatman) | Electrically isolates electrodes while allowing ionic conduction; inert and stable in battery electrolytes. |
| Potentiostat/Galvanostat with EIS Module (e.g., BioLogic, Metrohm) | Instrument for applying precise electrical perturbations (sine waves) and measuring the current/voltage response to generate EIS data. |
| Environmental Chamber (Binder, ESPEC) | Controls temperature with high accuracy (±0.1°C), as temperature dramatically affects kinetic parameters (Rct, diffusion). |
| Standard Battery Materials (NMC622, Graphite) | Well-characterized active materials serve as controls for validating ECM fitting protocols and software outputs. |
Within the broader thesis on Electrochemical Impedance Spectroscopy (EIS) for battery state-of-health and degradation analysis, the accurate fitting of equivalent circuit models (ECMs) is paramount. The Nonlinear Least Squares (NLLS) algorithm is the computational cornerstone for extracting physically meaningful parameters (e.g., charge-transfer resistance, double-layer capacitance) from complex impedance spectra. This protocol details the critical steps of defining parameter bounds, assigning weights, and selecting initial guesses to ensure a robust, physically plausible, and convergent fit.
Core NLLS Protocol for EIS ECM Fitting
Pre-Fitting Data Preparation
- Instrument: Potentiostat/Galvanostat with EIS capability (e.g., BioLogic, Autolab).
- Data Format: Ensure impedance data (Z(ω) = Z' + jZ") is corrected for cabling artifacts and open/short circuit calibration.
- Circuit Selection: Choose an appropriate ECM (e.g., R(CRW)) based on the battery chemistry and physical understanding of the electrode-electrolyte interface.
Defining Parameter Bounds (Constraints)
Bounds prevent the NLLS algorithm from converging to non-physical solutions. Constraints are defined as [lower bound, upper bound].
Table 1: Typical Parameter Bounds for Common EIS ECM Elements
| ECM Element | Symbol | Physical Meaning | Lower Bound | Upper Bound | Justification |
|---|---|---|---|---|---|
| Resistor | R | Ohmic/Electrolyte resistance | 1e-6 Ω | 1e3 Ω | Positive, finite resistance. |
| Constant Phase Element | Q0 | Capacitance pseudo-constant | 1e-12 F·s(α-1) | 1 F·s(α-1) | Positive value. |
| CPE Exponent | α | Surface heterogeneity (0<α≤1) | 0.5 | 1.0 | Represents ideal capacitor (α=1) or porous electrode (α~0.5). |
| Warburg Element | Wσ | Diffusion coefficient relation | 1e-3 Ω·s-0.5 | 1e3 Ω·s-0.5 | Positive value. |
| Inductor | L | Cable/connection inductance | 1e-12 H | 1e-3 H | Positive, small value. |
Assigning Data Weights
Weights (wi) adjust the influence of each data point in the minimization of χ² = Σ [wi * (Yi,meas - Yi,calc)²]. For EIS, the choice is critical due to the wide range of impedance magnitudes.
Table 2: Common Weighting Schemes for EIS NLLS Fitting
| Scheme | Formula (wi) | Application Context | Rationale | ||
|---|---|---|---|---|---|
| Unit Weighting | 1 | High SNR data across all frequencies. | Simplest form, assumes equal error. | ||
| Modulus Weighting | 1/ | Zi | ² | General purpose for broad impedance range. | Prevents high-impedance regions (low-frequency) from dominating the fit. |
| Proportional Weighting | 1/(Z'i² + Z"i²) | Similar to modulus weighting. | Emphasizes relative error. | ||
| Statistical (1/σ²) | 1/σi² | When experimental standard deviation (σi) at each point is known. | Theoretically most correct, but σi is rarely measured. |
Protocol: For most battery EIS data, modulus weighting (1/|Z|²) is recommended as a default starting point to balance the fit across the Nyquist plot.
Determining Initial Guesses
Poor initial guesses lead to convergence failures or local minima. Use heuristic methods:
- Visual Inspection: Estimate RΩ from the high-frequency real-axis intercept. Estimate charge-transfer resistance (Rct) from the diameter of the semicircle.
- Analytical Approximations: For a simple R(CR) circuit, initial C can be estimated from the frequency at the top of the semicircle (fmax): Cinit = 1 / (2πfmaxRct).
- Staged/Sequential Fitting: First fit the high-frequency region to get RΩ and inductance, then progressively fit lower frequency elements.
- Genetic Algorithm/ Monte Carlo: Use a global search algorithm on a bounded parameter space to generate a robust initial guess for the final NLLS refinement.
Iterative Fitting and Quality Assessment
- Run the NLLS optimizer (e.g., Levenberg-Marquardt) with bounds, weights, and initial guesses.
- Assess goodness-of-fit:
- χ² (Chi-squared): Lower values indicate a better fit.
- Visual Inspection: Overlay fitted curve on measured Nyquist and Bode plots.
- Residual Analysis: Plot weighted residuals (Zmeas - Zcalc) vs. frequency. Random scatter indicates a good fit; systematic trends suggest an inadequate model.
- If fit is poor, systematically:
- Re-evaluate the appropriateness of the ECM.
- Adjust weighting scheme.
- Slightly perturb initial guesses and re-fit to test for local minima.
Visualization of the NLLS Fitting Workflow
Diagram Title: NLLS Fitting Workflow for EIS Data
The Scientist's Toolkit: Key Research Reagents & Materials
Table 3: Essential Materials for EIS-Based Battery Cell Analysis and Fitting
| Item | Function in EIS Experiments |
|---|---|
| Potentiostat/Galvanostat with FRA | Core instrument for applying perturbative AC voltage/current and measuring impedance response across frequencies. |
| Electrochemical Cell (e.g., Coin Cell, Pouch Cell) | Housing for battery materials (cathode, anode, separator, electrolyte) under test. Must ensure stable electrical connections. |
| Reference Electrode (e.g., Li-metal) | Critical for three-electrode setups to deconvolute anode and cathode contributions to full-cell impedance. |
| Battery Cycler | For cell formation, state-of-charge (SOC) control, and cycle aging prior to EIS measurement. |
| Environmental Chamber | Maintains constant temperature during EIS measurement, as impedance is highly temperature-sensitive. |
| ECM/NLLS Fitting Software (e.g., ZView, EC-Lab, pyEIS) | Software containing NLLS algorithms to perform the fitting protocol, visualize results, and assess residuals. |
| High-Purity Electrolyte & Solvent | Defines ionic conductivity and directly impacts charge-transfer and diffusion-related impedance elements. |
| High-Surface Area Conductive Carbon (e.g., Super P) | Conductivity additive in composite electrodes, influencing ohmic resistance and CPE behavior. |
| Binder (e.g., PVDF, CMC/SBR) | Holds active material particles together; can influence interfacial properties and low-frequency impedance. |
This application note is presented within the framework of a broader thesis research project focused on advancing Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling (ECM) for battery state-of-health (SOH) diagnostics. The core thesis posits that dynamic ECM parameters, tracked over cycle life, provide a more mechanistic and predictive understanding of degradation than standalone capacity fade measurements. This case study applies this principle to a commercial Li-ion coin cell (CR2032 format), correlating cyclical aging with the evolution of a physics-informed ECM.
Core Experimental Protocol
Materials and Cell Preparation
- Cell: Commercial Lithium Cobalt Oxide (LCO)/Graphite CR2032 coin cell (e.g., Panasonic BR2032). Nominal voltage: 3.0V, Typical capacity: 190 mAh.
- Equipment: Biologic VSP-300 potentiostat/galvanostat/EIS analyzer, Arbin LBT battery cycler, Tenney environmental chamber.
- Software: EC-Lab for EIS fitting, Python with SciPy for data analysis.
Cycling Aging Protocol
- Initial Characterization: All cells undergo an initial characterization sequence at 25°C:
- EIS Measurement: At 50% State-of-Charge (SOC), from 1 MHz to 10 mHz, with a 10 mV RMS perturbation.
- Capacity Check: Three C/20 (approx. 9.5 mA) charge/discharge cycles to determine initial capacity (C0).
- Cycling Regime: Cells are placed in a temperature chamber at 25°C ± 0.5°C.
- Charge: Constant Current-Constant Voltage (CC-CV) at 1C (190 mA) to 4.2V, hold until current drops to C/20.
- Discharge: Constant Current (CC) at 1C to 2.5V cut-off.
- Interval Testing: Every 50 cycles, the cycling is paused. Cells are stabilized at 25°C and 50% SOC, followed by a repeat of the EIS Measurement and a Capacity Check (C/20 cycle).
EIS Data Fitting Protocol
- Model Selection: A modified Randles circuit with a Constant Phase Element (CPE) is used as the base ECM, augmented with a second parallel R-CPE element to represent solid-electrolyte interphase (SEI) effects.
- Fitting Procedure in EC-Lab:
- Import EIS spectra (Z' vs Z").
- Define the ECM topology using the software's graphical circuit builder.
- Set initial parameter estimates: R1 (Ohmic) from high-frequency x-intercept, R2 (SEI) and R3 (Charge Transfer) from mid-low frequency semicircles.
- Execute a complex non-linear least squares (CNLS) fit, weighting data by modulus.
- Validate fit quality via chi-squared (χ²) value (< 0.01) and visual overlay.
Table 1: Evolution of ECM Parameters and Capacity Over Cycle Life
| Cycle Count | Discharge Capacity (mAh) | Capacity Retention (%) | Rs (Ω) | RSEI (Ω) | Rct (Ω) | CPEdl-T (F) |
|---|---|---|---|---|---|---|
| 0 | 190.5 | 100.0 | 0.85 | 1.2 | 4.5 | 0.015 |
| 50 | 186.2 | 97.7 | 0.87 | 1.8 | 5.8 | 0.014 |
| 100 | 179.1 | 94.0 | 0.90 | 2.5 | 8.1 | 0.012 |
| 150 | 169.8 | 89.1 | 0.95 | 3.4 | 12.3 | 0.010 |
| 200 | 157.3 | 82.6 | 1.02 | 4.8 | 18.9 | 0.008 |
Table 2: The Scientist's Toolkit: Essential Research Reagents & Materials
| Item | Specification/Example | Primary Function in Experiment |
|---|---|---|
| Potentiostat/Galvanostat with EIS | Biologic VSP-300, GAMRY Interface 5000 | Applies precise electrical perturbations, measures current/voltage response, and performs frequency-domain impedance analysis. |
| Battery Cycler | Arbin LBT, Bio-Logic BCS-800 | Automates the application of charge/discharge profiles for controlled aging and capacity measurement. |
| Environmental Chamber | Tenney T20S, ESPEC BTL | Maintains constant, precise temperature to eliminate thermal variance as a degradation factor. |
| EIS Fitting Software | EC-Lab, ZView | Performs complex non-linear least squares fitting of EIS data to equivalent circuit models to extract physicochemical parameters. |
| Reference Electrode | Li-metal foil in Swagelok-type 3-electrode cell (for parallel validation) | Enables decoupling of anode and cathode impedance contributions in non-commercial cell studies. |
| Electrolyte (for post-mortem analysis) | 1M LiPF6 in EC:DMC (1:1) | Standard electrolyte used to refill disassembled cells for component-level EIS diagnosis. |
Degradation Modeling and Pathway Visualization
The data supports a degradation model where capacity fade is primarily driven by loss of lithium inventory (LLI) and active material loss (LAM), mechanistically linked to rising impedances.
Title: Li-ion Coin Cell Degradation Pathways Linking Stress to EIS Parameters
Title: Experimental and Modeling Workflow for Cycle Life Study
Discussion and Implications for the Broader Thesis
This case study validates the thesis premise. The systematic increase in RSEI directly quantifies LLI from SEI growth, while the rapid rise in Rct correlates with LAM and increased charge transfer difficulty, likely from cathode surface degradation and anode pore clogging. The ECM serves as a transfer function, converting spectral data into parameters with pseudo-physical meaning, enabling the development of a predictive model for SOH based on early-cycle EIS parameter drift rather than requiring full cycle life tests. This methodology is directly applicable to the screening of novel electrolyte formulations or electrode materials in pharmaceutical-grade environmental chambers used in stringent R&D settings.
Navigating Pitfalls: Expert Strategies for Troubleshooting and Refining Your Model Fit
Within the critical research domain of electrochemical impedance spectroscopy (EIS) for battery development, equivalent circuit modeling (ECM) is a cornerstone technique for elucidating internal processes such as charge transfer, diffusion, and SEI layer dynamics. The validity of the drawn conclusions, however, hinges entirely on the statistical integrity of the model fit to the experimental data. A poor fit can lead to incorrect mechanistic interpretations and flawed predictions of battery lifespan and performance. This protocol details the systematic diagnosis of poor fits through the interpretation of chi-squared (χ²) goodness-of-fit metrics and residual error analysis, framed within battery EIS research.
Core Statistical Metrics for Fit Quality Assessment
Chi-Squared (χ²) Value
The reduced chi-squared statistic is the primary quantitative measure of goodness-of-fit in nonlinear least squares (NLLS) fitting, common in EIS analysis software (e.g., Gamry, ZView). It is defined as the weighted sum of squared residuals between the model and the data.
Calculation:
χ² = (1/ν) * Σ [ (Z_real,exp - Z_real,mod)² / σ_real² + (Z_imag,exp - Z_imag,mod)² / σ_imag² ]
where ν is the degrees of freedom (number of data points minus number of fitted parameters), Z_exp is the experimental impedance, Z_mod is the model impedance, and σ is the standard error of the measurement (often estimated).
Interpretation Guide:
- Ideal Fit: χ² ≈ 1. The model explains the data within experimental error.
- Acceptable Fit: χ² < 3. May indicate slightly underestimated measurement errors or minor model imperfections.
- Poor Fit: χ² > 10. Strong evidence the model is inadequate, or measurement errors are poorly characterized.
Residual Error Analysis
Residuals (difference between data and fit) should be randomly distributed. Structured patterns reveal systematic model deficiencies.
Types of Residual Plots:
- Real vs. Frequency: Deviations indicate poor modeling of resistive components.
- Imaginary vs. Frequency: Deviations indicate poor modeling of capacitive/inductive components.
- Complex Residual Plot: Vector representation showing magnitude and phase of misfit.
Table 1: Interpretation of Chi-Squared Values and Corresponding Residual Patterns in Battery EIS
| Reduced χ² Range | Fit Quality | Typical Residual Pattern | Likely Cause in Battery ECM |
|---|---|---|---|
| 0.8 - 3 | Good to Acceptable | Random scatter around zero | Optimal fit; noise-dominated residuals. |
| 3 - 10 | Questionable | Low-frequency drift or periodic structure | Missing a low-frequency diffusion element (e.g., Warburg) or incorrect constant phase element (CPE) exponent. |
| > 10 | Poor | Systematic, high-magnitude trends across wide frequency range | Fundamental model error: e.g., missing a major interfacial (R-CPE) circuit branch, or incorrect topology for cell chemistry. |
| << 1 | Over-fitted / "Too Good" | Artificially random, very low magnitude | Unphysically small weighting (σ), or excessive, correlated circuit parameters without physical meaning. |
Table 2: Common Equivalent Circuit Elements & Their Impact on Residuals
| Circuit Element | Physical Process (Battery Context) | Effect if Missing on Residuals |
|---|---|---|
| Rs | Electrolyte/separator resistance | Offset in real residual at high frequency. |
| Rct∥CPEdl | Charge-transfer resistance & double-layer capacitance | Semicircular misfit in mid-frequency Nyquist plot; structured residuals in Bode magnitude/phase. |
| WO / WS | Semi-infinite / finite-length diffusion (Warburg) | Systematic drift in low-frequency imaginary residuals. |
| L | Inductive wiring/leads | High-frequency misfit in negative imaginary quadrant. |
Experimental Protocol: Systematic Diagnosis of Poor EIS Fits
Protocol Title: Stepwise Workflow for Diagnosing Poor Fits in Battery EIS Equivalent Circuit Modeling.
Objective: To identify the source of a high χ² value and structured residuals in an EIS spectrum fit and propose corrective model actions.
Materials & Software:
- EIS data from battery cell (e.g., Li-ion coin cell cycled at specific state-of-charge).
- ECM software (e.g., Gamry Echem Analyst, Bio-Logic EC-Lab, ZView).
- Statistical plotting software (e.g., Python with SciPy, Matplotlib; OriginLab).
Procedure:
- Initial Fit & Metric Capture:
- Fit your proposed equivalent circuit to the complex impedance data using NLLS.
- Record the final reduced χ² value and all fitted parameters with confidence intervals.
Residual Calculation & Plotting:
- Export the fitted model impedances (
Z_mod) and experimental impedances (Z_exp). - Calculate real and imaginary residuals:
ΔZ' = Z'_exp - Z'_mod;ΔZ" = Z"_exp - Z"_mod. - Create three plots: (i)
ΔZ'vs. log(frequency), (ii)ΔZ"vs. log(frequency), (iii) Complex plane plot ofΔZ'vs.ΔZ".
- Export the fitted model impedances (
Pattern Analysis & Hypothesis Generation:
- Random Scatter: If residuals are random and χ² < 3, the fit is statistically acceptable. Proceed with physical validation of parameter values.
- Low-Frequency Trend (ΔZ"): A dominant trend at low frequencies suggests an inadequate diffusion model. Hypothesis: Replace finite Warburg with infinite, or vice-versa.
- Mid-Frequency Correlation: Correlated peaks/troughs in mid-frequency range. Hypothesis: An additional R-CPE parallel circuit branch is needed to model a second electrochemical interface.
- High-Frequency Offset: Systematic offset in real component at highest frequencies. Hypothesis: Incorrect series inductance (L) or resistance (Rs) value.
Model Iteration & Validation:
- Modify the equivalent circuit topology based on the hypothesis from Step 3.
- Refit the model and compare the new χ² value. A significant decrease (e.g., order of magnitude) supports the hypothesis.
- Perform a likelihood-ratio test or compare Akaike Information Criterion (AIC) scores if models are nested to formally justify added parameters.
Final Physical Sanity Check:
- Ensure all fitted parameters (R, C, CPE exponents) are physically plausible for the battery chemistry and test conditions.
- Cross-validate with a second data set from a replicate cell.
Visualization of Diagnostic Workflow
Title: Diagnostic Workflow for EIS Model Fitting
The Scientist's Toolkit: Key Reagents & Materials for Battery EIS Research
Table 3: Essential Research Reagents & Solutions for Battery EIS Studies
| Item | Function / Role in EIS Experiment |
|---|---|
| Electrolyte Solution (e.g., 1M LiPF₆ in EC:DMC) | Provides ionic conduction medium; its composition directly influences charge-transfer resistance and SEI formation. |
| Electrode Material (e.g., NMC-811, Graphite) | Primary material under investigation; its impedance signature reveals kinetic and diffusional limitations. |
| Reference Electrode (e.g., Li-metal foil) | Provides stable potential reference in 3-electrode cell setups, enabling deconvolution of anode and cathode impedances. |
| Separator (e.g., Celgard polyolefin membrane) | Electrically isolates electrodes; contributes to ohmic series resistance (Rₛ) and may influence low-frequency behavior. |
| Cyclic Carbonate Solvents (EC, DMC, DEC) | Electrolyte solvents that decompose to form the Solid Electrolyte Interphase (SEI), modeled as a constant phase element (CPE). |
| Electrochemical Cell Hardware (Swagelok, Coin Cell) | Provides controlled, reproducible geometry crucial for quantitative comparison of impedance values between experiments. |
| Fitting Software with NLLS (e.g., ZView, MEISP) | Performs the complex regression analysis to extract equivalent circuit parameters from raw impedance spectra. |
Within the broader thesis on Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling for battery research, addressing non-ideal behavior is paramount. Real electrochemical interfaces rarely exhibit perfect capacitive behavior, often deviating from the ideal capacitor model described by a pure capacitance (C). This deviation, manifesting as depressed or skewed semicircles in Nyquist plots, is effectively modeled using Constant Phase Elements (CPEs). CPEs are empirical circuit elements whose impedance is defined as (Z_{CPE} = 1/[Q(j\omega)^n]), where (Q) is the CPE constant (in (S\cdot s^n)), (j) is the imaginary unit, (\omega) is the angular frequency, and (n) is the CPE exponent ((0 \leq n \leq 1)). The use of CPEs is critical for accurate physical interpretation of EIS data from batteries, as they account for distributed time constants due to surface roughness, porosity, and inhomogeneous reaction rates.
Key Principles: When to Use a CPE
A CPE should be used instead of an ideal capacitor when:
- The Nyquist plot shows a depressed semicircle (center below the real axis).
- The Bode phase angle plot shows a broad, frequency-dependent peak.
- Modeling interfaces known for intrinsic inhomogeneity (e.g., porous electrodes, solid-electrolyte interphase (SEI) layers, corroding surfaces).
- When n < 0.9. An ideal capacitor has n=1. Values of n significantly less than 1 indicate the need for a CPE.
Quantitative Data on CPE Parameters in Battery Systems
Table 1: Typical CPE Parameters for Common Battery Components
| Battery Component | Typical Q Value Range | Typical n Exponent Range | Physical Interpretation of n |
|---|---|---|---|
| Porous Cathode (e.g., NMC) | 1e-5 – 1e-3 S·sⁿ | 0.7 – 0.9 | Related to pore geometry and surface roughness. Lower n indicates higher disorder. |
| Solid-Electrolyte Interphase (SEI) | 1e-6 – 1e-5 S·sⁿ | 0.8 – 1.0 | n~1 indicates a near-capacitive, homogeneous layer. n<0.9 suggests inhomogeneous growth. |
| Double Layer at Liquid Electrolyte Interface | 1e-5 – 1e-3 S·sⁿ | 0.8 – 1.0 | Depression often due to atomic-level roughness or adsorption processes. |
| Grain Boundaries in Solid Electrolytes | 1e-8 – 1e-6 S·sⁿ | 0.5 – 0.8 | Reflects the distributed resistive and capacitive nature of interfaces between grains. |
| Lithium Metal Anode (during plating/stripping) | 1e-4 – 1e-2 S·sⁿ | 0.6 – 0.8 | Low n values linked to fractal surface growth and non-uniform current distribution. |
Table 2: Comparison of Equivalent Circuit Models for a Depressed Semicircle
| Model | Circuit Elements | Advantages | Limitations | Best Used For |
|---|---|---|---|---|
| Ideal (Randle-Type) | R(CT) + C | Simple, physically clear. | Fails to fit non-ideal data accurately. | Idealized, smooth surfaces. |
| CPE-Adjusted | R(CT) + CPE | Excellent fit to empirical data. Accounts for distribution. | Empirical; direct capacitance value not obtained. | Most real-world, inhomogeneous interfaces. |
| Distributed Element (Bounded) | R + Finite-Length Warburg (O) | Based on physical diffusion model. | More complex. Specific to finite-length diffusion. | Porous electrodes with restricted diffusion. |
| Multiple Parallel R-C | (R1+C1) // (R2+C2) // ... | Can deconvolute multiple physical processes. | Over-parameterization risk. Less unique fit. | Systems with discrete, separable time constants. |
Experimental Protocols for Effective CPE Application
Protocol 4.1: EIS Measurement for CPE Parameter Extraction
Objective: To acquire high-quality impedance data suitable for CPE-based equivalent circuit fitting.
- Cell Preparation: Assemble a coin cell or pouch cell with the battery materials under study (e.g., NMC cathode, graphite anode, separator, electrolyte). Ensure consistent pressure and temperature control (e.g., 25°C ± 0.5°C).
- Electrochemical Station Setup: Use a potentiostat/galvanostat with frequency response analyzer (FRA) capability. Connect the cell to the potentiostat's working, counter, and reference electrodes (for 3-electrode setup) or working and counter (for 2-electrode).
- Cell Conditioning: Cycle the cell for 3-5 formation cycles at C/10 rate to establish stable interfaces (e.g., SEI).
- State-of-Charge (SOC) Definition: Bring the cell to a specific SOC (e.g., 50%) using a constant-current constant-voltage (CCCV) protocol, followed by a long relaxation period (≥ 2 hours) to reach voltage equilibrium.
- EIS Measurement Parameters:
- Excitation Signal: Apply a sinusoidal potential perturbation with amplitude of 5-10 mV (rms). Ensure linearity by verifying the current response is sinusoidal.
- Frequency Range: Typically 1 MHz to 10 mHz. Log-distributed frequencies (e.g., 10 points per decade).
- Integration Time/AC Cycles: Set to ensure sufficient signal-to-noise ratio at low frequencies (e.g., ≥ 5 AC cycles per measurement point).
- Data Validation: Perform Kramers-Kronig transform tests on the acquired data to check for stability, linearity, and causality before proceeding to fitting.
Protocol 4.2: Equivalent Circuit Modeling with CPEs
Objective: To fit EIS data and extract meaningful parameters using CPE-containing circuits.
- Circuit Selection: Based on the physical system and Nyquist shape, propose an initial equivalent circuit. A common model for a single interfacial process is: ( R{electrolyte} + (R{ct} // CPE_{dl}) ).
- Initial Parameter Estimation: Use the impedance spectra for initial guesses:
- ( R{electrolyte} ): High-frequency real-axis intercept.
- ( R{ct} ): Diameter of the depressed semicircle.
- ( Q, n ): Adjust to fit the shape of the semicircle. Initial guess: n=0.8, Q=1e-5.
- Non-Linear Least Squares Fitting: Use fitting software (e.g., ZView, EC-Lab, or Python's
lmfit). Weight the data appropriately (often by the modulus). - Goodness-of-Fit Assessment: Evaluate using chi-squared ((χ^2)) and error percentages for each parameter. Visually inspect the fit overlaid on Nyquist and Bode plots.
- Physical Validation & Conversion (if needed): Calculate an effective capacitance, (C{eff}), from the CPE parameters using the Brug formula: (C{eff} = (Q \cdot R_{ct}^{1-n})^{1/n}). This allows for comparison with ideal capacitor values.
- Report: Report (Q), (n), (R{ct}), and derived (C{eff}) with their associated fitting errors. Always report the full circuit diagram.
Diagrams
Decision Flow for CPE Modeling
CPE Impedance Formula Breakdown
Circuit Model Selection Based on Interface
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials for EIS and CPE Analysis in Battery Research
| Item | Function / Rationale | Example Product / Specification |
|---|---|---|
| Potentiostat/Galvanostat with FRA | Applies potential/current perturbation and measures impedance response across frequency. Core instrument for EIS. | Biologic VMP-3, Metrohm Autolab PGSTAT302N. Requires µHz to MHz capability. |
| Temperature-Controlled Test Chamber | Maintains isothermal conditions (±0.5°C). Temperature fluctuations significantly impact kinetics and impedance. | ESPEC BTZ-133, Memmert IPP series. |
| Electrochemical Cell (3-Electrode) | Enables isolation of working electrode impedance. Critical for studying single electrodes (anode or cathode). | EL-CELL PAT-Cell, Swagelok-type T-cell with Li reference. |
| High-Purity Battery Electrolyte | Minimizes side reactions and parasitic impedance from impurities. Essential for reproducible interface studies. | 1M LiPF6 in EC:EMC (3:7), battery grade, <20 ppm H2O. |
| Reference Electrode | Provides stable potential reference in 3-electrode setup. | Lithium metal foil, Li4Ti5O12 (LTO) electrode. |
| Modeling & Fitting Software | Performs complex non-linear least squares fitting of equivalent circuits (including CPEs) to EIS data. | Scribner Associates ZView, BioLogic EC-Lab, Equivalent Circuits. |
| Kramers-Kronig Validation Tool | Checks EIS data for consistency, stability, and causality before fitting. | Integrated in EC-Lab, or custom code (Python, MATLAB). |
| Porous Electrode Sheets | Model systems for studying non-ideal, CPE-dominated behavior. | Commercial NMC622 or Graphite coated on Al/Cu foil. |
In Electrochemical Impedance Spectroscopy (EIS) modeling for battery research, a fundamental challenge lies in selecting an equivalent circuit model (ECM) that balances goodness-of-fit with predictive power and physical interpretability. Overfitting occurs when a model is excessively complex, capturing not only the underlying electrochemical processes but also experimental noise, leading to parameters that are statistically unstable and physically meaningless. This document outlines application notes and protocols to enforce parsimony and ensure the physical realism of extracted parameters, framed within a thesis on advanced battery diagnostics.
Principles of Parsimony in ECM Development
Parsimony, or Occam's razor, dictates that the simplest adequate model is preferable. In EIS, this translates to using the minimum number of circuit elements required to accurately represent the impedance data within the bounds of experimental error.
Quantitative Model Selection Criteria
The following criteria should be calculated and compared across nested or competing models. Lower values for AICc and BIC, and a p-value > 0.05 for the LRT, indicate a preferred, parsimonious model.
Table 1: Quantitative Criteria for Model Selection
| Criterion | Formula | Interpretation in EIS Context | Threshold/Goal |
|---|---|---|---|
| Adjusted R² | $R^2_{adj} = 1 - \frac{(1-R^2)(n-1)}{n-p-1}$ | Measures variance explained, penalizing extra parameters (p). | Maximize, but watch for diminishing returns. |
| Akaike Information Criterion (AIC) | $AIC = 2p + n \ln(SSE/n)$ | Estimates relative information loss; lower is better. | Prefer model with ΔAIC > 2 vs. next best. |
| Corrected AIC (AICc) | $AICc = AIC + \frac{2p(p+1)}{n-p-1}$ | Adjusted for small sample sizes (n). Essential for EIS. | Prefer model with lowest AICc. |
| Bayesian Information Criterion (BIC) | $BIC = p \ln(n) + n \ln(SSE/n)$ | Penalizes complexity more strongly than AIC for large n. | Prefer model with lowest BIC. |
| Likelihood Ratio Test (LRT) | $ \chi^2 = -2 \ln \frac{L{simple}}{L{complex}} $ | Tests if complex model fits significantly better. | p-value > 0.05 favors simpler model. |
Where: n = number of data points (frequencies × 2 for Z', Z''), p = number of free parameters, SSE = sum of squared errors, L = likelihood of the model.
Protocol for Stepwise Model Development and Validation
Protocol 1: Iterative ECM Identification and Pruning
Objective: To systematically identify a parsimonious ECM with physically meaningful parameters.
Materials:
- EIS spectrometer (e.g., Biologic VMP-3, GAMRY Interface 5000)
- Battery cycler and thermal chamber
- Software: ZView (Scribner), EC-Lab (BioLogic), or Python (impedance.py, lmfit packages)
Procedure:
- Initial Data Acquisition:
- Acquire EIS data over a relevant frequency range (e.g., 10 kHz to 10 mHz) at a defined state-of-charge (SOC), temperature, and state-of-health (SOH). Use a perturbation amplitude ensuring linearity (typically 5-10 mV).
- Perform Kramers-Kronig transformation or residual analysis to validate data consistency.
Baseline Model Postulation:
- Start with a basic, physically justified model (e.g., R
Ω-(RQ)SEI/CFL-(RQ)CT-WDifffor an intercalation electrode). - Fit the model. Visually inspect the fit on Nyquist and Bode plots. Calculate initial goodness-of-fit (χ², error on parameters).
- Start with a basic, physically justified model (e.g., R
Complexity Introduction (Only if Justified):
- If a systematic misfit is observed (e.g., a depressed semicircle is inadequately fitted by a single (RQ) element), consider a justifiable expansion (e.g., replacing one (RQ) with two series (RQ) elements for two distinct physical processes).
- Refit the new, more complex model.
Model Comparison & Pruning:
- Calculate criteria from Table 1 for both the simple (nested) and complex model.
- Perform a Likelihood Ratio Test. If the p-value is not significant (p > 0.05), the simpler model is statistically sufficient.
- Compare AICc/BIC values. If the complex model does not yield a decrease > 2-10 units (for AICc/BIC respectively), retain the simpler model.
Physical Plausibility Check:
- Proceed to Section 3.1 to validate the parameters of the selected model.
Cross-Validation:
- Validate the final model on EIS data from the same cell at a different SOC or temperature, or from a replicate cell.
- Refit only the parameters—not the model structure—to the new data. The parameters should vary smoothly and predictably with the changed condition.
Title: Workflow for Parsimonious Equivalent Circuit Model Selection
Ensuring Physical Meaning of Extracted Parameters
An ECM is only as good as the interpretability of its parameters. Extracted values must correspond to plausible electrochemical properties.
Protocols for Physical Validation
Protocol 2: Parameter Trend Analysis with Operating Condition
Objective: To verify that ECM parameters follow expected trends with changing cell conditions, confirming their link to physical properties.
Procedure:
- Design Experiment: Perform EIS measurements on a battery at multiple, well-defined states:
- SOC Series: e.g., 100%, 80%, 50%, 20%, 5% SOC at constant temperature.
- Temperature Series: e.g., 0°C, 10°C, 25°C, 40°C at constant SOC.
- SOH Series: Over many charge-discharge cycles (e.g., every 50 cycles).
- Model Application: Fit the same parsimonious ECM structure (from Protocol 1) to all datasets. Only the parameter values should be optimized.
- Trend Validation: Plot each parameter vs. the changing condition. Compare to established physical expectations:
Table 2: Expected Physical Trends for Common ECM Parameters
| Circuit Element | Typical Physical Origin | Expected Trend with Increasing SOC | Expected Trend with Increasing Temperature | Typical Physical Range |
|---|---|---|---|---|
| RΩ (Ohmic Res.) | Electrolyte, current collectors | Relatively constant | Decreases (ionic conductivity ↑) | 10-200 mΩ (for 18650 cell) |
| RSEI/CFL | Solid-Electrolyte Interphase | May increase slightly | Decreases (Arrhenius behavior) | 10-500 mΩ |
| QSEI/CFL (Y₀, n) | CPE for SEI capacitance | Y₀ may evolve; n constant | n constant (~0.8-1) | n: 0.7-1.0 |
| RCT | Charge Transfer Resistance | Often minimum at mid-SOC | Strong decrease (Arrhenius) | 10 mΩ - 2 Ω |
| QDL (Y₀, n) | CPE for Double Layer | Varies with potential | n constant | n: 0.8-1.0 |
| Wo (Warburg) | Solid-State Diffusion | Varies with Li⁺ concentration | Follows diffusion coefficient | - |
- Flagging Anomalies: Parameters that violate expected trends (e.g., RCT increasing with temperature) indicate potential overfitting, an incorrect model, or an unstable fit, requiring reinvestigation.
Title: Parameter Trend Validation Protocol Workflow
Protocol 3: Statistical Robustness and Error Correlation Analysis
Objective: To assess the stability of the fit and identify overly correlated parameters, a sign of over-parameterization.
Procedure:
- After fitting, extract the co-variance matrix or the confidence intervals for all parameters (standard output in good fitting software).
- Analyze confidence intervals: If the 95% confidence interval for any parameter spans an order of magnitude or includes zero for a resistive element, the parameter is not well-defined.
- Calculate parameter correlation coefficients from the co-variance matrix. High correlation (|r| > 0.9) between two parameters indicates they are not independent; the model may be trying to resolve two parameters for a process described by one. This often calls for model simplification.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Tools for Robust EIS Modeling
| Item / Reagent Solution | Function in EIS Modeling Research |
|---|---|
| Stable Reference Electrode (e.g., Li metal in Li-ion systems) | Enables precise decoupling of anode and cathode impedances for more fundamental, less ambiguous modeling. |
| Controlled Environment Chamber | Allows EIS at multiple, stable temperatures for Arrhenius analysis of resistive elements, a key physical validation. |
| Kramers-Kronig Validation Tool (Integrated in software or custom code) | Tests EIS data linearity, causality, and stability before modeling. Invalid data guarantees meaningless parameters. |
| Constant Phase Element (CPE) Implementation | Use instead of ideal capacitors in most battery ECMs to account for surface inhomogeneity and fractal geometry. |
| Distribution of Relaxation Times (DRT) Analysis Software | A model-free tool to identify the number and time constants of physical processes present before committing to an ECM, guiding parsimonious model choice. |
| Global Fitting Algorithm | Fits a single ECM to multiple EIS spectra (e.g., at different SOCs) simultaneously, enforcing shared parameters where physically justified, reducing overfitting risk. |
| Bootstrapping or Monte Carlo Script | Assesses parameter uncertainty by repeatedly fitting data with added synthetic noise, providing robust confidence intervals. |
Addressing Frequency Range Limitations and Artifacts in Experimental Data
Within the broader thesis on Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling for battery research, a critical challenge is the accurate acquisition and interpretation of impedance data. Experimental data are often constrained by the frequency range limitations of the potentiostat and corrupted by artifacts arising from instrumental, environmental, and cell setup factors. These issues directly compromise the validity of the derived equivalent circuit models (ECMs), leading to incorrect estimations of state-of-charge (SOC), state-of-health (SOH), and degradation mechanisms. This application note details protocols to identify, mitigate, and correct for these limitations and artifacts, ensuring robust data for reliable ECM fitting.
Common Artifacts and Limitations: Identification & Quantification
The table below summarizes the primary sources of error in EIS measurements for battery cells.
Table 1: Common EIS Artifacts, Causes, and Identifiable Features
| Artifact / Limitation | Primary Cause | Typical Frequency Manifestation | Impact on Nyquist Plot |
|---|---|---|---|
| Inductive Loop at High Frequency | Cabling inductance, cell fixture geometry. | >10 kHz | Positive imaginary impedance semicircle. |
| Time-Domain Drift | Changing cell potential (e.g., relaxation, slow side reactions). | Very Low Frequency (<1 mHz) | Open, non-closing low-frequency tail; data point scatter. |
| Instrumental Noise Floor | Potentiostat current measurement limits. | Very High & Very Low Frequencies | Scatter and erratic points at spectrum extremes. |
| Frequency Range Cap | Potentiostat hardware/software limits. | Typically <1 mHz lower limit, >1 MHz upper limit. | Incomplete semicircles or missing diffusion tail. |
| DC Offset Error | Incorrect OCP measurement or settling. | Affects all frequencies, most evident in model fit. | Vertical shift of entire spectrum on real axis. |
| Electrode Misalignment | Non-uniform current distribution in test cell. | Mid to Low Frequencies (1 kHz - 1 Hz) | Distorted or "smeared" semicircles. |
| Thermal Fluctuations | Lack of temperature control. | Low Frequencies (<0.1 Hz) | Distortion and poor reproducibility of the Warburg region. |
Title: Data Corruption Pathway from Artifacts to Faulty Models
Core Experimental Protocols
Protocol 3.1: Pre-Measurement Validation and Setup
Objective: Minimize introduction of artifacts through rigorous experimental preparation.
- Cell Conditioning: Prior to EIS, condition the battery cell (e.g., Li-ion) with 3 full charge-discharge cycles at a slow rate (C/10) to establish stable electrode interfaces.
- Open Circuit Potential (OCP) Stabilization: Hold the cell at the desired SOC until the potential drift is < 0.1 mV/min for at least 5 minutes. Record the final stable OCP.
- Cabling & Connection:
- Use shielded, twisted-pair cables of minimal practical length.
- Employ a 4-terminal (Kelvin) connection to the cell to eliminate lead resistance.
- Ensure all connections are clean and tight.
- Environmental Control: Place the cell in a temperature-controlled chamber (±0.5 °C). Allow thermal equilibration for at least 2 hours after chamber setpoint is reached.
Protocol 3.2: Staircase Frequency EIS with Drift Monitor
Objective: Acquire impedance data while monitoring for time-domain drift.
- Instrument Settings: Configure the potentiostat.
- AC Amplitude: Apply a sinusoidal voltage perturbation of 5-10 mV RMS, ensuring linear response (verify by checking amplitude independence).
- Frequency Range: Typically 1 MHz to 10 mHz. Acknowledge hardware limits.
- Points per Decade: 8-10 for initial surveys; increase to 15 for final model-fitting data.
- Staircase Measurement: Perform the EIS scan from high to low frequency. Critical Step: At each frequency, after the standard measurement, pause for a time equal to 3 perturbation periods. Measure the DC current. If the DC current shows a monotonic drift exceeding 1% of the AC current amplitude, flag the data point and consider truncating the sweep.
- Replicate Measurement: Immediately repeat the scan in the opposite direction (low to high frequency). Overlay the two spectra to identify hysteresis indicative of cell instability.
Protocol 3.3: Post-Measurement Data Validation & Correction
Objective: Identify and, where possible, correct artifacts in acquired data.
- Kramers-Kronig (KK) Transform Test: Apply a KK compatibility check to the impedance data. Most modern EIS software suites include this tool. Data points that fail the KK test should be considered corrupted and removed prior to ECM fitting.
- Inductance Correction:
- Inspect the high-frequency end of the Nyquist plot for a positive imaginary component.
- Model the lead inductance (L) as a series element. Estimate L from the high-frequency data (Z'' = ωL).
- Subtract the inductive impedance (Zcorr = Zmeas - jωL) from the entire dataset.
- Data Truncation: Based on the drift monitor (P.3.2) and KK test, truncate the unreliable low-frequency data. Report the exact frequency range used for final analysis.
Title: EIS Data Acquisition and Validation Workflow
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Materials for Artifact-Free EIS in Battery Research
| Item | Function & Relevance to Artifact Reduction |
|---|---|
| High-Precision Potentiostat/Galvanostat with FRA | Core instrument. Must have wide frequency range (e.g., 10 µHz to 1 MHz), low current noise floor (< 1 pA), and 4-terminal sensing capability to measure cell impedance accurately. |
| Shielded, Twisted-Pair Cables with Low Inductance | Minimizes electromagnetic interference (EMI) pickup and reduces cabling inductance that causes high-frequency artifacts. |
| Four-Electrode Battery Test Cell (e.g., Swagelok-type) | Enables true 4-point probing, separating current injection and voltage sensing electrodes to eliminate the impedance of leads and contacts from the measurement. |
| Temperature-Controlled Chamber (±0.1°C Stability) | Critical for stabilizing electrochemical kinetics and diffusion coefficients. Eliminates thermal drift, a major source of low-frequency distortion. |
| Electrochemical Impedance Modeling Software (e.g., ZView, EC-Lab) | Used for KK testing, visual inspection, artifact correction (e.g., inductance subtraction), and equivalent circuit modeling. |
| Reference Electrode (e.g., Li-metal foil) | For 3-electrode cell setups. Allows separation of anode and cathode impedances, resolving ambiguities and misalignment artifacts in 2-electrode spectra. |
| Ultra-Pure Electrolyte and Dry Room/Glovebox | Prevents parasitic side reactions and unstable SEI layers that cause time-domain drift during low-frequency measurements. |
Data Integration into Equivalent Circuit Modeling
The final, validated dataset is used for ECM fitting. The table below summarizes the consequences of uncorrected artifacts on model parameters for a common Li-ion battery Randles circuit model (RΩ + Rct//CPE + Warburg).
Table 3: Impact of Artifacts on Common Battery ECM Parameters
| Uncorrected Artifact | Most Affected ECM Parameter | Typical Error Introduced |
|---|---|---|
| Inductive Loop | Electrolyte Resistance (RΩ) | Overestimation, as HF intercept is shifted right. |
| Time-Domain Drift | Warburg Coefficient (σ) & Charge Transfer Resistance (Rct) | Severe overestimation and unphysical values. |
| DC Offset | Electrolyte Resistance (RΩ) | Constant over- or under-estimation across all SOCs. |
| High-Frequency Noise Floor | Constant Phase Element (CPE) exponent (n) | Underestimation, making CPE appear more resistive. |
| Frequency Range Cap (Low) | Warburg Coefficient (σ) | Underestimation or complete failure to resolve diffusion. |
Title: Impact of Data Quality on ECM Fitting Outcomes
Application Notes
This document provides advanced protocols for optimizing Equivalent Circuit Model (ECM) fitting in Electrochemical Impedance Spectroscopy (EIS) studies of batteries, critical for evaluating state-of-health, degradation mechanisms, and material performance within battery research.
1. The Role of Kramers-Kronig (K-K) Relations in EIS Data Validation EIS data must be causal, linear, and stable to be physically meaningful. The Kramers-Kronig relations provide an integral test for this adherence by linking the real and imaginary components of the complex impedance. In battery EIS, non-stationary behavior due to changing State-of-Charge (SoC) or temperature often leads to K-K violations, which must be identified before model fitting. Modern applications involve numerical validation algorithms (e.g., measurement model, polynomial fitting) to flag unreliable frequency data points.
2. Genetic Algorithm (GA) for Robust ECM Parameterization Traditional non-linear least squares (NLLS) fitting of ECMs (e.g., Randles circuit, distribution of relaxation times models) is prone to converging on local minima, especially with complex circuits representing porous electrodes and solid-electrolyte interphases (SEI). GAs, a global optimization heuristic, overcome this by evolving a population of parameter sets (genes) through selection, crossover, and mutation. This is particularly valuable for identifying unique parameter sets for Warburg elements, constant phase elements (CPE), and nested time constants in multi-particle battery systems.
3. Integrated Workflow: Validation Before Optimization The synergistic application first validates the frequency-domain dataset using K-K checks. Data points or frequency regions that violate K-K relations are either excluded or weighted less in the subsequent fitting process. The GA then operates on the validated data to find the global optimum for the ECM parameters, dramatically improving the physical interpretability and predictive accuracy of the model for battery dynamics.
Protocols
Protocol 1: Kramers-Kronig Validation of Battery EIS Data
Objective: To test the validity of acquired EIS spectra for causality, linearity, and stability.
Materials & Software:
- Potentiostat/Galvanostat with EIS capability.
- Battery cell under test (coin cell or pouch cell) at fixed SoC and temperature.
- Software with K-K validation (e.g., ZView, PyEIS, custom Python/Matlab scripts).
Procedure:
- Experimental Data Acquisition:
- Stabilize the battery at the desired SoC (e.g., 50%) and temperature (e.g., 25°C) for 2 hours.
- Acquire EIS spectrum in potentiostatic mode with a 10 mV RMS perturbation. Typical frequency range: 100 kHz to 10 mHz.
- Perform at least three replicate measurements to ensure consistency.
Data Pre-processing:
- Average the replicate spectra.
- Convert raw data to complex impedance Z(ω) = Z' + jZ''.
K-K Validation Algorithm (Measurement Model Approach):
- Fit the complex data to a measurement model comprising a linear combination of M Voigt elements (resistor + capacitor in parallel) and an inductor.
- The model impedance is: Zmodel(ω) = jωL + Σ [Rk / (1 + jωRkCk)] from k=1 to M.
- Use linear least squares to solve for Rk, Ck, and L.
- Calculate the residuals between the measured Z(ω) and the K-K compliant Z_model(ω).
- Data points where the residual exceeds a threshold (e.g., 5% of |Z|) are marked as invalid.
Output:
- A validated dataset and a report of excluded frequency points.
Protocol 2: Genetic Algorithm Fitting of an Equivalent Circuit Model
Objective: To determine the optimal parameters for a chosen battery ECM using a global search algorithm.
Materials & Software:
- Validated EIS dataset from Protocol 1.
- Computer with optimization software (e.g., MATLAB Global Optimization Toolbox, Python DEAP or SciPy).
Procedure:
- Define the ECM and Parameter Bounds:
- Select an appropriate circuit (e.g., modified Randles: Rs + (Qdl || (Rct + Zw))).
- Define realistic physical bounds for each parameter (e.g., Rs: [0.01, 0.1] Ω, Rct: [10, 200] Ω, Qdl-Y0: [1e-6, 1e-3], Zw-R: [5, 100] Ω).
Configure the Genetic Algorithm:
- Population Size: 100-500 individuals.
- Fitness Function: Weighted sum of squared errors (WSSE) between modeled and validated data, with inverse weighting on invalid points from Protocol 1.
- Selection: Tournament selection (size 3).
- Crossover: Scattered crossover (probability 0.8).
- Mutation: Adaptive feasible (probability 0.1).
- Stopping Criteria: Stall generations (50) or maximum generations (500).
Execution and Analysis:
- Run the GA for 10 independent trials to ensure convergence to a consistent global minimum.
- Extract the best-fit parameter set and its confidence intervals via bootstrap analysis.
- Simulate the best-fit model and overlay it on the experimental data to assess goodness-of-fit (χ², relative error).
Data Tables
Table 1: Comparison of ECM Fitting Methods for a Li-ion Coin Cell EIS Dataset (1 MHz - 0.01 Hz)
| Fitting Method | Number of Valid Data Points (Post K-K) | Optimal χ² Value | R_s (Ω) | R_ct (Ω) | Q_dl-Y0 (S·s^a) | Q_dl-a | Z_w-R (Ω) | Total Computation Time (s) |
|---|---|---|---|---|---|---|---|---|
| NLLS (Local) | 48 | 1.2e-3 | 0.051 | 85.3 | 4.7e-4 | 0.92 | 12.1 | 2.1 |
| GA (Global) | 48 | 3.8e-4 | 0.049 | 87.5 | 4.2e-4 | 0.94 | 11.8 | 124.7 |
Table 2: K-K Validation Output for a Degraded Battery Module
| Frequency Region | Violation Severity (Residual %) | Probable Physical Cause in Battery | Recommended Action for Fitting |
|---|---|---|---|
| > 10 kHz | 1.2% | Inductive wiring artifacts | Include series inductance (L) in ECM |
| 100 Hz - 1 Hz | 8.7% | SoC drift during measurement | Exclude region from fit |
| < 0.05 Hz | 15.4% | Non-stationary diffusion process | Exclude region from fit |
Visualizations
Short Title: EIS Data Validation and Fitting Workflow
Short Title: Genetic Algorithm Iteration Cycle
The Scientist's Toolkit
Table 3: Key Research Reagent Solutions & Materials for Advanced EIS Analysis
| Item Name | Function/Benefit in Battery EIS Research |
|---|---|
| Gamry Reference 3000AE | Potentiostat with advanced EIS suite for high-precision, low-impedance measurements on battery cells. |
| Bio-Logic VMP-3B | Multi-channel potentiostat enabling high-throughput EIS testing of multiple battery cells under identical conditions. |
| ZView Software (Scribner) | Commercial software offering robust K-K validation routines and flexible non-linear fitting tools for ECMs. |
| PyEIS (Open-Source Python Package) | Customizable toolkit for implementing numerical K-K checks, GA fitting, and distribution of relaxation times analysis. |
| MATLAB Global Optimization Toolbox | Provides a stable, well-documented GA function (ga) for scripting complex, multi-parameter ECM fitting workflows. |
| High-Stability Battery Climate Chamber | Maintains precise temperature control (±0.1°C) during EIS measurement to ensure data stability and K-K compliance. |
| Symyx/Citrine Informatics Battery Database | Cloud platform for comparing fitted ECM parameters against a large corpus of published battery EIS data. |
Ensuring Accuracy: Validating ECM Results and Comparing Model Architectures
Within the broader thesis on Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling (ECM) for battery research, model validation is the critical step that transitions a hypothesized circuit from a mathematical construct to a reliable tool for predicting battery state-of-health (SOH), state-of-charge (SOC), and degradation mechanisms. This document provides Application Notes and Protocols for three fundamental validation pillars, enabling researchers to rigorously assess model credibility before deployment in applications such as battery management systems (BMS) or lifetime prediction.
Error Analysis for EIS Circuit Models
Error analysis quantifies the discrepancy between the model's impedance prediction and the experimental EIS measurement across the entire frequency spectrum.
Protocol 2.1: Calculation of Goodness-of-Fit Metrics
- Objective: To compute quantitative error metrics after ECM parameterization via non-linear least squares (NLLS) fitting.
- Materials:
- Experimental impedance data:
Z_exp(ω) = Re_exp + j*Im_exp - Model impedance function:
Z_model(ω, p), wherepis the vector of fitted parameters (e.g., R, C, CPE). - Software: Python (with SciPy, lmfit), MATLAB, ZView, or equivalent.
- Experimental impedance data:
- Procedure:
- Complete the NLLS fitting routine to obtain the optimal parameter set
p_opt. - Calculate the model impedance
Z_modelat all experimental frequencies. - Compute the residual errors for real and imaginary components.
- Calculate the chi-squared (χ²) statistic:
χ² = Σ [ (Re_exp - Re_model)²/σ_Re² + (Im_exp - Im_model)²/σ_Im² ], where σ are weight estimates. - Calculate the root mean square error (RMSE) for impedance magnitude.
- Complete the NLLS fitting routine to obtain the optimal parameter set
- Data Presentation: Standard Goodness-of-Fit Metrics Table
Prediction vs. Experiment Under Dynamic Conditions
This method validates the model's predictive power by comparing its forward simulation against independent experimental data not used during parameter fitting, often under varying operational conditions (temperature, C-rate, cycling age).
Protocol 3.1: Temporal Validation of Model Parameters
- Objective: To assess if ECM parameters, identified at one time/cycle, can predict EIS behavior at a later cycle.
- Materials:
- EIS datasets from a cycling aging study (e.g., at cycles 1, 100, 200).
- ECM structure finalized from initial error analysis.
- Scripting environment for batch fitting and simulation.
- Procedure:
- Parameterization: Fit the ECM to the EIS data from Cycle 1. Store parameters
p_cycle1. - Prediction: Use the model with
p_cycle1to simulate the impedance spectrum expected at Cycle 100. Do not re-fit to Cycle 100 data. - Experimental Comparison: Acquire the actual experimental EIS data at Cycle 100.
- Validation: Compare the simulated (predicted) spectrum from Step 2 directly with the experimental spectrum from Step 3 using RMSE or visual overlay on a Nyquist plot.
- Analysis: Repeat, fitting at cycle N and predicting cycle N+X. A robust model will show low prediction error over short intervals, with error increasing as physical degradation mechanisms not captured by the model evolve.
- Parameterization: Fit the ECM to the EIS data from Cycle 1. Store parameters
Visualization 3.1: Prediction vs. Experiment Workflow
Title: Workflow for Temporal Model Prediction Validation
Cross-Validation in ECM Selection
Cross-validation (CV) mitigates overfitting by assessing model performance on data subsets not used for training. It is crucial for selecting the most robust circuit topology among candidates.
Protocol 4.1: k-Fold Cross-Validation for ECM Topology Selection
- Objective: To compare multiple candidate ECMs (e.g., R(QR)(QR) vs. R(QR)(Q)) and select the one with the most generalizable performance.
- Materials:
- A full, high-quality EIS dataset (typically >50 frequency points).
- 2-3 proposed ECM topologies.
- Automated scripting for data partitioning, fitting, and error calculation.
- Procedure:
- Partitioning: Randomly shuffle the EIS data points (frequency by frequency) and split them into
kequal-sized folds (e.g., k=5). - Iteration: For each candidate ECM, perform
kiterations. In iterationi:- Designate fold
ias the validation set. The remaining k-1 folds form the training set. - Fit the ECM parameters using only the training set data.
- Use the fitted model to calculate the RMSE on the held-out validation set (
RMSE_val_i).
- Designate fold
- Scoring: For each ECM, compute the mean RMSE across all
kvalidation folds. - Selection: The ECM topology with the lowest mean validation RMSE is preferred, as it demonstrates consistent performance without overfitting to the complete dataset.
- Partitioning: Randomly shuffle the EIS data points (frequency by frequency) and split them into
Visualization 4.1: k-Fold Cross-Validation Process
Title: k-Fold Cross-Validation Iterative Scheme
The Scientist's Toolkit: Research Reagent Solutions for EIS Validation
Table 5.1: Essential Materials and Computational Tools
| Item / Solution | Function in EIS Model Validation |
|---|---|
| Potentiostat/Galvanostat with FRA | The core hardware for acquiring experimental EIS data. Frequency Response Analyzer (FRA) capability is essential for precise impedance measurement. |
| Environmental Chamber | Provides controlled temperature for experiments, crucial for validating model performance across temperature-dependent kinetic and transport phenomena. |
| Cycling Equipment (Battery Testers) | Used to age cells under controlled protocols, generating the independent data sets required for "Prediction vs. Experiment" validation over time. |
| High-Precision LCR Meter | For independent validation of component values in constructed analog circuits, sometimes used as a sanity check for model parameters. |
| ECM Fitting Software (e.g., ZView, RelaxIS, EC-Lab) | Commercial packages with robust NLLS algorithms for parameter estimation and initial error analysis. |
| Scientific Programming Environment (Python, MATLAB) | Essential for automating validation protocols, implementing custom cross-validation splits, and batch processing of data. Libraries: SciPy, lmfit (Py); impedance.py (Py). |
| Reference Electrodes (for 3-electrode cells) | Enables deconvolution of anode and cathode impedances, allowing for validation of electrode-specific sub-models within a full-cell ECM. |
| Symmetric Cell Configurations | Cells with identical electrodes simplify the EIS response, serving as a critical validation system for isolating and validating models for SEI or charge transfer at a single electrode. |
1. Introduction Within battery research, Electrochemical Impedance Spectroscopy (EIS) data interpretation hinges on model selection. The choice between a phenomenological Equivalent Circuit Model (ECM) and a physics-based model (e.g., derived from the Pseudo-Two-Dimensional model) is critical. This document provides protocols for systematic benchmarking to determine the sufficiency of a phenomenological ECM for a given research objective, framed within a broader thesis on EIS for batteries.
2. Key Comparison Framework & Quantitative Benchmarks The decision to use an ECM is guided by the research question's required depth of mechanistic insight versus the need for computational speed and parametric simplicity.
Table 1: Comparative Metrics for Model Selection
| Metric | Phenomenological ECM | Physics-Based Model | Benchmark for ECM Sufficiency |
|---|---|---|---|
| Parametric Physicality | Circuit elements (R, C, W) represent lumped phenomena; parameters may not be directly physical. | Parameters directly linked to material properties & transport (D, k, ε, c). | ECM is sufficient if only relative changes or "health indicators" are needed, not absolute property values. |
| Extrapolation Fidelity | Poor outside fitted conditions (T, SOC, SOH, C-rate). | Good, as it captures governing physical equations. | ECM is insufficient for predicting performance under unseen operational protocols. |
| Computational Cost | Very low (seconds). | High (minutes to hours). | ECM is sufficient for real-time or on-board applications (e.g., BMS). |
| Frequency Range Validity | Valid only within the frequency range of fitting. | Theoretically valid across all relevant time scales. | ECM is sufficient if the experimental EIS frequency range is fixed and representative. |
| Identifiability | High risk of correlation between non-physical elements. | Parameters are structurally identifiable but may require multi-modal data. | ECM is insufficient if unique parameter resolution is critical for diagnosis. |
| Typical R² Fit (EIS) | > 0.99 (excellent fit common). | > 0.98 (slightly lower due to model complexity). | A high R² alone is not a sufficient benchmark for ECM validity. |
3. Experimental Protocol for Systematic Benchmarking This protocol outlines steps to rigorously test an ECM's sufficiency against a physical model.
Protocol 1: The Multi-Conditional ECM Stress Test Objective: To evaluate the robustness and predictive failure of an ECM across varying operational conditions. Materials: Cycled battery cells, potentiostat/EIS analyzer, environmental chamber. Procedure:
- Define the Application Context: State the primary use case (e.g., SOH tracking, electrolyte degradation sensing).
- Select Candidate Models:
- Phenomenological ECM: e.g., R(CR)(CR)(W) (Ohmic, SEI, charge transfer, diffusion).
- Physics-Based Benchmark: e.g., Single Particle Model with Electrolyte (SPMe) impedance implementation.
- Acquire Multi-Dimensional EIS Dataset:
- Perform EIS measurements at 5-10 discrete SOC points (e.g., 10%, 50%, 90%).
- Repeat at 3+ temperatures (e.g., 0°C, 25°C, 45°C) using an environmental chamber.
- Include cells at different States of Health (SOH: 100%, 90%, 80%).
- Global Parameterization:
- Fit the phenomenological ECM independently to each EIS spectrum.
- Fit the physical model globally to the entire dataset, sharing physical parameters (e.g., diffusion coefficient) across conditions where they should be constant.
- Benchmarking Analysis:
- Trend Analysis: Plot ECM parameters (e.g., Rct) vs. SOC/T/SOH. Compare trends to those of physical parameters (e.g., reaction rate constant k).
- Predictive Test: Train ECM parameters on a subset of conditions (e.g., 25°C only). Attempt to predict the EIS spectrum at an untrained condition (e.g., 0°C). Compare prediction error to that of the physical model.
- Identifiability Check: Perform a sensitivity analysis on the ECM. Calculate correlation matrices of its fitted parameters. High correlation (>0.9) indicates phenomenological lumping that reduces physical insight.
4. Visualizing the Benchmarking Workflow
Title: Decision Workflow for Benchmarking ECM Sufficiency
5. The Scientist's Toolkit: Key Research Reagents & Materials Table 2: Essential Materials for EIS Model Benchmarking Studies
| Item | Function / Relevance |
|---|---|
| Potentiostat/Galvanostat with FRA | Core instrument for performing accurate EIS measurements. Must have low-current and low-impedance capabilities. |
| Environmental Chamber | For controlling temperature during EIS tests, critical for evaluating model performance across temperatures. |
| Cycled Battery Cells (Various SOH) | Test subjects representing different degradation states. Commercial 18650 or custom pouch cells are typical. |
| Reference Electrodes (3-electrode setup) | Enables decoupling of anode and cathode impedances, providing richer data for physical model validation. |
| Physics-Based Modeling Software (e.g., COMSOL, PyBaMM, DANDELION) | Platform for implementing and simulating the impedance of physics-based models for benchmarking. |
| ECM Fitting Software (e.g., ZView, pyimpspec, EquivalentCircuit.jl) | Specialized software for robust, often non-linear, least squares fitting of circuit models to EIS data. |
| Global Fitting Optimization Toolbox (e.g., MEIGO, SciPy optimize) | Essential for parameterizing physical models by fitting multiple datasets simultaneously. |
Within the broader thesis on Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling (ECM) for battery research, the selection of an appropriate circuit topology is a critical step. This application note provides a detailed protocol for the systematic evaluation and comparison of multiple ECMs when fitted to impedance data from the same battery cell. The objective is to establish a reproducible methodology for researchers to determine the most physically representative, yet parsimonious, model for their specific cell chemistry and state-of-charge (SOC).
Key Research Reagent Solutions & Materials
| Item | Function in ECM Analysis |
|---|---|
| Potentiostat/Galvanostat with FRA | Generates the small-signal AC perturbation and measures the current/voltage response across a frequency spectrum. |
| High-Precision Battery Cycler | Conditions the cell to a precise SOC and maintains voltage stability during EIS measurement. |
| Thermal Chamber | Maintains the cell at a constant, known temperature (e.g., 25°C ± 0.1°C) to eliminate temperature-induced variance. |
| EIS Software (e.g., EC-Lab, VersaStudio) | Controls the measurement hardware, acquires the Nyquist and Bode data. |
| ECM Fitting Software (e.g., ZView, MEISP) | Performs complex nonlinear least squares (CNLS) fitting of the impedance data to user-defined circuit topologies. |
| Reference Electrode (3-electrode cell) | (Optional but recommended) Isolates and decouples the impedance contributions of the positive and negative electrodes. |
Experimental Protocol for Data Acquisition
Title: Standardized EIS Measurement Protocol for LIBs. Objective: To acquire high-fidelity, reproducible impedance spectra for subsequent ECM analysis.
Procedure:
- Cell Conditioning: Cycle the commercial Li-ion cell (e.g., NMC811/Graphite) between specified voltage limits (e.g., 3.0 - 4.2V) for 3 full cycles at C/10 to establish stable SEI.
- SOC Setting: Charge or discharge the cell to the target SOC (e.g., 50% SOC) using a constant-current constant-voltage (CCCV) protocol. Apply a constant voltage hold until the current reaches C/100 to ensure electrochemical equilibrium.
- Thermal Equilibration: Place the cell in a thermal chamber set to 25°C. Hold for a minimum of 2 hours to ensure temperature uniformity.
- EIS Measurement:
- Settings: Apply a sinusoidal voltage perturbation with amplitude of 10 mV (rms). Sweep frequency from 100 kHz to 10 mHz.
- Points per Decade: 10.
- Integration: Use the "Potentiostatic EIS" mode with auto-integration for accuracy at low frequencies.
- Validation: Perform a Kramers-Kronig test on the acquired data to confirm stability, linearity, and causality.
Protocol for Comparative ECM Fitting & Evaluation
Title: Workflow for Comparative ECM Topology Analysis. Objective: To fit, evaluate, and compare candidate ECMs using a standardized set of criteria.
Procedure:
- Topology Selection: Define 3-4 candidate ECMs of varying complexity (see Table 1 and Section 5).
- Initial Parameter Estimation: For each model, use fitting software to make intelligent initial guesses (e.g., from the Nyquist plot: high-frequency intercept for RΩ, semicircle diameter for Rct).
- CNLS Fitting: Perform the fit for each topology. Constrain physically nonsensical parameters (e.g., capacitance, CPE exponent) to positive values between 0 and 1.
- Goodness-of-Fit Evaluation: Record the chi-squared (χ²) value and the weighted sum of squares for each fit.
- Parameter Validation: Assess the physical plausibility of fitted values (e.g., Rct should be positive, CPE-n for a double layer should be ~0.8-1.0).
- Residual Analysis: Examine the relative residual plots (real and imaginary) across the frequency range. A good fit shows random, low-magnitude residuals.
- Model Selection: Apply the Akaike Information Criterion (AIC) to penalize overfitting and select the most justified model.
Common ECM Topologies & Data Presentation
Table 1 summarizes four common ECM topologies fitted to the same NMC/Graphite cell dataset at 50% SOC and 25°C.
Table 1: Quantitative Comparison of Fitted ECM Topologies
| Model Name & Diagram | Topology (R=Resistor, C=Capacitor, Q=CPE, W=Warburg) | Fitted Parameters (Example Values) | Goodness-of-Fit (χ²) | AICc | Key Interpretation |
|---|---|---|---|---|---|
| Randle's Model | RΩ-(Rct//Cdl) | RΩ=1.5 mΩ, Rct=45.2 mΩ, Cdl=1.2 F | 8.7E-3 | -1254 | Poor fit. Misses diffusion and second time constant. |
| Randle's w/ Warburg | RΩ-(Rct//Cdl)-Ws | RΩ=1.5 mΩ, Rct=42.1 mΩ, Cdl=1.1 F, Ws-σ=0.25 Ω s⁻⁰·⁵ | 3.1E-3 | -1421 | Captures diffusion but misses mid-frequency electrode asymmetry. |
| Dual-RC with CPE | RΩ-(RSEI//QSEI)-(Rct//Qdl) | RΩ=1.5 mΩ, RSEI=6.3 mΩ, QSEI=2.5 F, n=0.85, Rct=38.5 mΩ, Qdl=0.9 F, n=0.92 | 9.2E-4 | -1588 | Good fit. Separates SEI and charge-transfer processes. CPE accounts for surface heterogeneity. |
| Dual-RC w/ Finite Warburg | RΩ-(RSEI//QSEI)-(Rct//Qdl)-O | RΩ=1.5 mΩ, RSEI=6.5 mΩ, QSEI=2.4 F, n=0.86, Rct=38.0 mΩ, Qdl=0.9 F, n=0.93, O-R=0.15 Ω, O-T=150 s | 7.5E-4 | -1623 | Best fit. Accounts for SEI, charge-transfer, and finite-length diffusion (O-element). Most physically complete. |
Visualization of ECM Evaluation Workflow
Title: ECM Selection & Validation Workflow
Visualization of Common ECM Topologies
Title: Hierarchy of Common Battery ECM Topologies
Correlating ECM Parameters with Complementary Techniques (e.g., Cyclic Voltammetry, Post-Mortem Analysis)
Within the broader thesis on Electrochemical Impedance Spectroscopy (EIS) equivalent circuit modeling (ECM) for battery research, a critical step is the validation and physical interpretation of the fitted circuit parameters. ECM alone provides a phenomenological representation of electrochemical processes. Correlating these parameters (e.g., charge-transfer resistance, double-layer capacitance, Warburg coefficient) with data from complementary techniques is essential to deconvolute their physical meaning, such as linking them to specific degradation modes (SEI growth, lithium plating, active material loss). This Application Note details protocols for integrating Cyclic Voltammetry (CV) and Post-Mortem Analysis with ECM parameter tracking.
Key Correlative Data Tables
Table 1: Correlation of ECM Parameters with CV-Derived Metrics
| ECM Parameter (from Nyquist Fit) | Complementary CV Metric | Correlation Purpose | Typical Quantitative Relationship (Example Li-ion) |
|---|---|---|---|
| Charge-Transfer Resistance (R_ct) | Peak Separation (ΔE_p) | Kinetics of redox reaction | ΔEp increases linearly with increasing Rct. For reversible system, Rct ∝ (ΔEp - (59/n) mV). |
| Double-Layer Capacitance (C_dl) | Capacitive Current (i_c) at fixed potential | Electrode/electrolyte interfacial area | Cdl estimated from slope of ic vs. scan rate plot. |
| Warburg Coefficient (σ_w) | Peak Current Ratio (ipa/ipc) | Diffusion limitations & reversibility | Deviation from ipa/ipc = 1 correlates with increased σ_w, indicating diffusion hindrance. |
| SEI Resistance (R_SEI) | Integrated Charge in SEI formation region | SEI growth and stability | Increase in R_SEI correlates with increased charge passed in low-voltage CV sweeps. |
Table 2: Post-Mortem Analysis Findings vs. Degraded ECM Parameters
| Post-Mortem Observation (e.g., SEM/EDX/XPS) | Affected ECM Parameter | Proposed Degradation Mode |
|---|---|---|
| Visible lithium dendrites on anode surface | Increased R_ct, new low-frequency element | Lithium plating & increased interfacial resistance. |
| Thick, inhomogeneous SEI layer (XPS: LiF, Li₂CO₃) | Significant increase in R_SEI | Excessive electrolyte reduction & SEI growth. |
| Cathode particle cracking (SEM) | Increased σ_w (diffusion) | Loss of electrical contact, increased diffusion path. |
| Transition metal dissolution (EDX) | Decrease in Cdl, increase in Rct | Loss of active material, degraded electrode surface. |
Experimental Protocols
Protocol 1: Synchronized EIS and Cyclic Voltammetry for Kinetics Decoupling
Objective: To directly correlate Rct and Cdl with faradaic and capacitive currents across a potential window.
Materials: (See "Scientist's Toolkit" below). Procedure:
- Cell Stabilization: Cycle the battery (e.g., Li-ion coin cell) for 5 cycles at C/10 to form a stable SEI.
- State-of-Charge (SOC) Definition: Bring the cell to a specific SOC (e.g., 50%) using a constant current-constant voltage (CCCV) protocol.
- EIS Measurement: At the defined SOC, perform EIS measurement from 1 MHz to 10 mHz with a 10 mV perturbation amplitude. Record the data.
- ECM Fitting: Fit the EIS spectrum using an appropriate circuit (e.g., R(QR)(QRW)) to extract parameters: RΩ, RSEI, CSEI, Rct, Cdl, σw.
- CV Measurement: Immediately following EIS, at the same SOC and temperature, initiate a cyclic voltammetry scan over a narrow, non-destructive potential window centered on the open-circuit potential (e.g., OCV ± 50 mV). Use scan rates from 0.05 to 1.0 mV/s.
- Data Analysis:
- From CV: Calculate peak separation (ΔEp) for each scan rate. Plot ΔEp vs. scan rate; extrapolate to v→0 to estimate kinetic overpotential.
- From CV: Plot the capacitive current (at a potential with no faradaic activity) vs. scan rate. The slope is proportional to C_dl.
- Correlate: Plot extracted Rct from EIS against 1/(intercept of ΔEp vs. v plot). Plot extracted Cdl from EIS against the CV-derived Cdl.
Protocol 2: Post-Mortem Analysis Driven ECM Parameter Validation
Objective: To physically identify the root cause of observed changes in ECM parameters after cycle life testing.
Materials: (See "Scientist's Toolkit" below). Procedure:
- In-Situ ECM Parameter Tracking: For a set of identical cells (N≥3), perform periodic EIS measurements at a reference SOC (e.g., 50%) throughout extended cycling. Fit all spectra with a consistent ECM, creating a database of parameter evolution (e.g., RSEI(t), Rct(t)).
- Cell Selection & Disassembly: At End-of-Life (EOL) criteria (e.g., 80% capacity retention), select cells representing key degradation states. In an argon-filled glovebox (<1 ppm O₂/H₂O):
- Carefully disassemble the cell.
- Extract the electrodes, rinse with appropriate solvent (e.g., DMC for Li-ion), and dry.
- Sample Preparation for Ex-Situ Analysis:
- SEM: Cut small electrode samples and mount on stubs. Transfer using an air-tight transfer vessel.
- XPS: Cut samples and adhere to XPS holders. Use a vacuum-transfer module to avoid air exposure.
- Analysis & Correlation:
- SEM/EDX: Image electrode cross-sections and surfaces. Measure SEI/plating layer thickness. Correlate morphological changes (cracks, deposits) with increases in σw or RSEI.
- XPS: Perform depth profiling on electrode surfaces. Quantify atomic concentrations of SEI components (F, P, C). Correlate increases in specific components (e.g., LiF) with increases in R_SEI from the final EIS measurement.
- Compile findings into a table format as in Table 2.
Diagrams
Title: Workflow for Correlating EIS-ECM with Cyclic Voltammetry
Title: Post-Mortem Analysis and ECM Correlation Workflow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Correlative Studies
| Item | Function in Protocol | Example Product/Chemical |
|---|---|---|
| High-Precision Potentiostat/Galvanostat with EIS Module | Instrument for performing EIS and CV measurements. | Biologic VMP-3, Gamry Interface 5000. |
| Argon-Filled Glovebox | Provides inert environment for cell assembly, disassembly, and post-mortem sample handling to prevent air/moisture degradation. | MBraun Unilab or equivalent (O₂/H₂O < 1 ppm). |
| Electrolyte Solution | Medium for ion transport. Composition critically affects all ECM parameters. | 1M LiPF₆ in EC:EMC (3:7 wt%) for Li-ion studies. |
| Reference Electrodes | For 3-electrode cell setups to isolate anode/cathode EIS responses. | Lithium metal ring for Li-ion coin cell kits (e.g., from EL-CELL). |
| SEM Sample Transfer Vessel | Allows vacuum-secure transfer of air-sensitive electrode samples from glovebox to SEM chamber. | Leuco VTS or similar. |
| XPS Vacuum Transfer Module | Maintains ultra-high vacuum and prevents air exposure during transfer of samples for surface analysis. | Specific to XPS manufacturer (e.g., Thermo Fisher, Kratos). |
| ECM/Fitting Software | Software to model EIS data and extract quantitative parameters. | ZView (Scribner Associates), EC-Lab (BioLogic), or equivalent. |
The Role of Machine Learning in Enhancing and Complementing Traditional ECM Analysis
Traditional Equivalent Circuit Model (ECM) analysis of Electrochemical Impedance Spectroscopy (EIS) data for batteries is a well-established method for deconvolving complex electrochemical processes into discrete electrical components (e.g., resistors, capacitors, Warburg elements). However, this approach has inherent limitations: model selection is often subjective, it struggles with highly non-linear or coupled processes, and it may fail to capture the full complexity of real-world battery degradation.
Machine Learning (ML) introduces a paradigm shift, not by replacing ECM, but by augmenting it. ML algorithms can handle high-dimensional, non-linear data, identify patterns beyond human intuition, and provide predictive insights. The synergy between ECM and ML is creating more robust, accurate, and predictive models for battery state estimation (State-of-Charge, State-of-Health), fault diagnosis, and lifetime prediction.
Core Synergistic Applications:
- Automated ECM Topology Selection: ML classifiers (e.g., Random Forest, SVMs) can analyze raw EIS spectra to recommend the most probable ECM topology, reducing analyst bias.
- Parameter Initialization and Extraction: ML-based regression models (e.g., Neural Networks) can provide highly accurate initial guesses for ECM parameters, accelerating non-linear fitting routines and avoiding local minima.
- Beyond-Circuit Phenomena Modeling: ML models (e.g., Gaussian Process Regression) can model residuals or phenomena that the ECM cannot capture, such as complex diffusion behaviors or coupling between degradation modes.
- Fusion with Multi-Modal Data: ML excels at integrating EIS-derived ECM parameters with other data streams (e.g., cycling data, temperature, pressure) for holistic performance and health forecasting.
Table 1: Performance Comparison of Traditional ECM vs. ML-Augmented ECM for SOC/SOH Estimation
| Method | Application | Key Metric | Reported Performance | Reference Year | Advantage |
|---|---|---|---|---|---|
| Traditional ECM (Randles) | SOC Estimation | Mean Absolute Error (MAE) | 2.1 - 5.0% SOC | N/A (Baseline) | Physically interpretable, simple. |
| ML (CNN on EIS spectra) | SOC Estimation | MAE | 0.6 - 1.2% SOC | 2023 | Eliminates model selection, superior accuracy. |
| ECM with Genetic Algorithm | Parameter Extraction | Fitting Convergence Time | 30-50% faster | 2022 | Better global optimum search. |
| Gradient Boosting on ECM Params | SOH Prediction (RUL) | Root Mean Square Error (RMSE) | < 1.5% Capacity | 2024 | Integrates degradation dynamics. |
| Hybrid ECM-LSTM Model | Voltage Prediction | Voltage RMSE | ~10 mV | 2023 | Captures temporal dependencies. |
Table 2: Common ML Algorithms and Their Role in ECM Enhancement
| Algorithm Type | Example Algorithms | Primary Role in ECM Workflow | Typical Input Data | Output |
|---|---|---|---|---|
| Unsupervised Learning | k-Means, PCA | EIS Data Clustering, DRT pre-processing | Raw EIS spectra (Zreal, Zimag) | Data clusters, reduced dimensions |
| Supervised Learning | Random Forest, SVM | ECM Topology Classification | EIS features or full spectra | Recommended ECM circuit |
| Regression | ANN, Gaussian Process | ECM Parameter Prediction, SOH estimation | EIS data + operational conditions | R, C, W values; SOH% |
| Hybrid/Sequential | GA + Levenberg-Marquardt | Parameter Optimization | Initial ECM parameters | Optimized, physically-plausible parameters |
| Deep Learning | CNN, LSTM | Direct End-to-End Prediction | Sequential EIS or cycling data | SOC, SOH, RUL |
Experimental Protocols
Protocol 1: ML-Assisted ECM Topology Selection and Parameter Extraction
Objective: To systematically select an appropriate ECM for a Li-ion battery EIS dataset and extract its parameters using a hybrid ML-nonlinear least squares (NLLS) approach.
Materials: (See Scientist's Toolkit below).
Procedure:
- EIS Data Acquisition & Preprocessing:
- Perform EIS measurements on the battery cell across the desired SOC and SOH range. Typical range: 10 kHz to 10 mHz, 10 mV perturbation.
- Clean data: remove inductive tails at high frequency, ensure Kramers-Kronig validity.
- Normalize impedance data if using data from multiple cells.
Feature Engineering for ML:
- Extract both heuristic features (e.g., real impedance at 1 kHz, low-frequency slope, characteristic frequency of semicircle) and transform-based features (e.g., coefficients from a Discrete Fourier Transform).
- Assemble a feature vector for each EIS spectrum.
Automated Topology Selection:
- Train a multi-class classifier (e.g., Random Forest) on a labeled dataset where the "ground truth" ECM for each spectrum is known from expert analysis.
- Input the feature vector of a new spectrum into the trained classifier.
- The classifier outputs a probability for candidate ECMs (e.g., Randles, Randles with 2 time constants, etc.). Select the topology with the highest probability.
Hybrid Parameter Extraction:
- ML Initialization: Use a pre-trained Neural Network regression model (trained on [EIS features] -> [ECM parameters]) to predict initial parameter values (R1, R2, CPE-Y, etc.) for the selected topology.
- Refinement via NLLS: Feed the ML-predicted parameters as the initial guess into a standard NLLS fitting algorithm (e.g., Levenberg-Marquardt).
- Apply appropriate physical constraints (e.g., all resistances > 0) during NLLS.
- Validate the final fitted parameters by comparing the reconstructed spectrum to the original data (calculate NRMSE).
Protocol 2: Building a Hybrid ECM-ML Model for State-of-Health Prediction
Objective: To create a predictive model for battery Remaining Useful Life (RUL) by fusing ECM parameters with operational data using a sequential ML model.
Procedure:
- Longitudinal Data Collection:
- Subject battery cells to cyclic aging tests under varied conditions (temperature, C-rate, DOD).
- At regular intervals (e.g., every 25 cycles), pause cycling and perform EIS measurements and reference performance tests (RPT) for capacity/energy fade (true SOH).
- Record operational metadata (cycle number, average temperature, depth of discharge history).
ECM Parameter Extraction per Interval:
- For each EIS measurement in the aging series, fit a consistent, physically-relevant ECM (e.g., one with a charge-transfer and a solid-state diffusion element).
- Extract the time-series of each parameter (e.g., Rohm, Rct, CPE-diffusion).
Dataset Construction for ML:
- Create a sequential dataset where each sample is a window of the last 'n' cycles (e.g., n=10).
- For each window, the features are: the ECM parameter trends within that window and the operational metadata.
- The target label is the capacity fade (SOH) or RUL at the end of a prediction horizon (e.g., 50 cycles after the window).
Model Training and Validation:
- Use a sequence-aware model like an LSTM or 1D-CNN to train on the constructed dataset.
- Perform time-series cross-validation to avoid data leakage.
- The model learns the complex mapping from the evolution of ECM parameters (degradation modes) to future SOH.
Visualization: Workflows and Relationships
Diagram Title: ML-Augmented ECM Analysis Workflow
Diagram Title: Multi-Modal Data Fusion for Battery Prognostics
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Materials and Tools for ML-Enhanced ECM Research
| Item / Solution | Function / Purpose in ML-ECM Research | Example/Note |
|---|---|---|
| Potentiostat/Galvanostat with EIS | Acquires the fundamental impedance spectrum data. Requires low-current capability and wide frequency range for batteries. | Biologic VSP-300, Gamry Interface 5000. |
| Battery Cycler with Environmental Chamber | Provides controlled aging and testing conditions to generate the necessary datasets for model training. | Arbin LBT, Neware Cycler, with thermal chamber. |
| EIS Data Analysis Software | Performs initial visualization, Kramers-Kronig testing, and traditional ECM fitting. Acts as a baseline and data exporter. | EC-Lab (Biologic), Gamry Echem Analyst, ZView. |
| Python/R with Scientific Libraries | The core platform for developing and deploying ML models, custom data processing, and hybrid analysis scripts. | Python: scikit-learn, TensorFlow/PyTorch, SciPy, NumPy, Pandas, Matplotlib. |
| DRT (Distribution of Relaxation Times) Tool | A complementary model-free analysis method used to inform ECM topology selection and generate features for ML. | DRTtools (Python), BEIS (Matlab). |
| High-Performance Computing (HPC) / GPU | Accelerates the training of complex ML models (especially deep learning) and hyperparameter optimization. | Cloud platforms (Google Colab Pro, AWS) or local GPU workstations. |
| Curated Public/Private Battery Datasets | Provides essential training and benchmarking data, especially for teams lacking resources for large-scale testing. | NASA PCoE Dataset, CALCE Battery Data. |
| Version Control System | Manages code, model architectures, and hyperparameters to ensure reproducibility of ML-ECM research. | Git (GitHub, GitLab). |
Conclusion
Equivalent Circuit Modeling for EIS remains an indispensable, accessible tool for translating the complex electrochemical language of batteries into actionable insights for researchers. Mastering the foundational principles, meticulous application, rigorous troubleshooting, and robust validation outlined in this guide transforms impedance data into a powerful diagnostic for SOH, degradation mechanisms, and performance limits. Future directions point toward tighter integration with physics-based models, automated model selection via machine learning, and the development of standardized ECM protocols for emerging chemistries like solid-state and sodium-ion batteries. This continued evolution will further solidify EIS-ECM as a cornerstone methodology in the quest for more reliable, high-performance energy storage solutions.